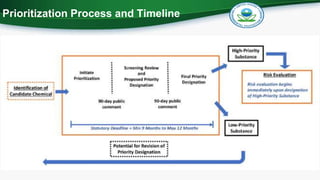
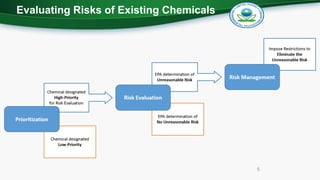
The document outlines the statutory requirements for the U.S. Environmental Protection Agency's risk-based screening process under the Toxic Substances Control Act, emphasizing the criteria for designating chemicals as high-priority or low-priority substances. By December 2019, the EPA aims to designate 20 high-priority chemicals for risk evaluations and 20 low-priority chemicals, focusing on factors like hazard, exposure, persistence, and potential risks to susceptible populations. Additionally, the prioritization process will prioritize chemicals from the 2014 TSCA work plan that have high toxicity and bioaccumulation scores.