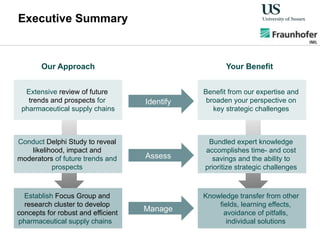
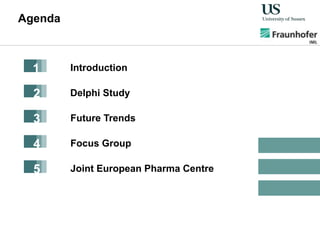
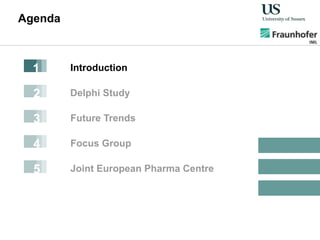
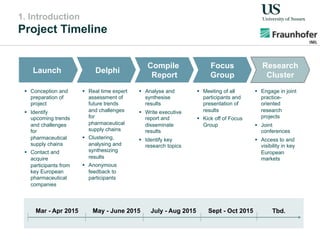
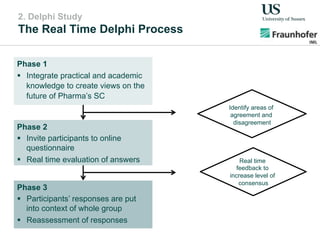
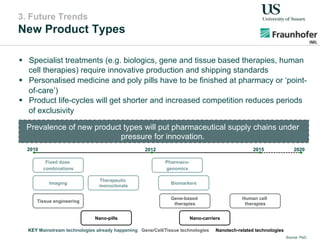
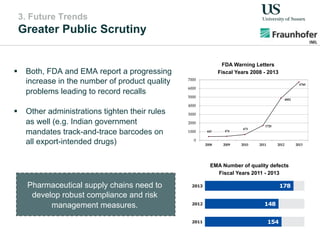
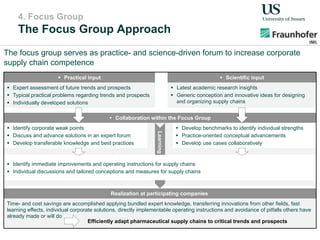
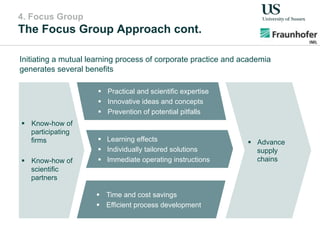
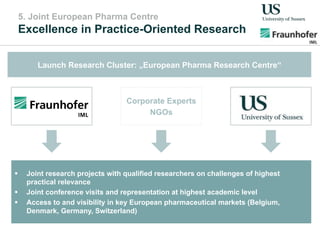
The document outlines a Delphi-based expert assessment focusing on future challenges for pharmaceutical supply chains, highlighting critical trends such as new product types, healthcare delivery methods, and emerging market opportunities. The study aims to gather insights from industry experts to develop effective strategies for managing complex supply chain dynamics and regulatory changes. Additionally, it emphasizes the importance of a collaborative approach among stakeholders to enhance performance and adapt to evolving market needs.