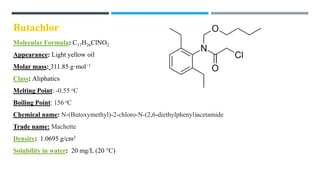
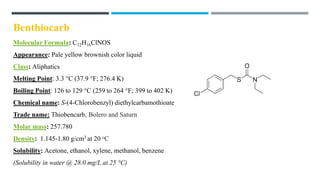
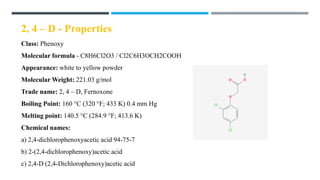
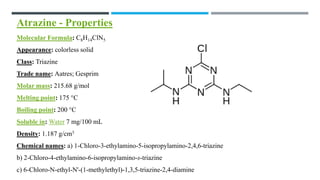
The document discusses various herbicides, including 2,4-D, atrazine, glyphosate, butachlor, and benthiocarb, detailing their chemical properties, uses, modes of action, and safety evaluations. 2,4-D is notable for its historical use and recent regulatory reviews, while atrazine is restricted due to its environmental impacts. Glyphosate is widely used in agriculture and has significant herbicidal activity, and other herbicides like butachlor and benthiocarb are highlighted for their specific applications in rice cultivation.








![Molecular Formula: C3H8NO5P
Appearance: white crystalline powder
Molar mass: 169.073 g·mol−1
Class: Aliphatics
Melting Point: 184.5 °C (364.1 °F; 457.6 K)
Boiling Point: 187 °C (369 °F; 460 K) decomposes
Chemical name: [(phosphonomethyl)amino]acetic acid
Trade name: Round up, Glycel
Molar mass: 169.07 g/mol
Density: 1.7 g/cm³
Solubility in water: 1.01 g/100 mL (20 °C)](https://image.slidesharecdn.com/herbicideschemicalpropertiesanduses-200924151246/85/Herbicides-chemical-properties-and-uses-11-320.jpg)