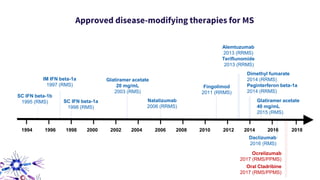
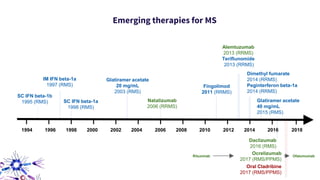
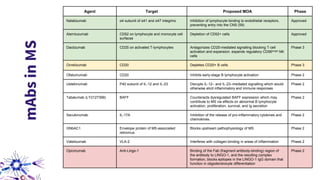
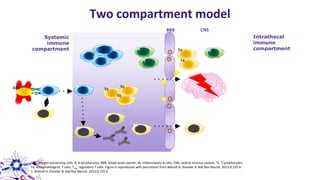
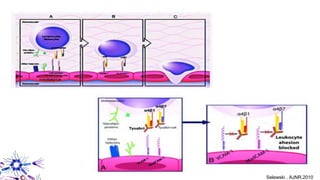
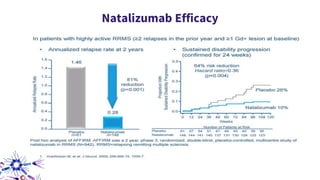
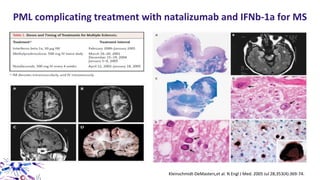
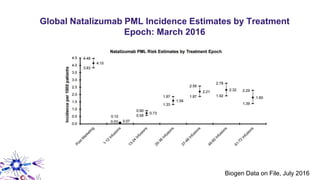
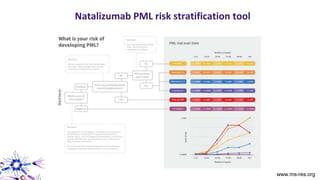
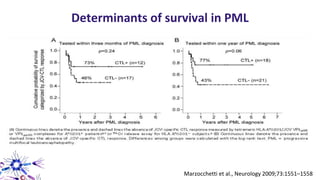
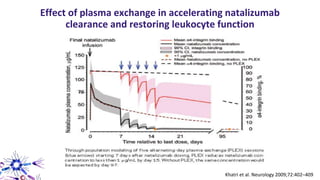
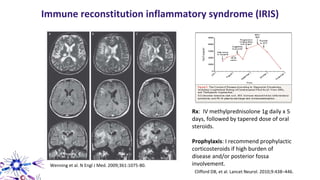
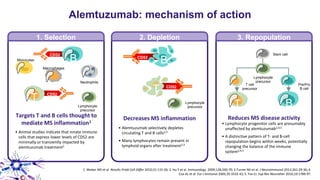
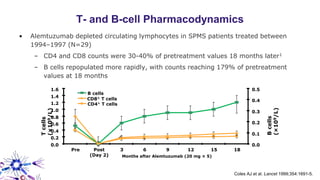
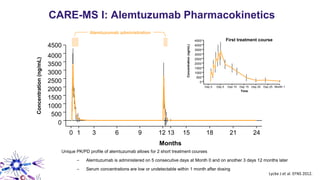
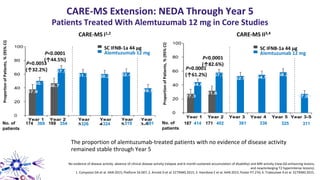
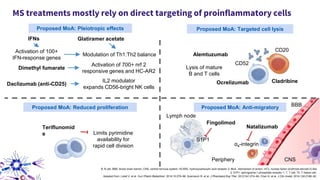
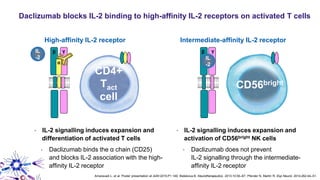
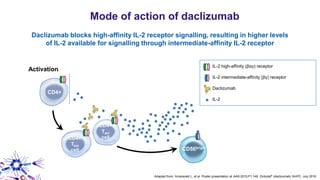
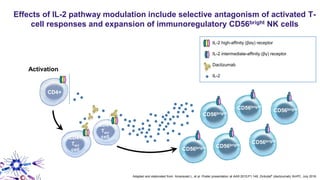
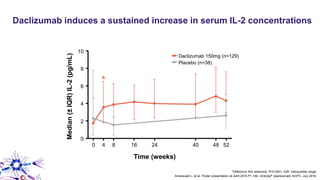
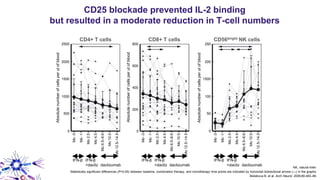
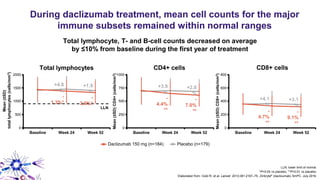
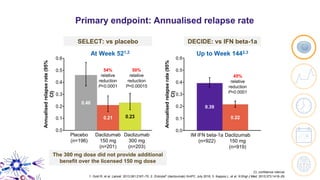
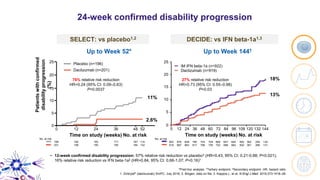
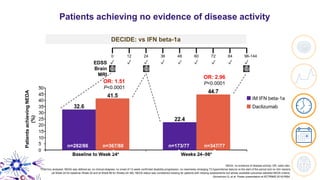
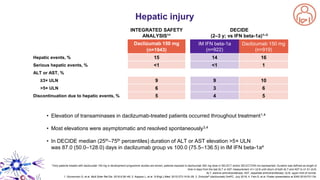
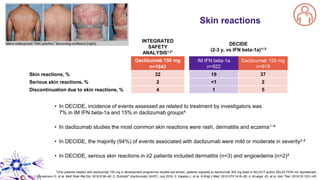
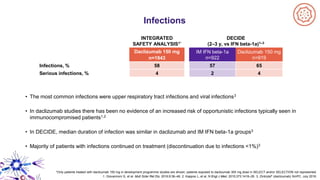
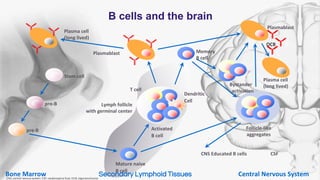
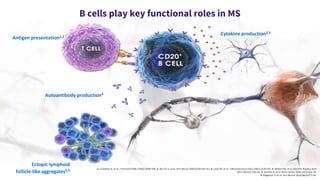
