lez12_01_23_468addbc67a5c01af41aa3545cebed20.pptx
•Download as PPTX, PDF•
0 likes•1 view
protein
Report
Share
Report
Share
Recommended
Recommended
More Related Content
Similar to lez12_01_23_468addbc67a5c01af41aa3545cebed20.pptx
Similar to lez12_01_23_468addbc67a5c01af41aa3545cebed20.pptx (20)
In biology, the roles of transition metal ions can be broadly groupe.pdf

In biology, the roles of transition metal ions can be broadly groupe.pdf
Heavy metals and its effects on plants and environment

Heavy metals and its effects on plants and environment
Charge-Transfer-Spectra. metal to metal, metal to ligand

Charge-Transfer-Spectra. metal to metal, metal to ligand
Structure and Catalytic Function of Superoxide Dismutase.pptx

Structure and Catalytic Function of Superoxide Dismutase.pptx
Effects of heavy metals and radiation pollution on biological system.pptx

Effects of heavy metals and radiation pollution on biological system.pptx
Recently uploaded
Recently uploaded (20)
Biogenic Sulfur Gases as Biosignatures on Temperate Sub-Neptune Waterworlds

Biogenic Sulfur Gases as Biosignatures on Temperate Sub-Neptune Waterworlds
Vip profile Call Girls In Lonavala 9748763073 For Genuine Sex Service At Just...

Vip profile Call Girls In Lonavala 9748763073 For Genuine Sex Service At Just...
TEST BANK For Radiologic Science for Technologists, 12th Edition by Stewart C...

TEST BANK For Radiologic Science for Technologists, 12th Edition by Stewart C...
Kochi ❤CALL GIRL 84099*07087 ❤CALL GIRLS IN Kochi ESCORT SERVICE❤CALL GIRL

Kochi ❤CALL GIRL 84099*07087 ❤CALL GIRLS IN Kochi ESCORT SERVICE❤CALL GIRL
Chemical Tests; flame test, positive and negative ions test Edexcel Internati...

Chemical Tests; flame test, positive and negative ions test Edexcel Internati...
Formation of low mass protostars and their circumstellar disks

Formation of low mass protostars and their circumstellar disks
Creating and Analyzing Definitive Screening Designs

Creating and Analyzing Definitive Screening Designs
Biopesticide (2).pptx .This slides helps to know the different types of biop...

Biopesticide (2).pptx .This slides helps to know the different types of biop...
Botany krishna series 2nd semester Only Mcq type questions

Botany krishna series 2nd semester Only Mcq type questions
All-domain Anomaly Resolution Office U.S. Department of Defense (U) Case: “Eg...

All-domain Anomaly Resolution Office U.S. Department of Defense (U) Case: “Eg...
VIRUSES structure and classification ppt by Dr.Prince C P

VIRUSES structure and classification ppt by Dr.Prince C P
SAMASTIPUR CALL GIRL 7857803690 LOW PRICE ESCORT SERVICE

SAMASTIPUR CALL GIRL 7857803690 LOW PRICE ESCORT SERVICE
Seismic Method Estimate velocity from seismic data.pptx

Seismic Method Estimate velocity from seismic data.pptx
Discovery of an Accretion Streamer and a Slow Wide-angle Outflow around FUOri...

