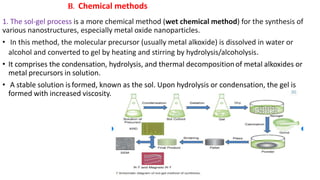
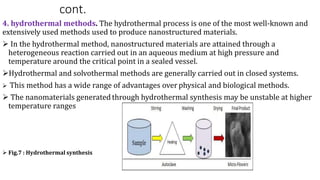
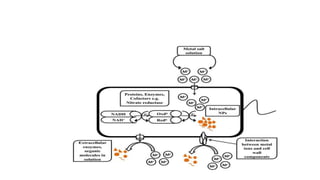
This document provides an overview of various methods for synthesizing nanoparticles. It discusses factors that affect nanoparticle synthesis and outlines two main approaches: top-down and bottom-up. For top-down, it mentions physical vapor deposition and chemical vapor deposition. For bottom-up, it describes methods like sol-gel synthesis, colloidal precipitation, and hydrothermal synthesis. It then examines specific physical, chemical, and biological synthesis methods in more detail like mechanical milling, pulsed laser ablation, and using microorganisms.