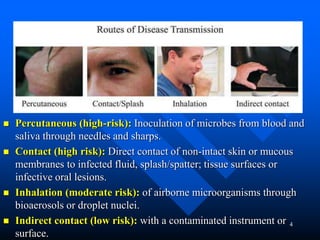
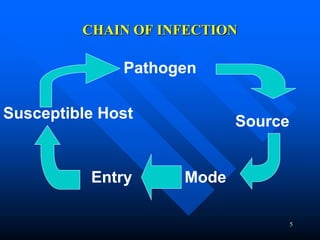
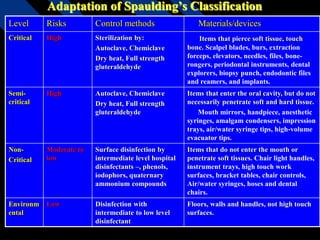
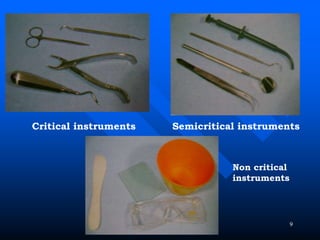
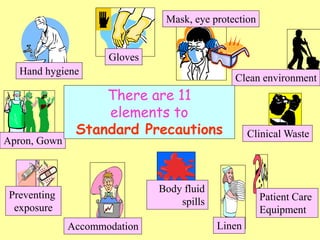
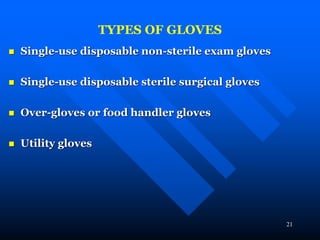
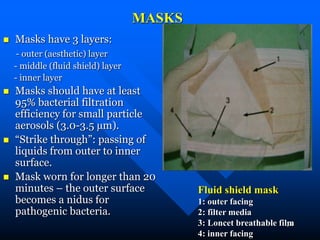
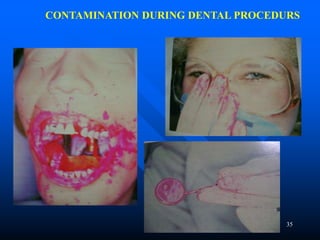
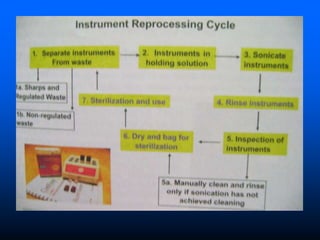
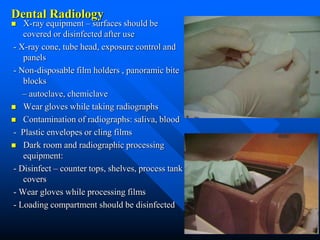
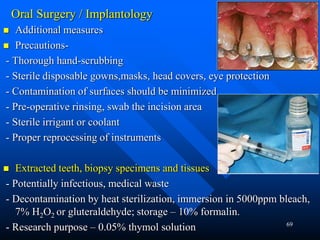
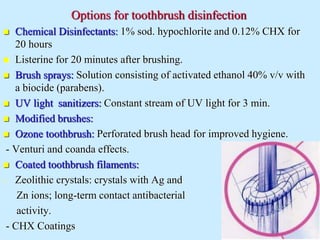
Infection control in dentistry is crucial to prevent the transmission of pathogens between dental healthcare providers and patients, as various routes of disease transmission exist in the dental environment. Proper decontamination procedures, including sterilization and the use of personal protective equipment, help mitigate risks associated with potential infections. Regulatory guidelines emphasize standard precautions for all patient interactions to enhance safety and minimize infection risks.







































































![84
6. World Health Organization. SEARO Regional Health Papers no. 18. A Manual on
Infection Control in Health Facilities. World Health Organization. Regional Office
for South-East Asia. New Delhi, 1988.
7. Kohn WJ et al. (2003). Guidelines for Infection Control in Dental Health-Care
Settings – 2003. [Online] MMWR Recommendations and Reports 2003 Dec 19; 52
(No. RR—17). Available: http://www.cdc.gov/mmwR/PDF/rr/rr5217.pdf [20th May
2008]
8. Sterigenics. Sterilization Alternatives. Electron Beam Radiation. [Online]. Available:
http://www.sterigenics.com/services/medical_sterilization/contract_sterilization/elec
tron_beam_radiation/sterilization_alternatives__electron_beam_radiation.pdf [29th
May 2008]
9. Allen D (1998). The Changing Face of Sterilization. [Online] Pharmaceutical and
Medical News Packaging Magazine 1998 Nov; 35-46. Available:
http://www.devicelink.com/pmpn/archive/98/11/004.html [29th May 2008]
10. Molinari JA. Infection control: Its evolution to the current standard precautions. J
Am Dent Assoc 2003; 134: 569-574.](https://image.slidesharecdn.com/infectioncontrolindentistry-231025042648-309bfd04/85/INFECTION-CONTROL-IN-DENTISTRY-ppt-84-320.jpg)
![85
11. Park K. Park’s Textbook of Preventive and Social Medicine. 19th ed. Bhanot
Publishers, Jabalpur.
12. Thraenhart O, Jursch C. Measures for Disinfection and Control of Viral Hepatitis.
In: Block SS. Disinfection, Sterilization and Preservation. 5th Ed. Lippincott
Williams & Wilkins, 2001.
13. Curran E 2003. A Self-directed Learning Unit ON Standard Precautions. [Online]
developed from NHS Greater Glasgow Control of Infection Policy 2003. Available:
shscintranet.gcal.ac.uk/student/Documents/PodDocs/sdlu_sp1.ppt [29th December
2008]](https://image.slidesharecdn.com/infectioncontrolindentistry-231025042648-309bfd04/85/INFECTION-CONTROL-IN-DENTISTRY-ppt-85-320.jpg)
