Embed presentation
Download to read offline



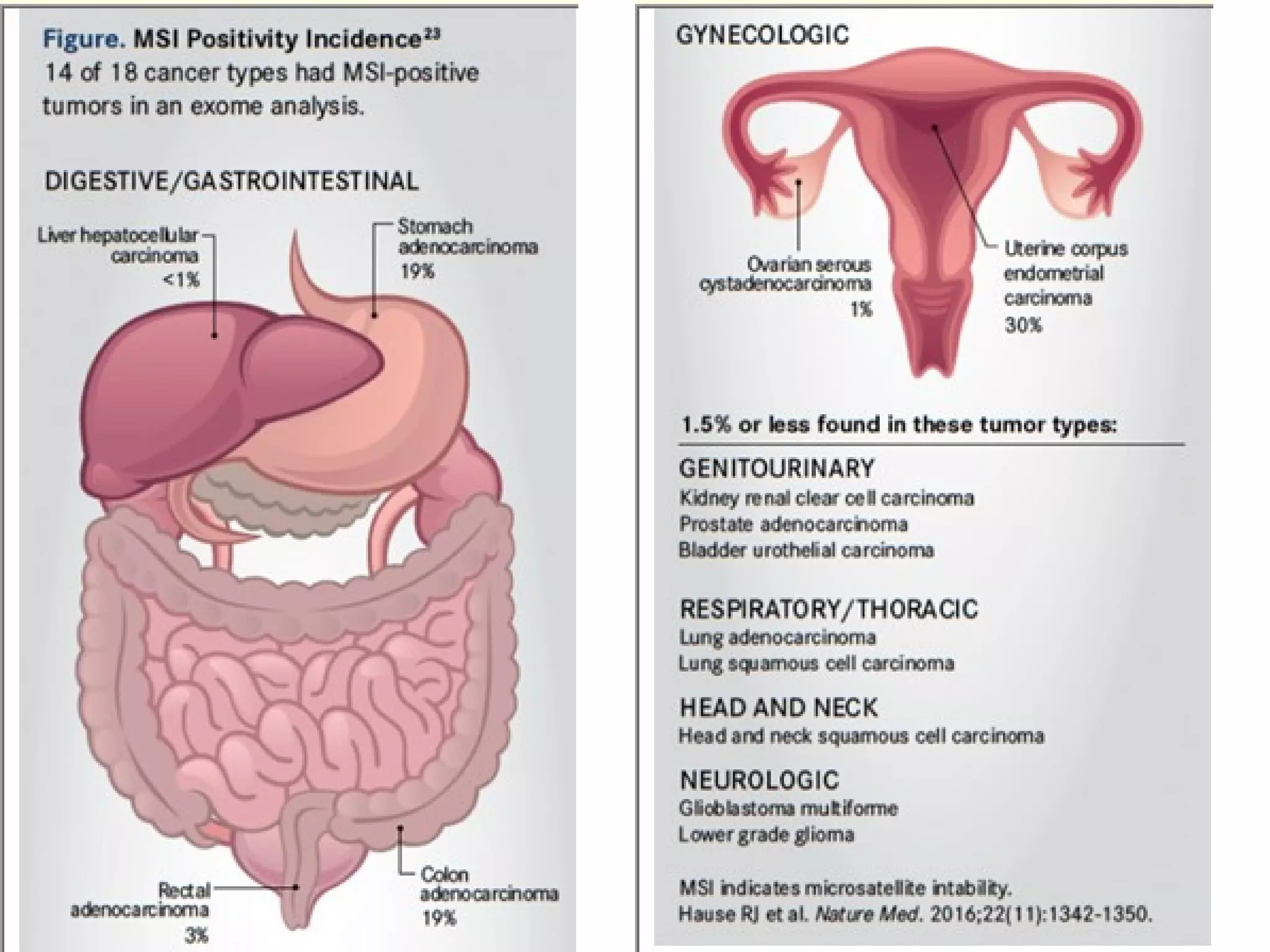
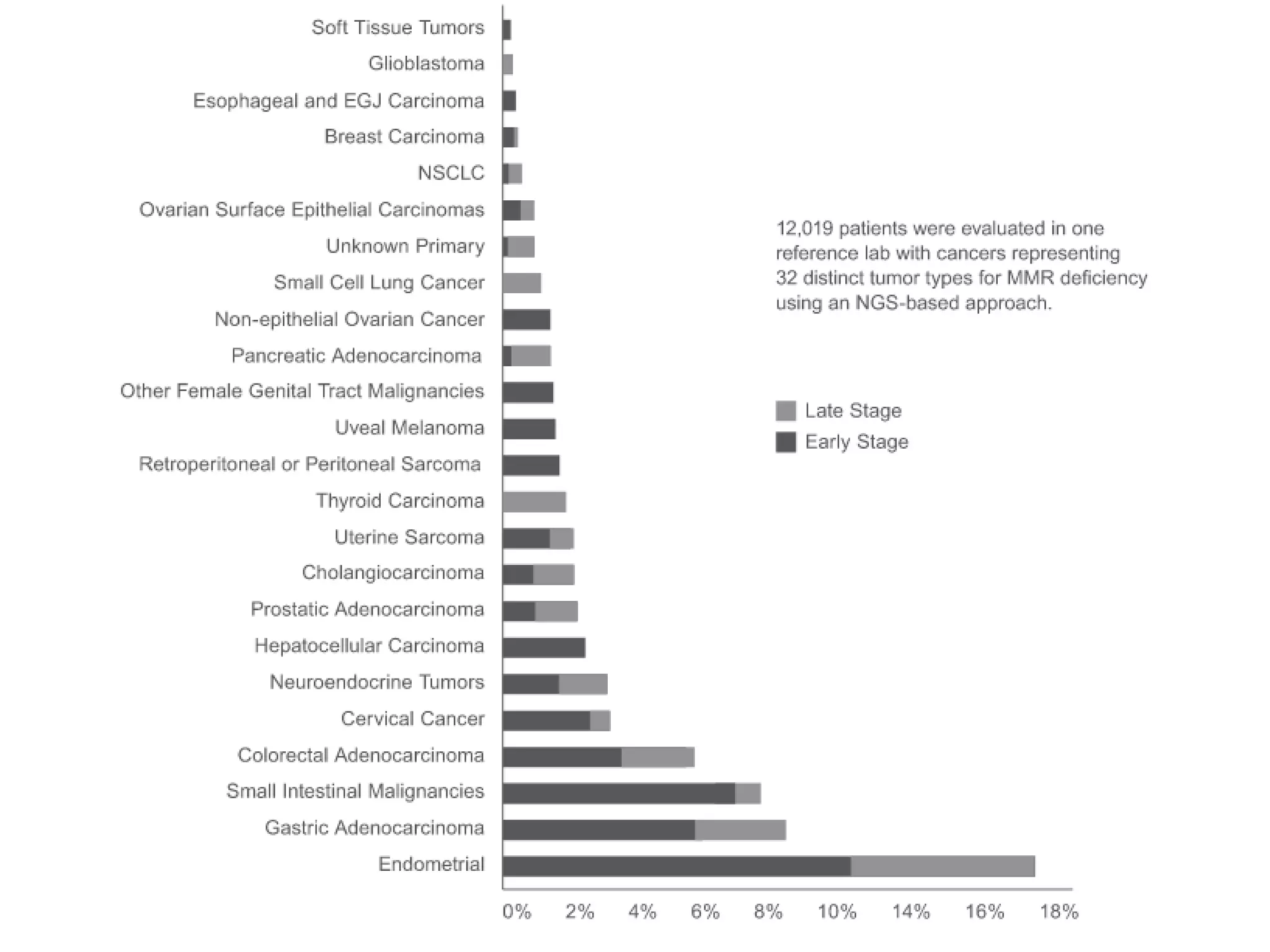

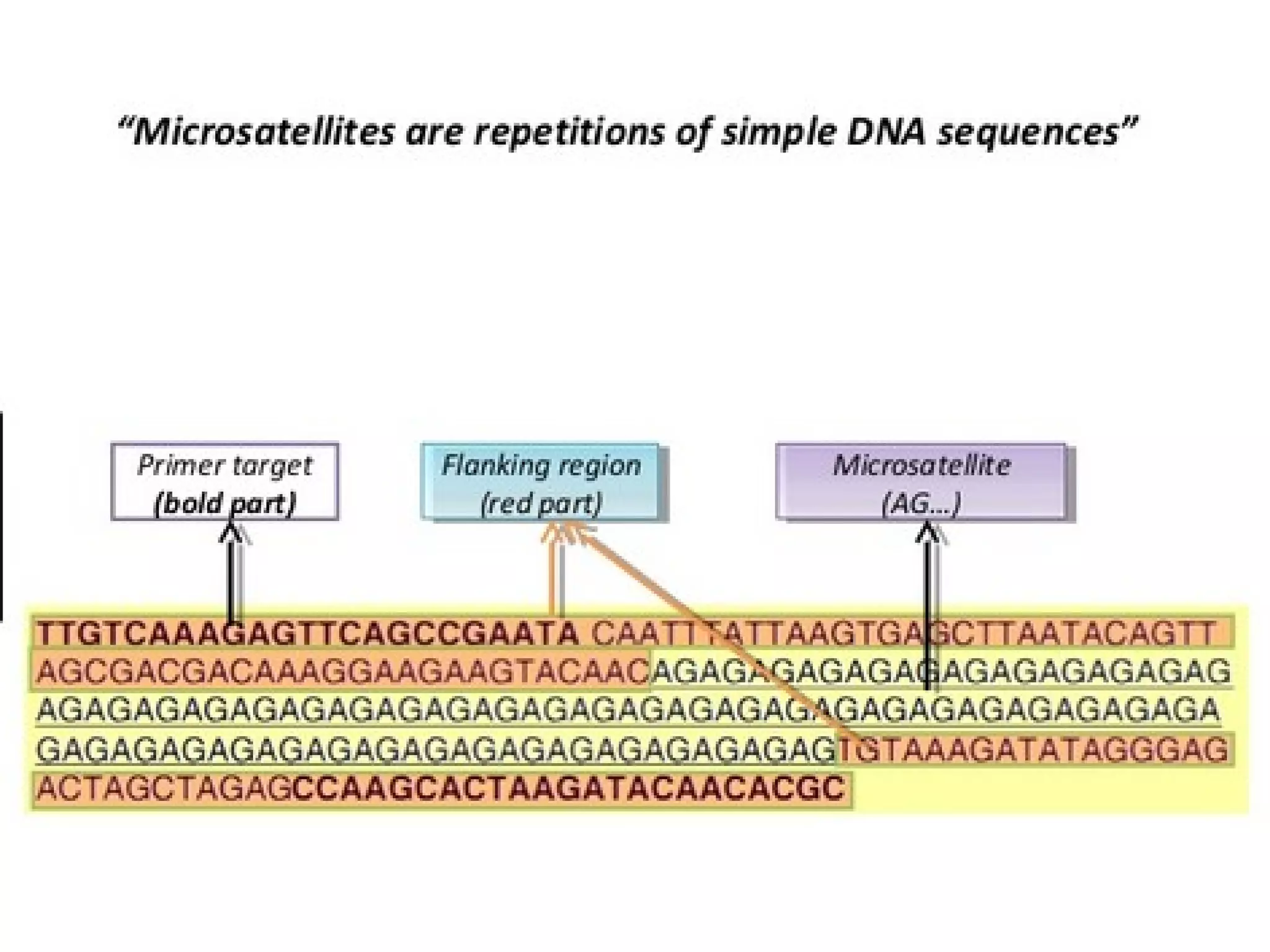