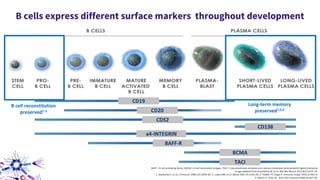
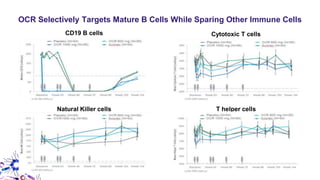
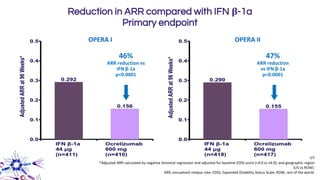
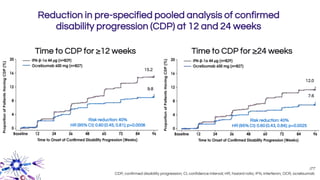
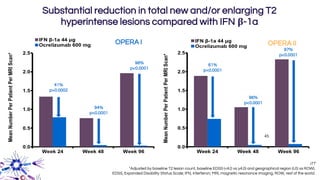
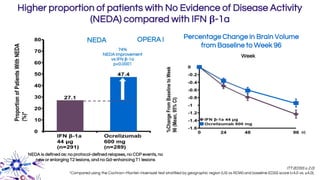
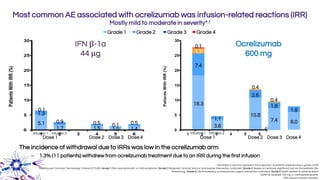
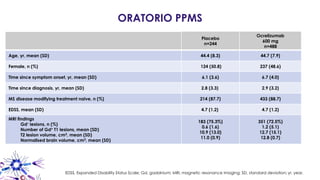
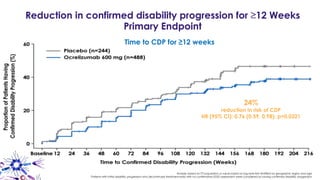
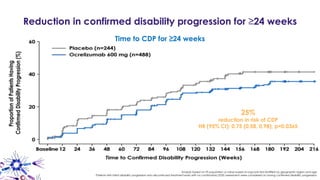
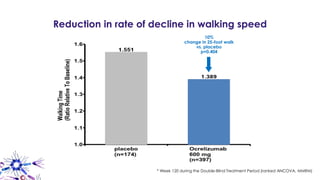
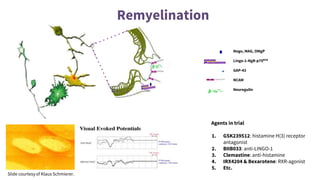
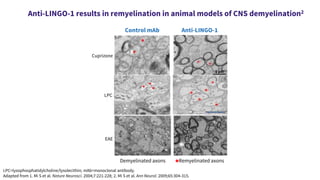
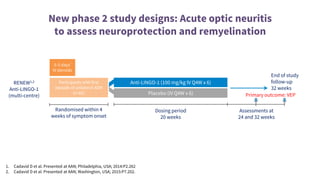
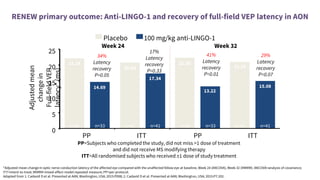
This document provides an overview of current and emerging antibody treatments for multiple sclerosis (MS). It discusses approved therapies like natalizumab, alemtuzumab, and daclizumab. Natalizumab works by inhibiting lymphocyte binding to endothelial cells. Alemtuzumab depletes CD52+ lymphocytes. Daclizumab antagonizes CD25-mediated T cell activation. Emerging therapies discussed include ofatumumab, ustekinumab, and secukinumab which target CD20, IL-12/IL-23, and IL-17A respectively. The document also reviews the mechanisms of action, efficacy data, safety issues like PML risk, and ongoing clinical trials of these antibody treatments for MS.