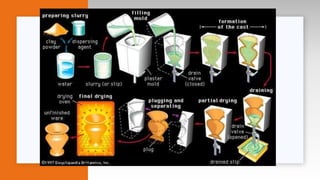
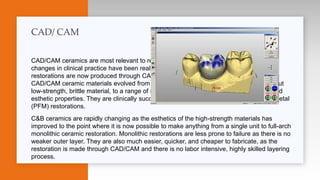
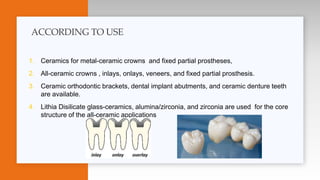
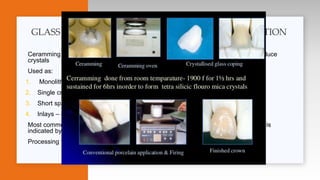
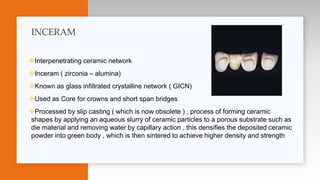
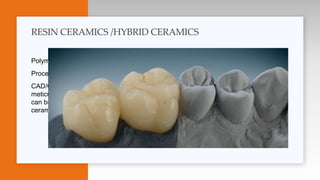
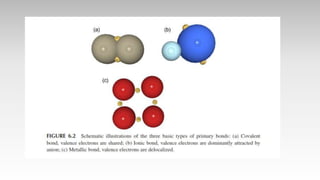
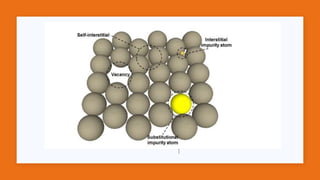
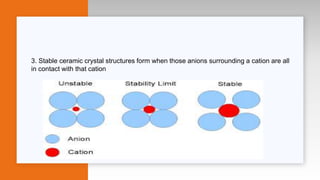
This document provides an overview of ceramics, including:
- Definitions of ceramics as inorganic, non-metallic materials formed from powders and strengthened through firing.
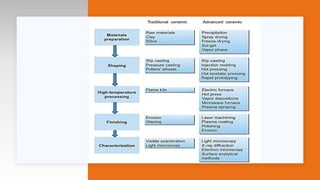
- Classifications based on fabrication method, crystalline phase, use, firing temperature, composition, microstructure, and properties.
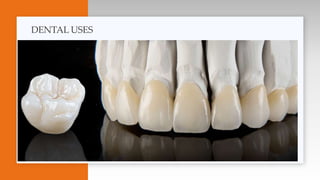
- A brief history of ceramics development and uses in dentistry and other applications.
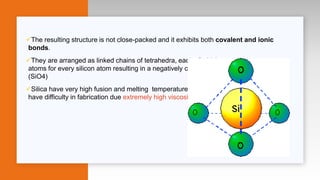
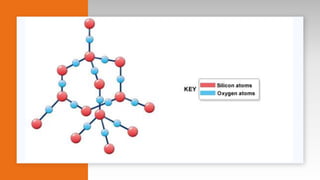
- Descriptions of microstructure, common crystalline phases, and how structure influences properties.