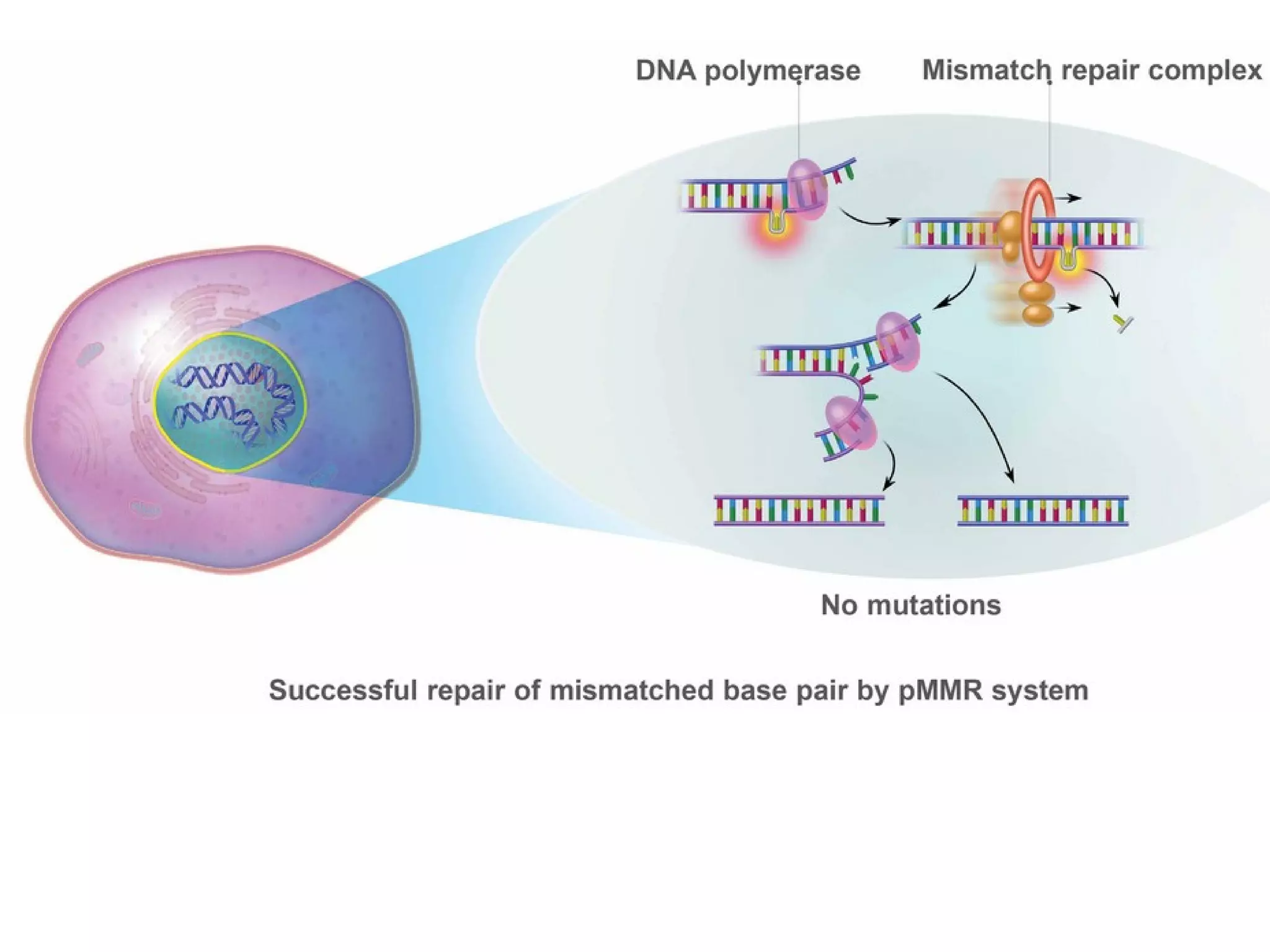
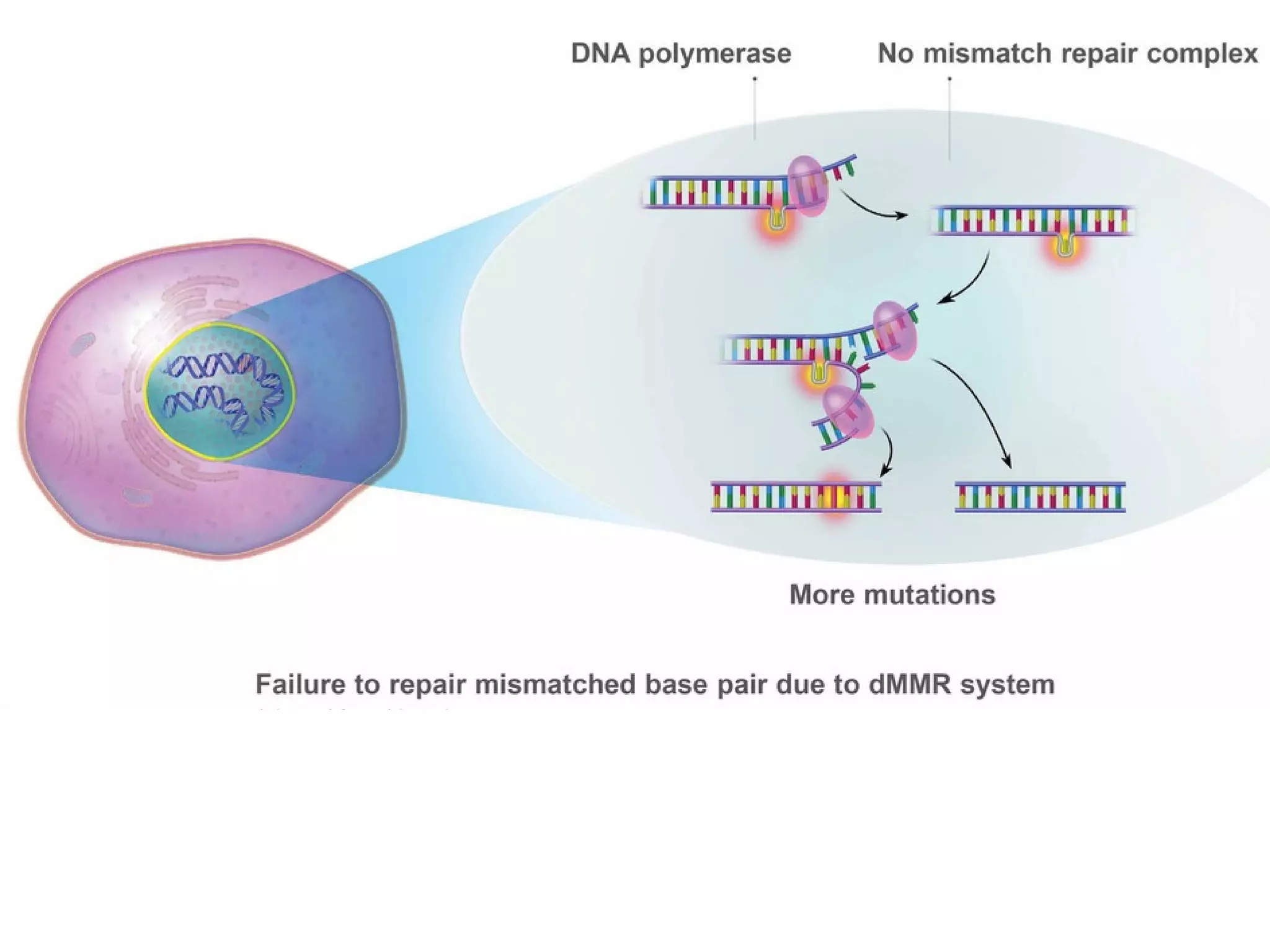
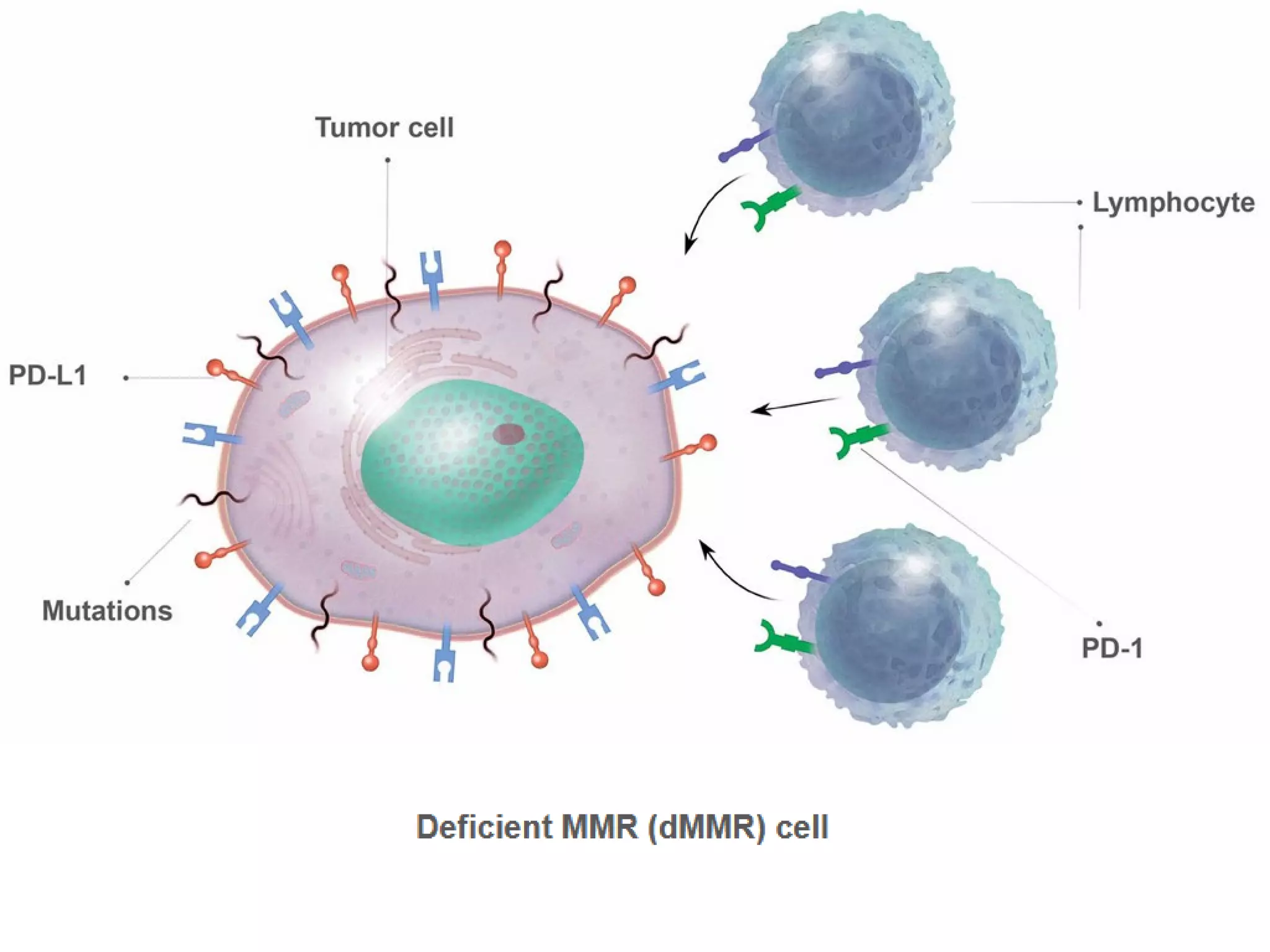
