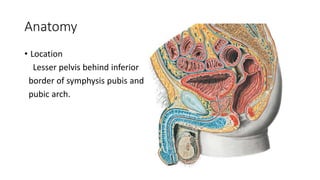
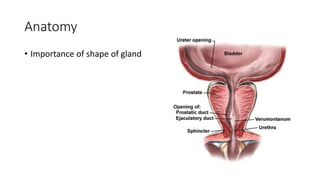
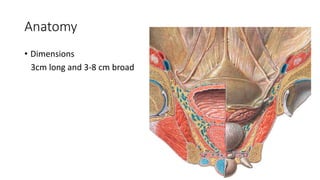
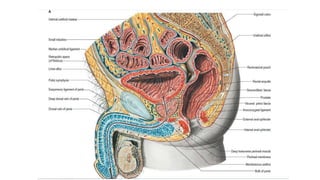
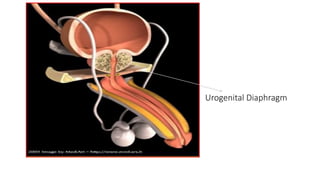
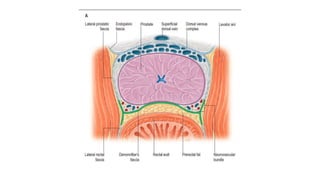
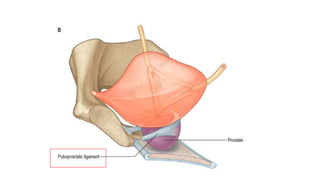
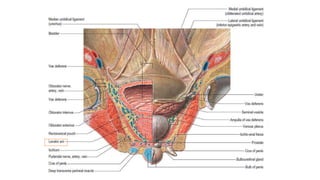
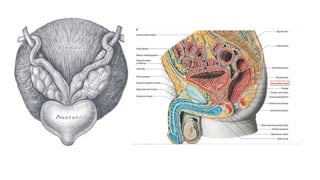
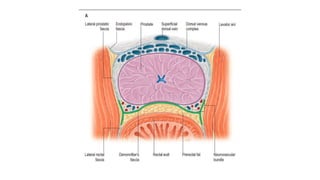
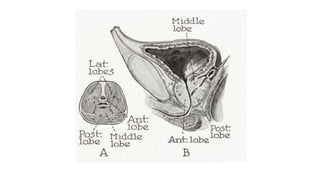
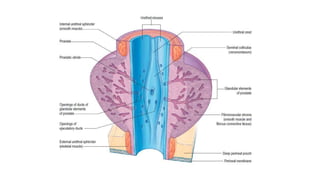
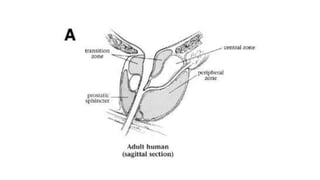
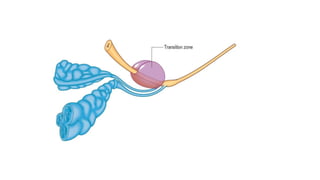
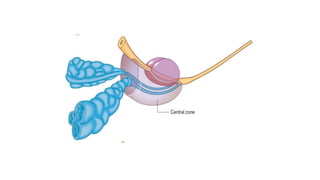
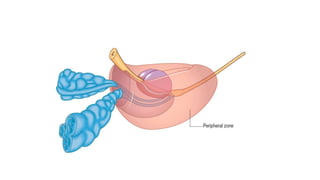
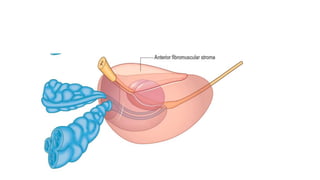
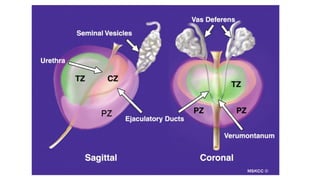
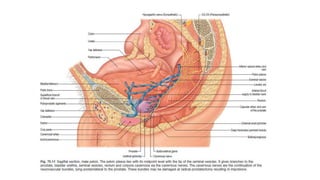
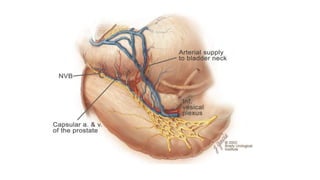
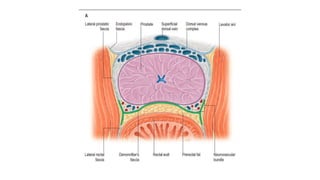
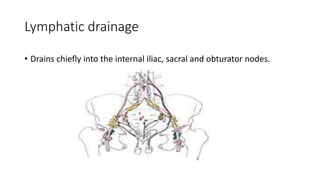
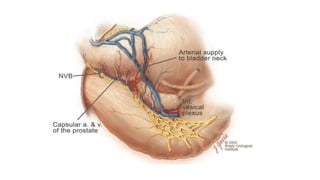
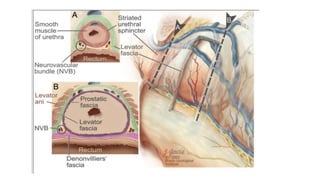
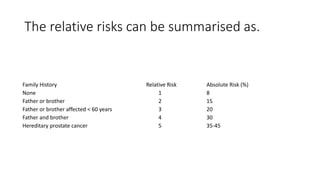
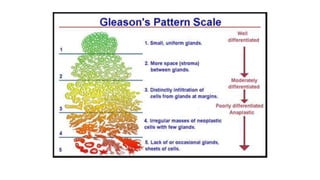
