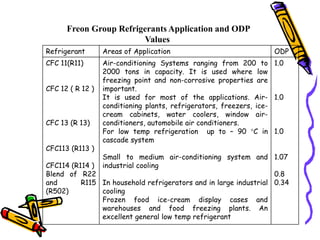
- Refrigerants have evolved over time from early experiments using ether to modern halocarbon refrigerants like Freon. Environmental concerns have led to a shift away from ozone depleting substances.
- The ideal refrigerant has favorable thermodynamic properties, low environmental impact, and is economical. Key criteria include pressures, heat transfer rates, toxicity, and contribution to global warming. Natural refrigerants like ammonia and hydrocarbons are gaining acceptance as sustainable alternatives.