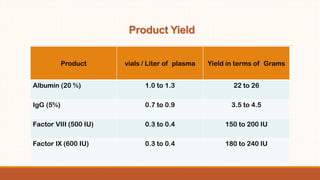
The document discusses establishing a human plasma fractionation project in Pakistan. It recommends a pilot contract fractionation project to make Pakistan self-sufficient in plasma derived medicines by pooling donated plasma, purifying it, and processing it into useful products. Contract fractionation would allow Pakistan to save on infrastructure costs by sending plasma abroad for fractionation and returning the finished products. A minimum of 30,000-50,000 liters of plasma is required annually to make contract fractionation cost-effective. The document also provides details on fractionation plant capacity, projected yields of various plasma products, capital costs, and timelines for developing a domestic fractionation capability.






















![lasma fractionation process: unique technology.
Adapted from reference [2] and [16].](https://image.slidesharecdn.com/plasmafractionationmaster-221203014325-5dc5f3a0/85/Plasma-Fractionation-Master-pptx-23-320.jpg)
![Plasma requirement (million liters) for albumin, factor and
intravenous immunoglobulin between 1974 and 2014 (source:the
MarketingResearchBureauwithpermission).Adaptedfromreference [22].](https://image.slidesharecdn.com/plasmafractionationmaster-221203014325-5dc5f3a0/85/Plasma-Fractionation-Master-pptx-24-320.jpg)