Inflammatory Bowel Disease Drug Development Solutions
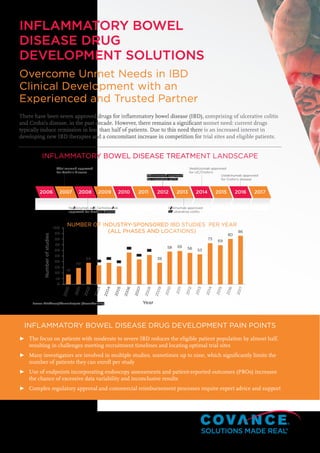
- 1. 2006 2007 2008 2009 2010 2011 2012 2013 2014 2015 2016 2017 Adalimumab approved for Crohn’s disease Adalimumab approved for ulcerative colitis Ustekinumab approved for Crohn’s disease Vedolizumab approved for UC/Crohn’s Natalizumab and Certolizumab approved for Crohn’s disease Golimumab approved for ulcerative colitis Year Numberofstudies 2000 2001 2002 2003 2004 2005 2006 2007 2008 2009 2010 2011 2012 2013 2014 2015 2016 2017 100 17 29 38 34 38 32 56 44 52 38 58 59 56 53 73 69 80 8690 80 70 60 50 40 30 20 10 0 ► The focus on patients with moderate to severe IBD reduces the eligible patient population by almost half, resulting in challenges meeting recruitment timelines and locating optimal trial sites ► Many investigators are involved in multiple studies, sometimes up to nine, which significantly limits the number of patients they can enroll per study ► Use of endpoints incorporating endoscopy assessments and patient-reported outcomes (PROs) increases the chance of excessive data variability and inconclusive results ► Complex regulatory approval and commercial reimbursement processes require expert advice and support INFLAMMATORY BOWEL DISEASE DRUG DEVELOPMENT PAIN POINTS Source: TrialTrove/PharmaProjects (November 2017) INFLAMMATORY BOWEL DISEASE DRUG DEVELOPMENT SOLUTIONS Overcome Unmet Needs in IBD Clinical Development with an Experienced and Trusted Partner There have been seven approved drugs for inflammatory bowel disease (IBD), comprising of ulcerative colitis and Crohn’s disease, in the past decade. However, there remains a significant unmet need: current drugs typically induce remission in less than half of patients. Due to this need there is an increased interest in developing new IBD therapies and a concomitant increase in competition for trial sites and eligible patients. INFLAMMATORY BOWEL DISEASE TREATMENT LANDSCAPE NUMBER OF INDUSTRY-SPONSORED IBD STUDIES PER YEAR (ALL PHASES AND LOCATIONS)
- 2. LOCATING THE OPTIMAL TRIAL SITES ► Xcellerate® Trial Design leverages data in the Covance clinical trial knowledgebase, which houses >40% global trial data at any one time – this real-world data can be used to match areas of high patient density with the location of high-performing investigators to find optimal trial sites, and includes: –– Global data on >2,000 experienced IBD sites –– LabCorp network of approximately 55,000 physicians with IBD patients REACHING RECRUITMENT TARGETS AND MAXIMIZING PATIENT RETENTION ► Covance leverages established relationships with IBD patient advocacy groups worldwide ► LabCorp database of approximately 237,000 IBD patients in the U.S., 1,380 of whom have opted to be contacted directly for clinical trial participation ► Use voice of patient insights to support participating patients and reduce study burden MAINTAINING HIGH-QUALITY DATA ► Centralized assessment of endoscopy data ensures consistency of interpretation across trial sites ► We provide PRO training to site staff and patients to ensure PROs are collected and interpreted correctly ► We implement measures to control placebo response, such as ensuring patients are assessed by the same independent assessor throughout the trial and proactively discussing the potential for placebo response with investigators ► Xcellerate Monitoring, part of our proprietary informatics suite, enables rapid identification of potential issues so that immediate preventative action can be taken NAVIGATING REGULATORY PATHWAYS ► Dedicated regulatory team have nearly 30 years’ experience each with local and global regulatory bodies ► Generate real-world evidence to communicate product value via Covance Market Access Learn more about our drug development solutions at www.covance.com Covance Inc., headquartered in Princeton, NJ, USA is the drug development business of Laboratory Corporation of America Holdings (LabCorp). COVANCE is registered trademark and themarketing name for Covance Inc. and its subsidiaries around the world. The Americas +1.888.COVANCE (+1.888.268.2623) +1.609.452.4440 Europe / Africa +00.800.2682.2682 +44.1423.500888 Asia Pacific +800.6568.3000 +65.6.5686588 © Copyright 2018 Covance Inc. INFCDS012-0118 INFLAMMATORY BOWEL DISEASE DRUG DEVELOPMENT SOLUTIONS Avoid Common Clinical Development Challenges with Covance IBD Solutions