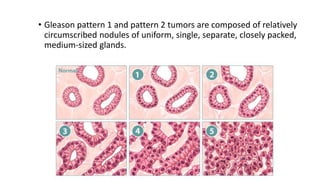
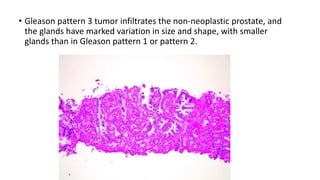
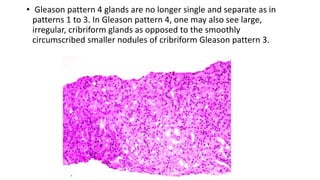
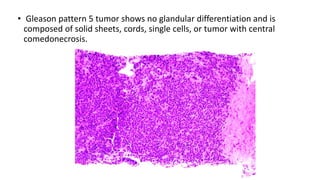
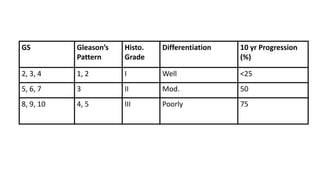
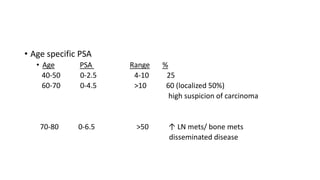
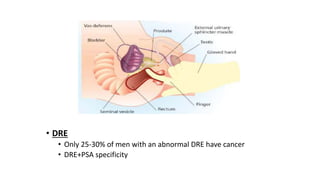
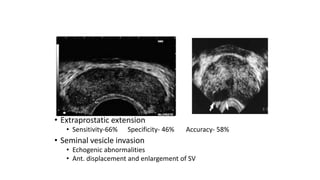
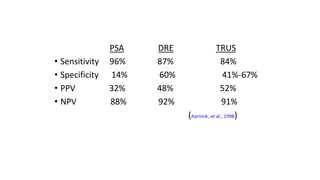
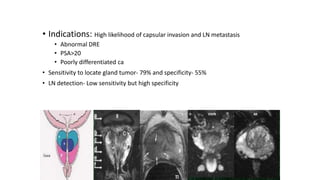
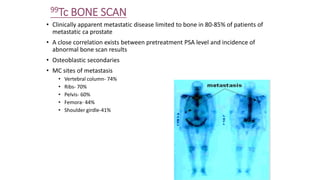
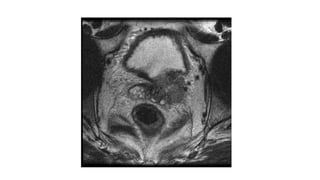
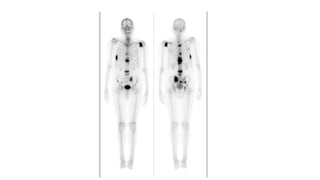
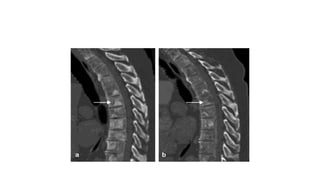
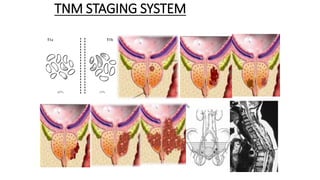
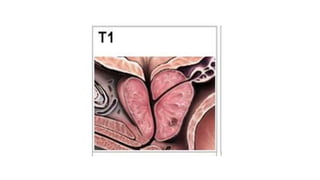
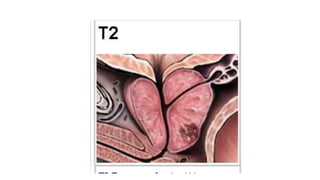
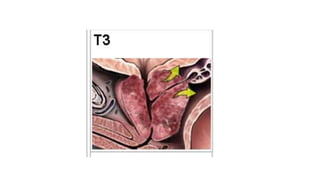
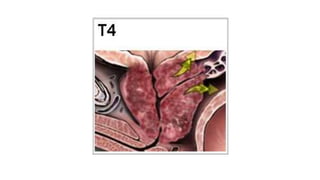
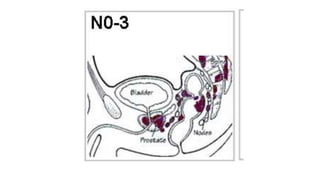
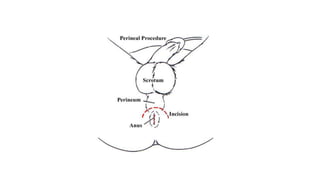
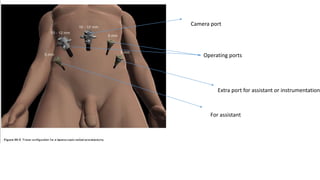
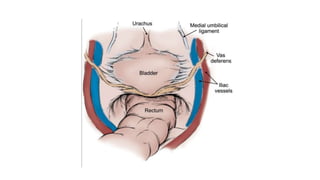
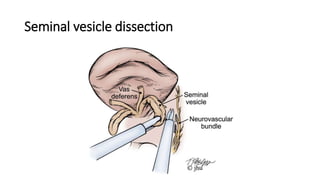
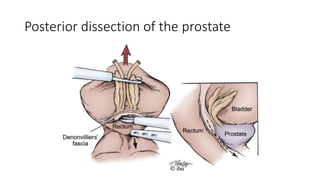
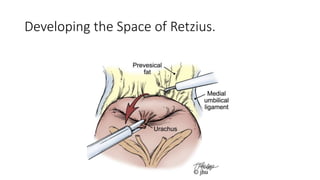
