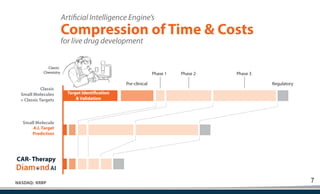
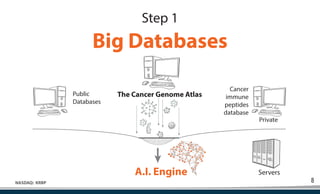
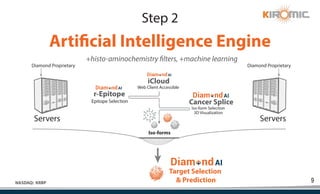
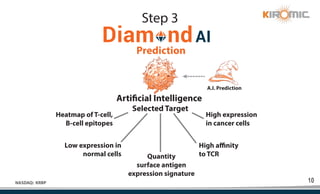
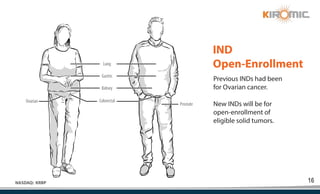
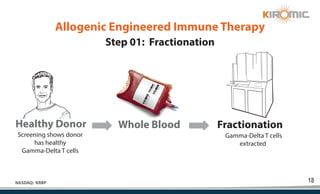
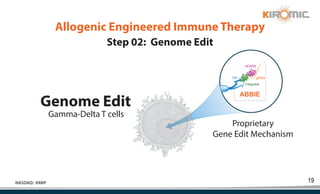
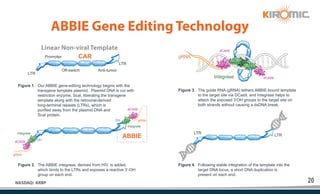
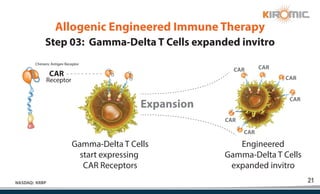
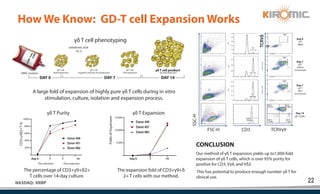
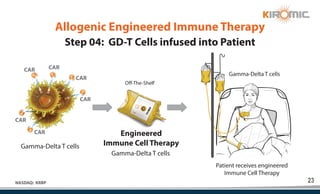
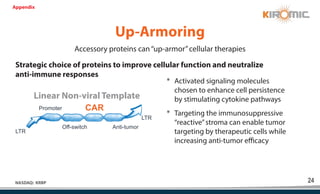
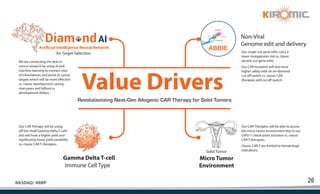
Kiromic Biopharma, Inc. is leveraging artificial intelligence and machine learning to enhance allogenic CAR therapy for solid tumors, aiming to identify effective cancer targets more efficiently and cost-effectively compared to traditional methods. Their platform, 'Diamond', is designed to discover novel cancer immunological targets and facilitate the development of therapies using off-the-shelf gamma-delta T cells. The company highlights innovations in genetic modifications and the potential to treat multiple solid tumors through advanced CAR-T technologies.