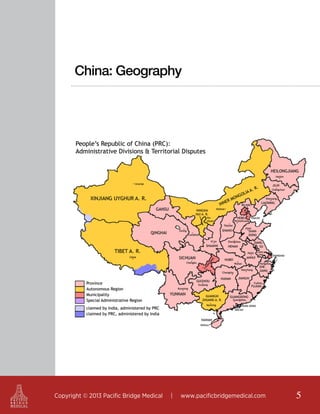
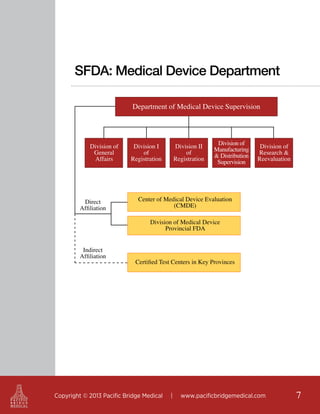
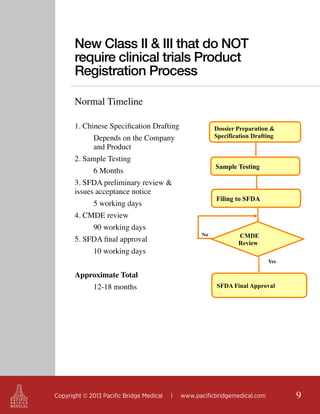
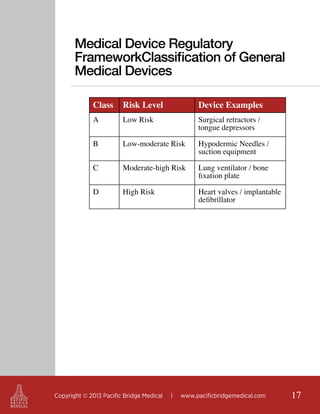
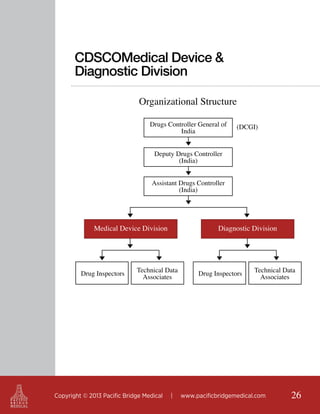
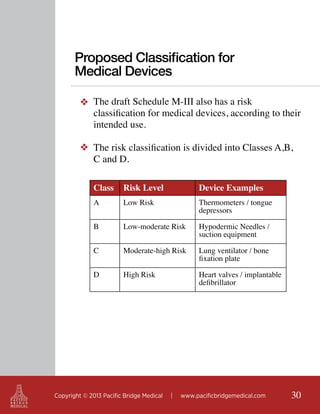
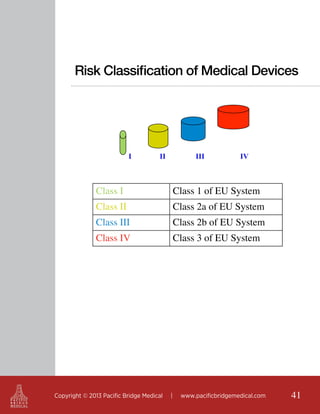
This document outlines regulatory strategies for medical device companies operating in the Asia-Pacific region, focusing on specific regulations in China, Singapore, India, and Hong Kong. It highlights key regulatory authorities, product classification, registration processes, and the importance of compliance with local laws. The white paper offers insights into market dynamics and the required documentation for successful navigation in these diverse regulatory landscapes.