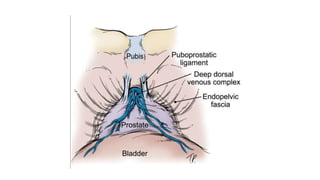
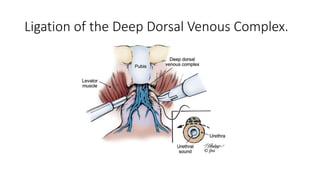
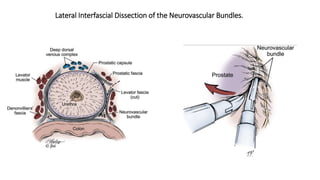
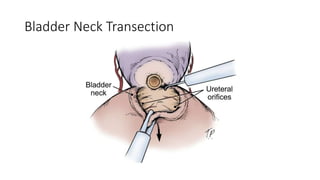
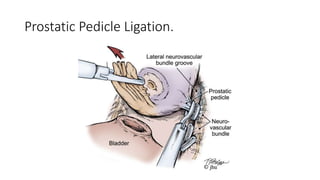
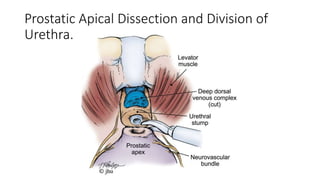
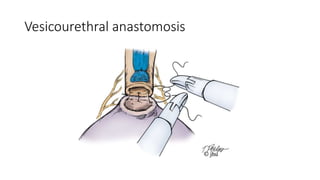
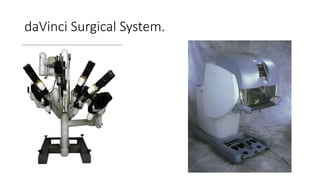
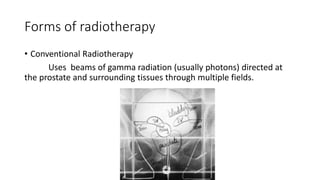
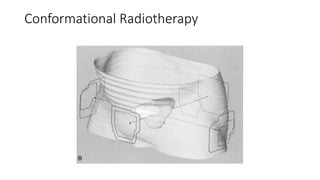
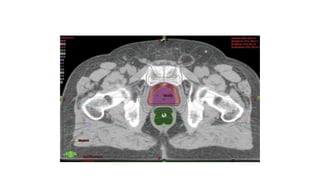
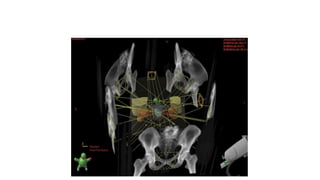
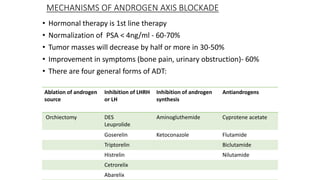
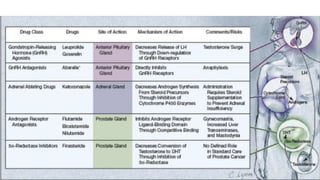
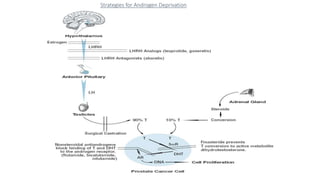
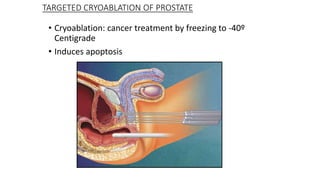
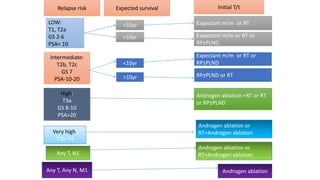
Prostate carcinoma is the most common cancer in men. It typically arises from the peripheral zone and spreads along the prostatic capsule. Diagnosis is usually made in the late 60s and treatment includes surgery, radiation, hormone therapy or active surveillance depending on staging. Having a family history, genetic factors, chronic inflammation and hormonal imbalances can increase risk.