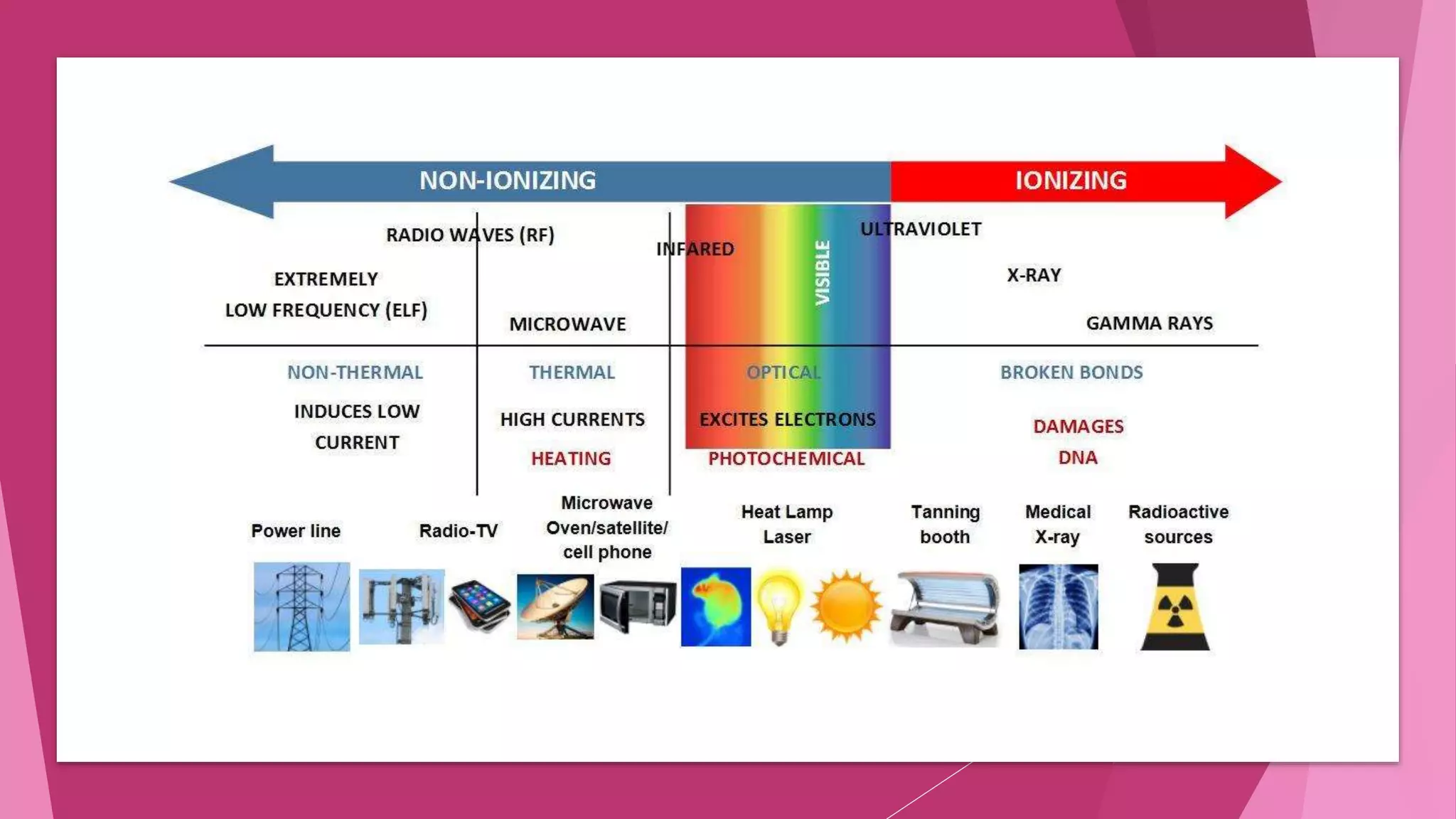
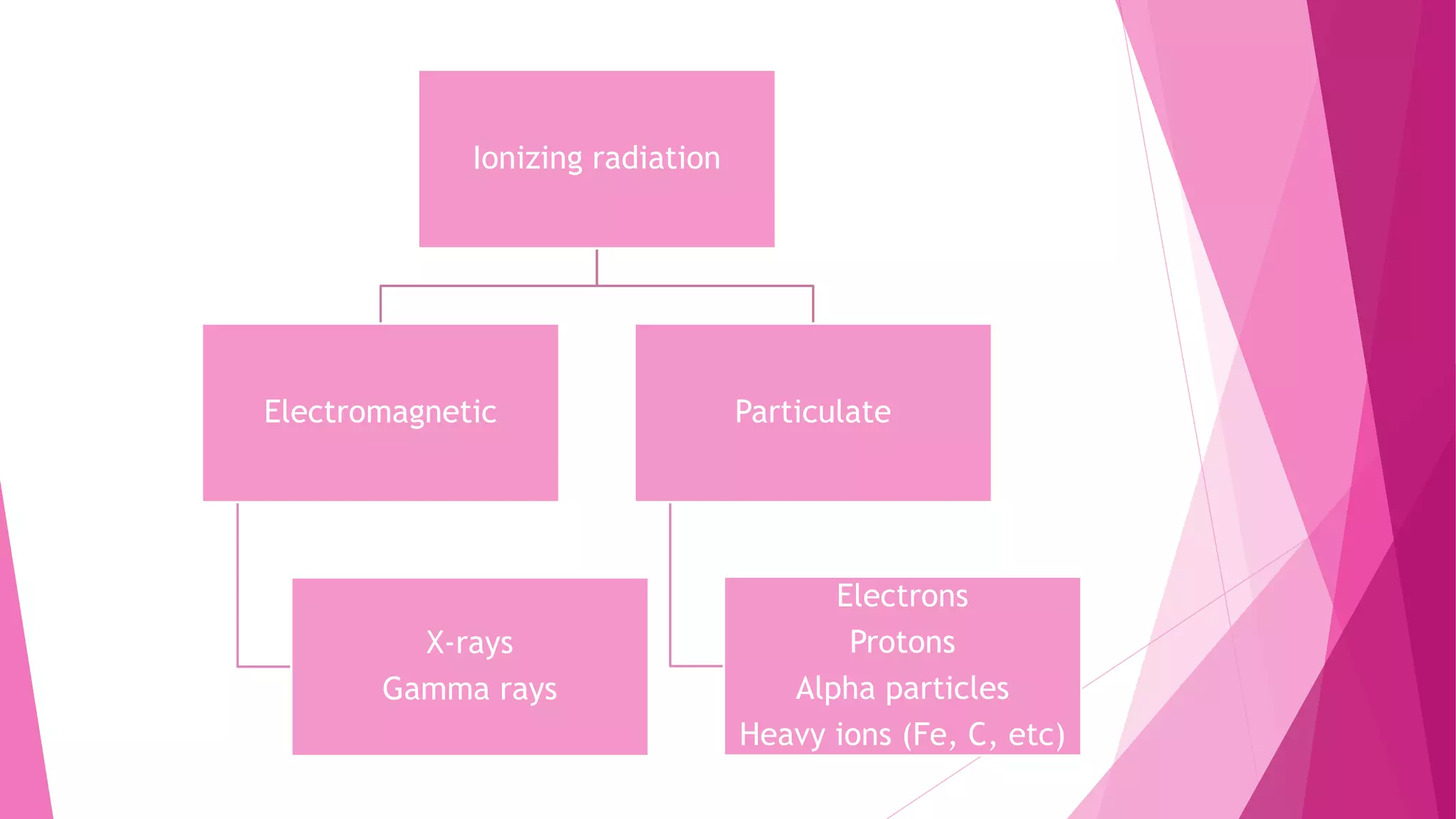
Radiobiology is the study of how ionizing radiation interacts with living things. This document provides an overview of different types of ionizing radiation including electromagnetic radiation like x-rays and gamma rays, and particulate radiation like electrons, protons, alpha particles, and heavy ions. It describes the physics of how this radiation is absorbed and can cause excitation or ionization in biological materials. Specifically, it notes that x-rays and gamma rays are indirectly ionizing since they produce fast-moving electrons upon absorption, while particulate radiations can directly ionize materials. Around 10,000 to 20,000 cases of lung cancer per year in the US are attributed to alpha particles from radon gas in homes.