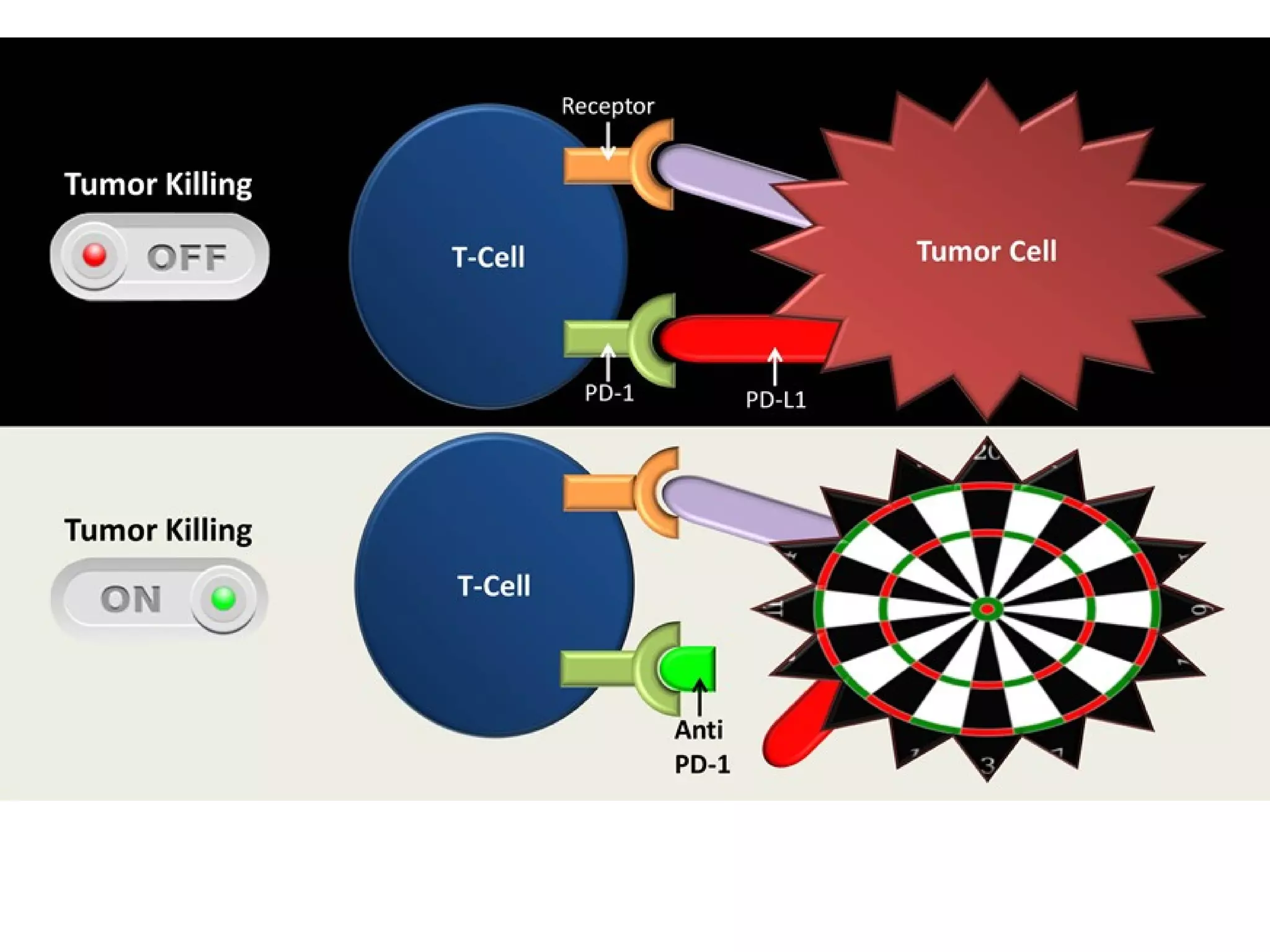

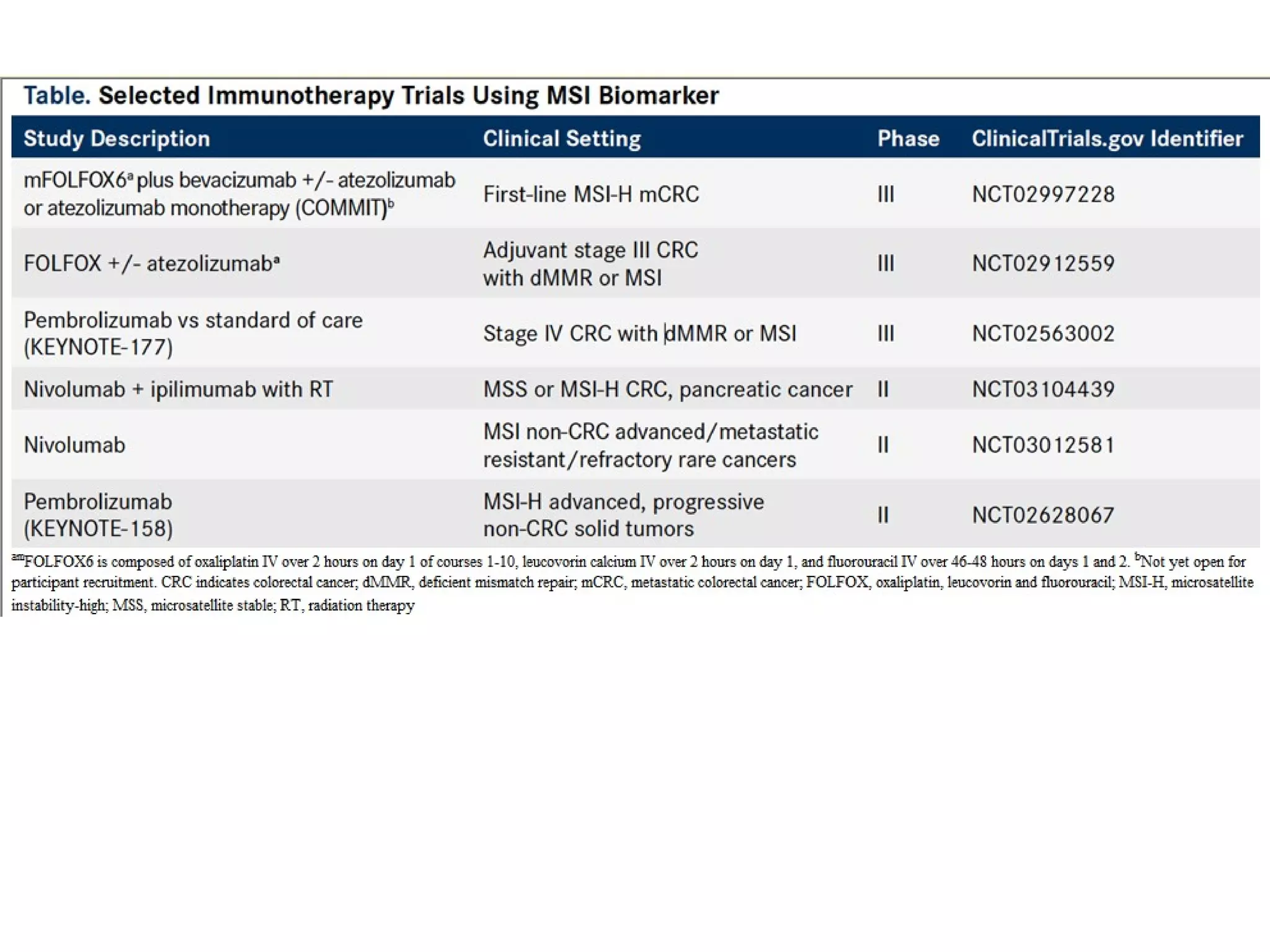
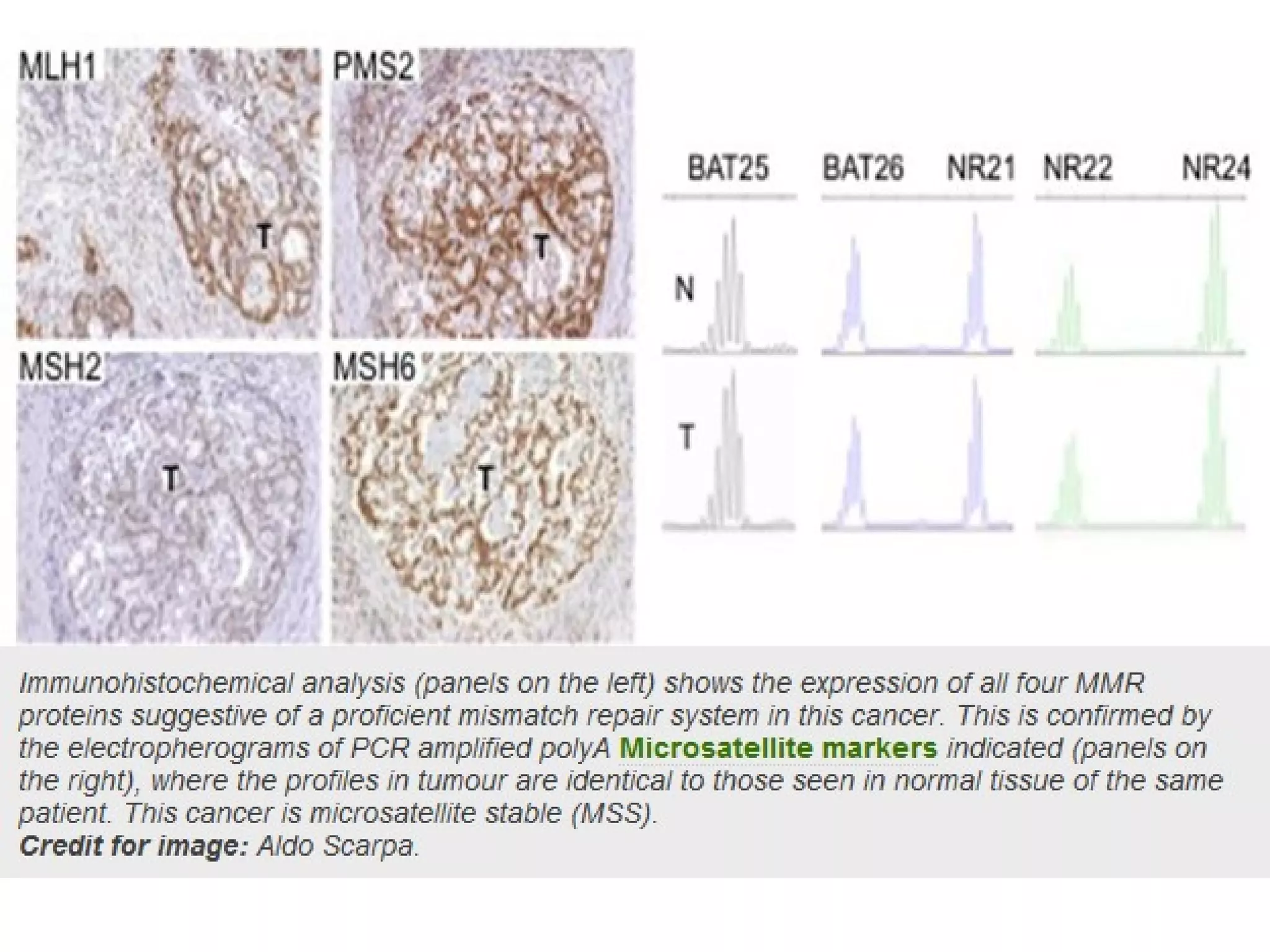
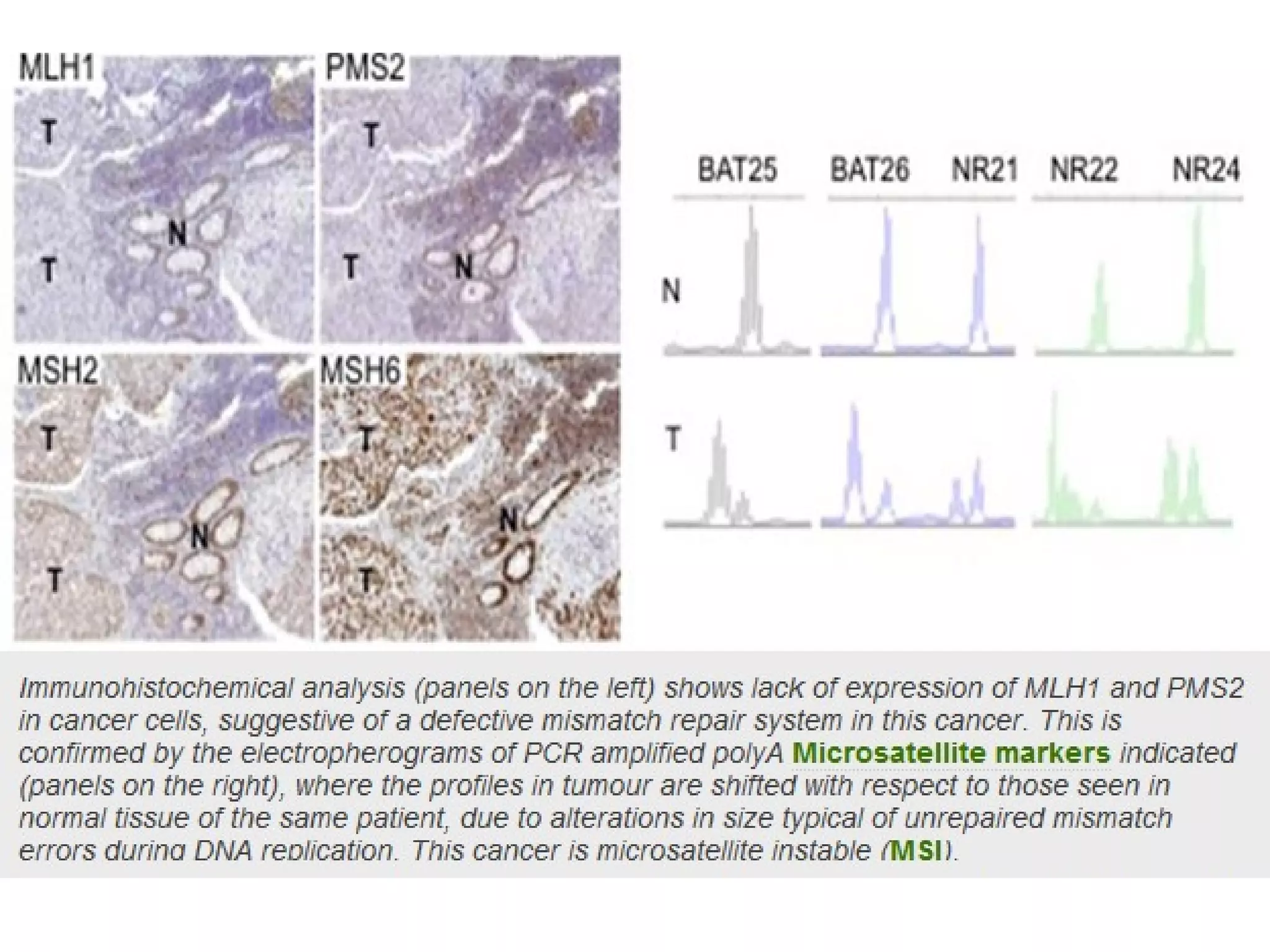

