Module C5 Summary
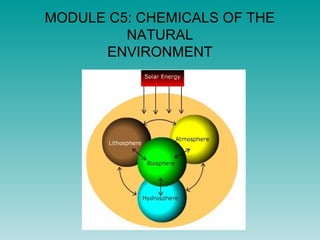
- 1. MODULE C5: CHEMICALS OF THE NATURAL ENVIRONMENT
- 2. Atmosphere • Molecular elements (e.g. oxygen, nitrogen, ozone) and compounds (e.g.carbon dioxide, water) • Monoatomic elements (e.g. argon) hydrosphere • Mainly water plus… • Solution of ionic compounds (e.g. sodium chloride) Q. Give the chemical symbols and state symbols for all the chemicals above.
- 3. Properties Molecular compounds Ionic compounds Made from Two or more non-metal atoms bonded Metal ions with non metal ions Electrical properties Insulators (no ions!) Conduct only when molten or dissolved in water Forces holding them together Strong covalent bonds within the molecule but weak forces between molecules Strong attractions between oppositely charged ions Melting and boiling points Low… found as gases at room temperature High… found as solids or in solution
- 4. Formulae of salts • Metal ions (+) combine with non-metal ions (-) to form neutral compounds. • The formula of magnesium oxide is MgO • The formula of sodium oxide is Na2O Mg 2+ Mg 2+ Mg 2+ Mg 2+ Mg 2+ Mg 2+ O 2- O 2- O 2- O 2- O 2- Mg 2+ O 2- Na + O 2- Na + O 2- O 2- O 2- O 2- O 2- O 2- O 2- O 2- O 2- O 2- O 2- O 2- O 2- O 2- O 2- O 2- O 2- O 2- O 2- O 2- O 2- O 2- O 2- O 2- O 2- O 2- O 2- O 2- O 2- O 2- O 2- O 2- O 2- O 2- Find the formula of… aluminium with fluorine, calcium with chlorine, sulphur with potassium The group number of the atom can tell us which ion it will form.. group ion 1 1+ 2 2+ 3 3+ 4 None 5 3- 6 2- 7 1- 0 None
- 5. Lithosphere • Made of the crust and upper mantle • Composed of minerals e.g. quartz containing silicon dioxide found in granite and sandstone • Rich in the atoms of silicon, oxygen and aluminium Treasures of the Earth: Gemstones for beauty, hardness, rarity
- 6. Chemical Brothers • Carbon dioxide and silicon dioxide are both covalently bonded compounds but with very different physical properties Carbon dioxide Silicon dioxide Molecular structure (limited covalent bonding) Giant structure (more extensive covalent bonding) Low melting point High melting point insulator Semi-conductor gas Hard solid Soluble in water Insoluble in water negatively charged electrons are attracted to the positively charged nuclei to form strong covalent bonds
- 7. Biosphere • the total sum of living organisms • Contains large molecules such as fats, carbohydrates, proteins, DNA • Mostly: carbon, hydrogen, oxygen, nitrogen • Minor contributions from phosphorus and sulphur What’s the formula of the glucose molecule above?
- 8. Cycling of nutrients • Many elements are cycled in nature including carbon, nitrogen and oxygen. • Identify in the cycle opposite where carbon can be found as… (i) simple molecular compounds, (ii) large molecules and (iii) as the element
- 9. Extracting metals • For zinc, iron and copper (fairly un-reactive metals) the ore (often an oxide) is mined, concentrated then the metal is extracted by heating with carbon Copper oxide + carbon copper + carbon dioxide lost oxygen gained oxygen When oxygen is lost: REDUCTION When oxygen is gained: OXIDATION Overall the process is REDOX Re-write the equation above using symbols (chemical and state symbols!) Write out the equivalent equation for iron (III) oxide (where iron has a 3+ charge)
- 10. Extracting reactive metals • The oxides of the more reactive metals are very stable so they require large amounts of energy supplied by electricity in order to extract the metal. The electricity decomposes the compound into simpler substances. Electrolyte: aluminium oxide (bauxite) with cryolite added
- 11. … in more detail • The cryolite makes the aluminium oxide (bauxite) easier to melt so the ions in it are free to move to each electrode to become neutral atoms Al 3+ Al 3+ Al 3+ O 2- O 2- O 2- cathode anode oxygen gas released Molten aluminium metal drops to the bottom Use an ionic equation to show what happens at each electrode
- 12. Metals • The typical properties of high melting/ boiling points, high strength, malleable and conduction of heat/ electricity can all be explained by their structure M + M + M + M +M + M + M + M + M + Giant structure held together by ionic bonding Metal ions in a lattice Sea of electrons (free to move)
- 13. Environmental issues • The (i) mining, (ii) extraction, (iii) transport, (iv) maintenance and (v) disposal of metals all have an impact on the environment. • For each stage above suggest how it may affect the surroundings
