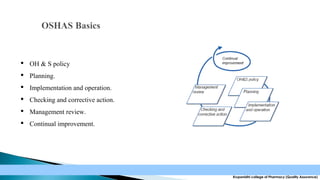
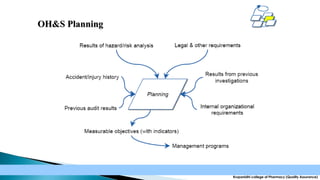
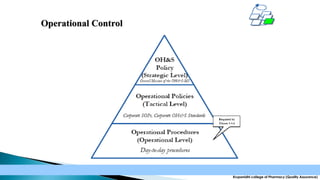
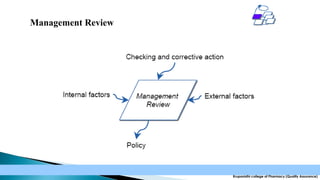
The document discusses the key elements of an Occupational Health and Safety Assessment Series (OHSAS) 18001 management system. It notes that OHSAS 18001 was originally developed in the early 1990s as British Standard 8800 and revised in 1999 to be more compatible with ISO 14001. The document then outlines the main components of an OHSAS 18001 system, including policy, planning, implementation, checking and corrective action, and management review.