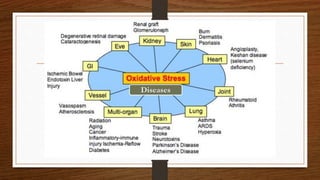
The document discusses oxidative stress and reactive oxygen species (ROS). It defines ROS and lists some examples like superoxide anion radical, hydroxyl radical, and hydrogen peroxide. It describes how ROS are produced endogenously through processes like mitochondrial electron transport, and exogenously through factors like pollution, radiation, and xenobiotics. The effects of ROS include DNA damage and modulation of signal transduction pathways. It also discusses antioxidants, dividing them into antioxidant enzymes like superoxide dismutase and catalase, chain breaking antioxidants like vitamins C and E, and transition metal binding proteins.