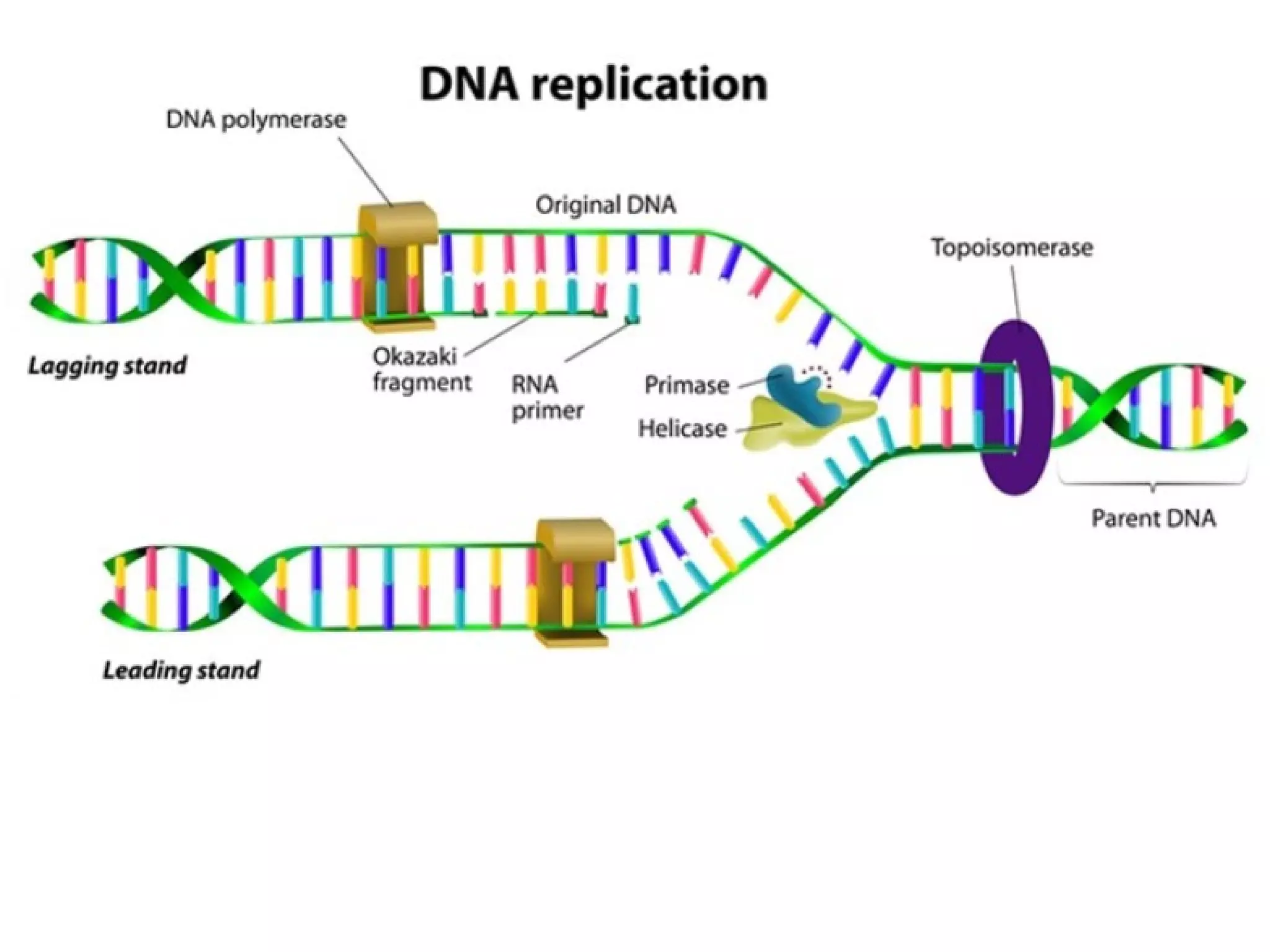
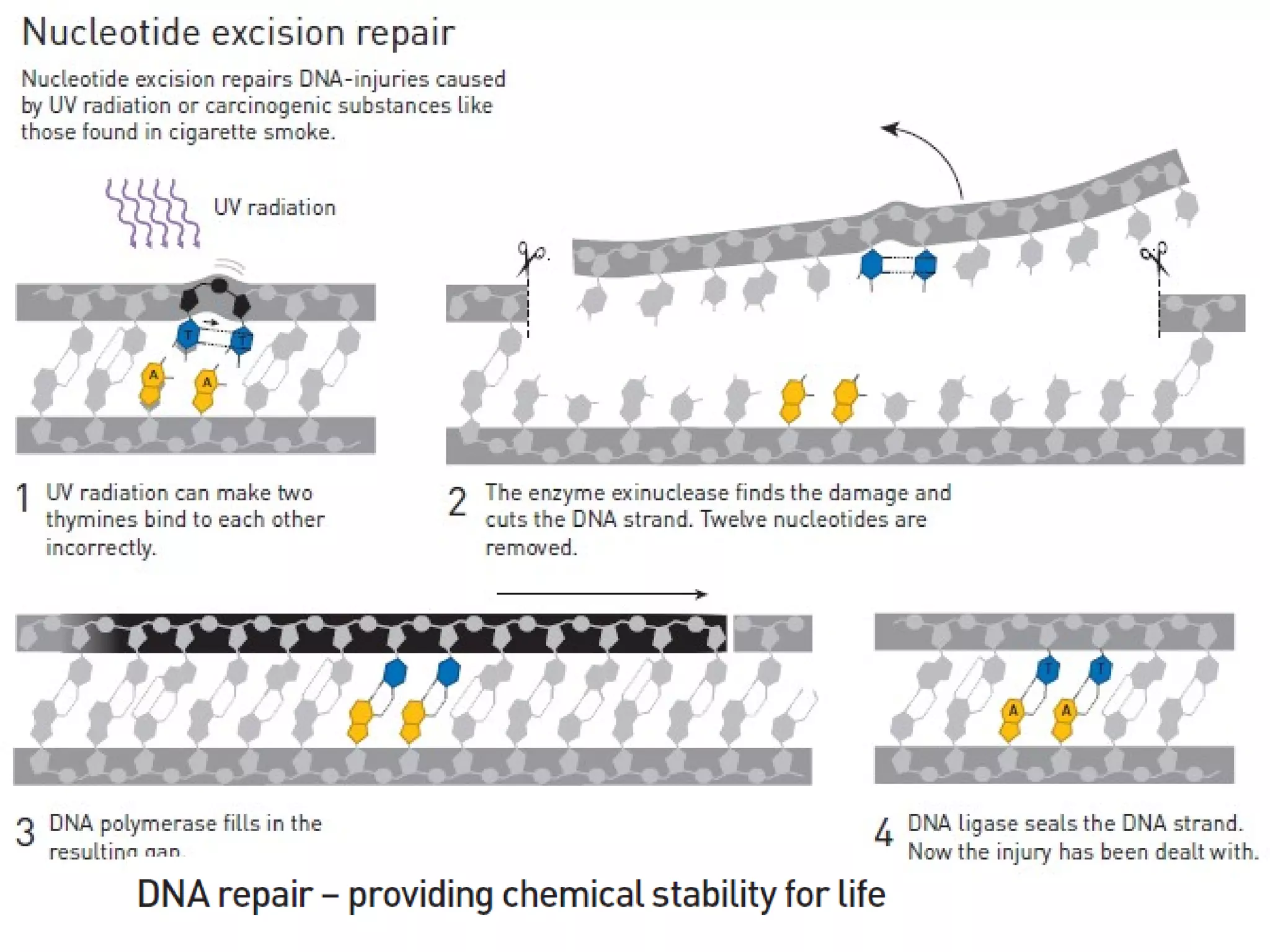
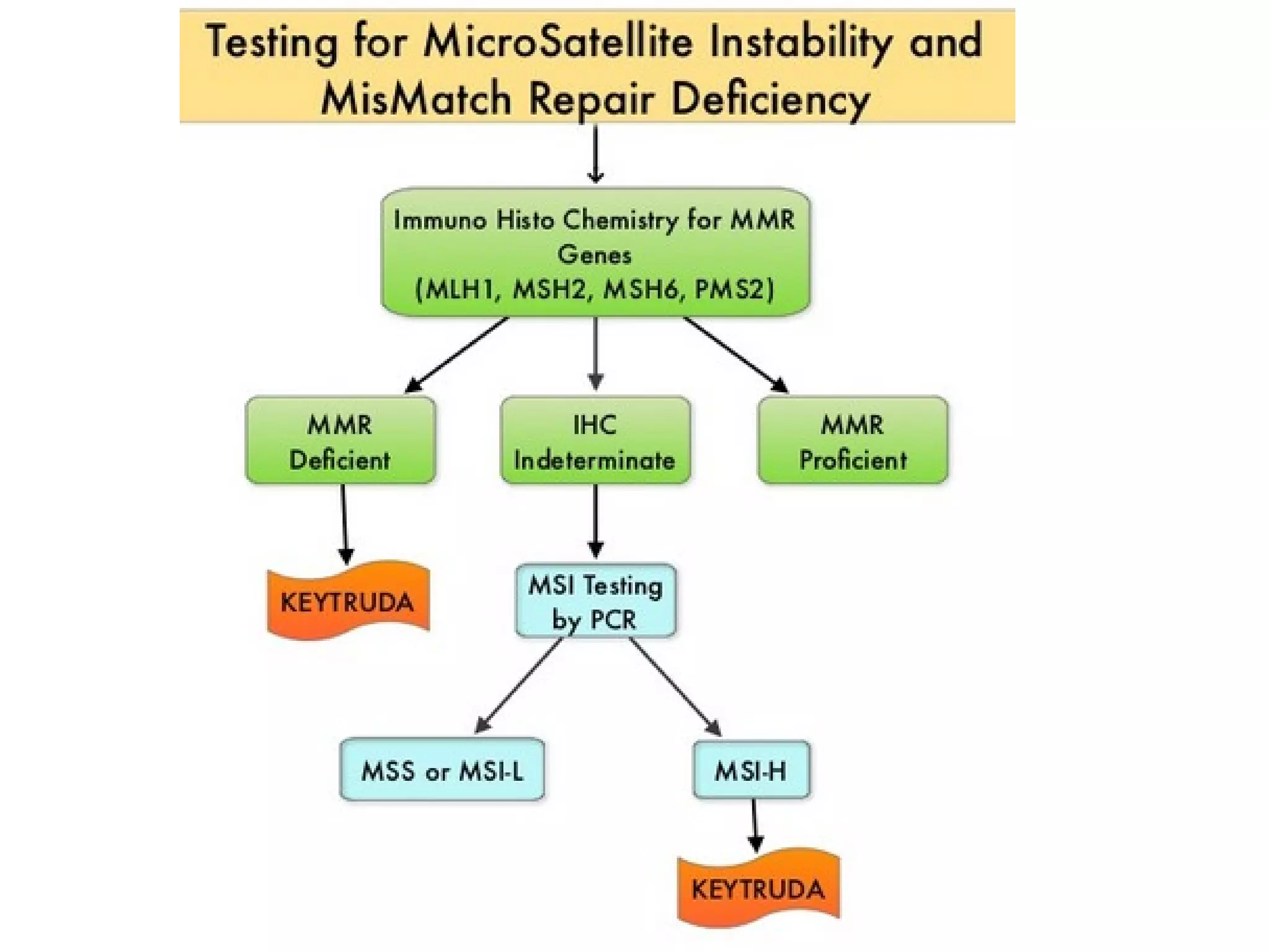
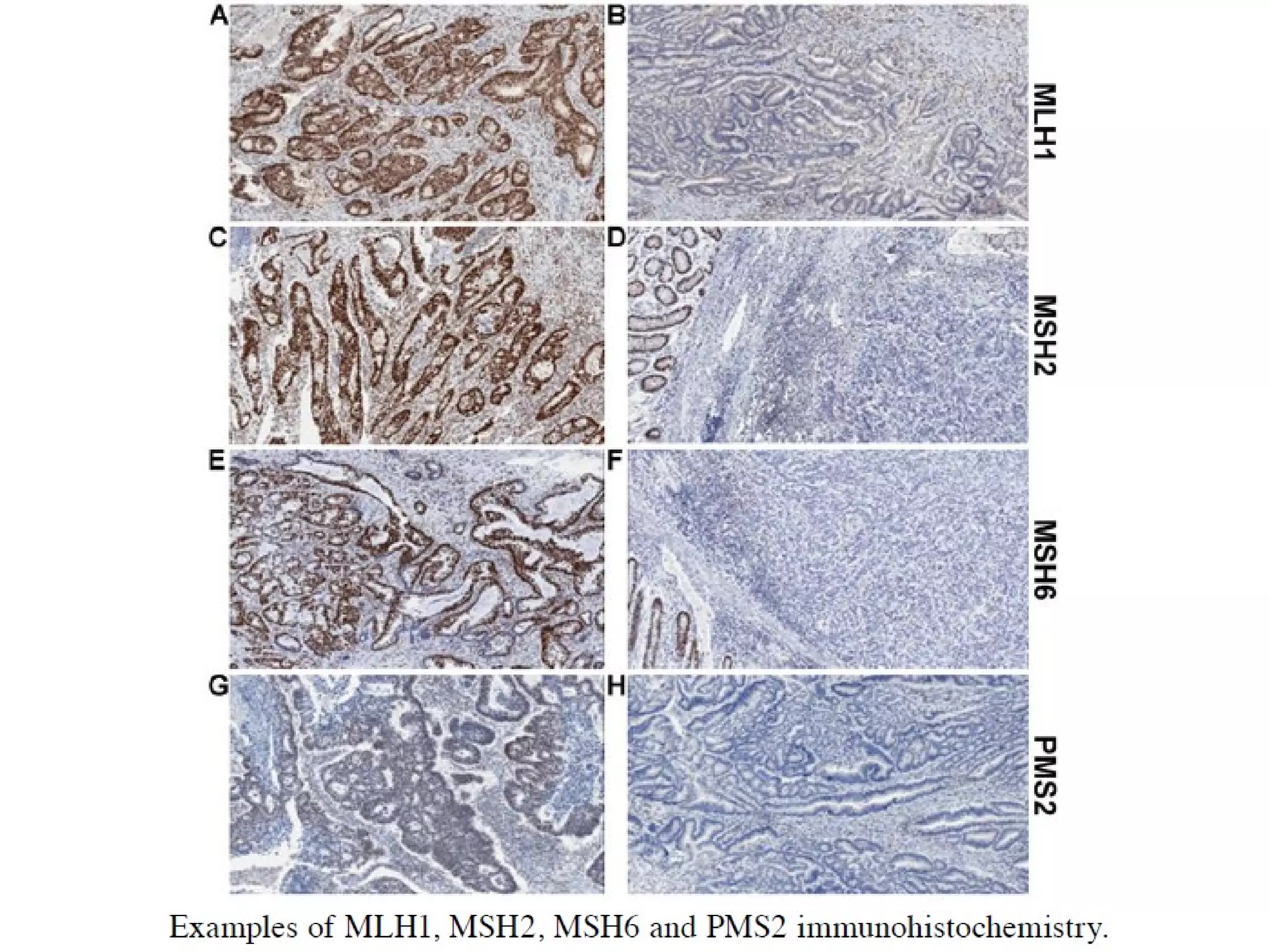
The FDA has granted accelerated approval to pembrolizumab (Keytruda) for treating adult and pediatric patients with unresectable or metastatic, microsatellite instability-high (MSI-H) or mismatch repair deficient (dMMR) solid tumors regardless of where in the body the cancer started. Defects in DNA mismatch repair proteins can result in microsatellite instability, which occurs in up to 15% of sporadic colorectal cancers. Researchers used immunohistochemistry testing on tumor tissue samples to determine dMMR status by looking for expression of the MLH1 and MSH2 mismatch repair proteins.






