Discovery of an Accretion Streamer and a Slow Wide-angle Outflow around FUOri...
lez12_01_23_468addbc67a5c01af41aa3545cebed20.pptx
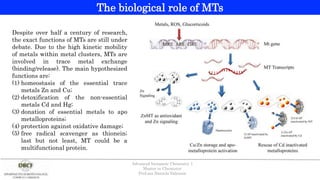
- 1. Despite over half a century of research, the exact functions of MTs are still under debate. Due to the high kinetic mobility of metals within metal clusters, MTs are involved in trace metal exchange (binding/release). The main hypothesized functions are: (1) homeostasis of the essential trace metals Zn and Cu; (2) detoxification of the non-essential metals Cd and Hg; (3) donation of essential metals to apo metalloproteins; (4) protection against oxidative damage; (5) free radical scavenger as thionein; last but not least, MT could be a multifunctional protein. The biological role of MTs Advanced Inorganic Chemistry 1 Master in Chemistry Prof.ssa Daniela Valensin
- 2. Their basic functions depend on the organism and the peptide variants: As metal storing proteins they participate in the homeostasis of metal ions first of all that of copper and zinc. As detoxification molecules they are active in the removal of detrimental soft metal ions (such as CdII, HgII, AgI and AuI). Their synthesis is induced by some essential, Zn and Cu, but some toxic metal ions, Cd, too. Inorganic-Hg does, but organic-Hg does not induce formation of metallothioneins. Biological role of MTs Advanced Inorganic Chemistry 1 Master in Chemistry Prof.ssa Daniela Valensin
- 3. A metallothioneins occur in human, in animals and plants (phytochelatins), they are low molecular mass (6-7 kDa) proteins, which bind soft metal ions (CuI, ZnII, CdII, Hg2 II, HgII, AgI and CoII) in cluster structure. Their sulphur and metal contents are very high, may reach 10%. Generally they consisit of two clusters, in which the metal ions coordinate through Cys-thiolates. The polypeptid part features repeated Cys-X-Cys sequences. In the middle of the Figure 12 terminal and 8 bridging CYS side chain bind all together 7 Cd2+-ions, in a chair conformation [3M-3S] cluster (Cd3S9) and an adamantane conformation [4M-5S] cluster (Cd4S11). Metal Detoxification Advanced Inorganic Chemistry 1 Master in Chemistry Prof.ssa Daniela Valensin
- 4. Metal Detoxification Advanced Inorganic Chemistry 1 Master in Chemistry AA 20/21 Prof.ssa Daniela Valensin
- 5. Metal Detoxification Advanced Inorganic Chemistry 1 Master in Chemistry Prof.ssa Daniela Valensin
- 6. (a) Entering of metal ions into a cell. (b) The ions interact with metal synthesis inhibitor (MTI). (c) Released metal transcription factor 1 (MTF-1) binds to metal responsive element (MRE). (d) Synthesis of mRNA to translate MT. (e) MT binds a heavy metal ion, the MT–heavy-metal-ion complex is transported to kidney or (f) to heavy metal dependent regulation proteins. MTF-1 Heavy metals (Zn, Cd, Pb, Cu ?) ? Phosphorylation? Nucleus Cytoplasm MRE mRNA MT MTF-1 MTI MTI MT excretion kidney (a) (b) (c) (d) (f) (e) MTF-1 Metal Detoxification Advanced Inorganic Chemistry 1 Master in Chemistry Prof.ssa Daniela Valensin
- 7. Metallothionein scavenging of reactive oxygen species. Presence of redox metals, such as Cu and Fe, in a cell can produce reactive oxygen species (ROS), leading to damaging of DNA and cell structures. The cell protects itself using various molecules as scavengers of the radicals. One of the most crucial cell pathways to scavenge the radicals is the glutathione redox complex. However, free –SH moieties of MT can be also involved in the scavenging of ROS in the MT redox cycle. Under physiologic conditions, zinc bound to MT is released through oxidation of the thiolate cluster when the environment becomes oxidized. Formation of MT-disulfide would be subjected to degradation; however, when the oxidized environment became reduced—through, for example, an increase in the glutathione (GSH)/glutathione disulfide (GSSG) ratio—MT disulfide is reduced to MT-thiol. This reduction process is greatly enhanced in the presence of selenium catalyst. In the presence of zinc, MT is quickly reconstituted. This process constitutes the MT redox cycle, which plays a crucial role in the biologic function of MT. Metal Detoxification Advanced Inorganic Chemistry 1 Master in Chemistry Prof.ssa Daniela Valensin
- 8. MRE MTF-1 (d) Nucleus Cytoplasm (a) (b) (c) MT S S H2O2 ●OH Reactive oxygen species (ROS) GSSG GSH DNA damage MT S S MT S S Cell damage MT S S MT S S Reactive oxygen species Heavy metals (Zn, Cd, Pb, Cu ? (a) Presence of heavy metals in a cell can produce reactive oxygen species (ROS). (b) To scavenge ROS glutathione and MT can be used. (c) Free heavy metal ions lead to activation of MTF-1 → to synthesis of MT. (d) MT can bind metal ions and scavenge ROS. Metal Detoxification Advanced Inorganic Chemistry 1 Master in Chemistry Prof.ssa Daniela Valensin
- 9. Neurodegenrative Disease Advanced Inorganic Chemistry 1 Master in Chemistry Prof.ssa Daniela Valensin
- 10. MTs and Alzheimer Disease Advanced Inorganic Chemistry 1 Master in Chemistry Prof.ssa Daniela Valensin
- 11. MTs and Alzheimer Disease Advanced Inorganic Chemistry 1 Master in Chemistry Prof.ssa Daniela Valensin
- 12. MTs and Alzheimer Disease Advanced Inorganic Chemistry 1 Master in Chemistry Prof.ssa Daniela Valensin
- 13. MTs and Alzheimer Disease Advanced Inorganic Chemistry 1 Master in Chemistry Prof.ssa Daniela Valensin
- 14. MTs and Alzheimer Disease Advanced Inorganic Chemistry 1 Master in Chemistry Prof.ssa Daniela Valensin
- 15. MTs and Alzheimer Disease Advanced Inorganic Chemistry 1 Master in Chemistry AA 20/21 Prof.ssa Daniela Valensin
- 16. MTs and Alzheimer Disease Advanced Inorganic Chemistry 1 Master in Chemistry Prof.ssa Daniela Valensin