Handout-Structure-of-med-res-paper-RG-checklists_FINAL.pdf
•
0 likes•10 views
This document outlines the key sections and content of a medical research paper, including reporting guidelines. It discusses sections like the title, abstract, introduction, methods, results, discussion, and conclusions. The methods section should describe the study design, participants, interventions, outcomes, and statistical analysis. The results section should report answers to the study questions using text, tables, and figures. The discussion should interpret the findings in the context of other research and discuss implications and recommendations. Reporting guidelines provide standards for transparently reporting different types of studies and their methodology.
Report
Share
Report
Share
Download to read offline
Recommended
An Introduction to writing an Systematic review of literature - Scientific re...

A systematic review is a type of literature review that uses a systematic method to collect Secondary data in which there is a comprehensive search for relevant studies on a particular topic, and those identified are then evaluated and synthesized according to a predetermined and explicit method.
This framework especially is essential for early career researchers and medical students to enhance their writing knowledge on the systematic review of the literature.
Why Pubrica:
When you order our services, we promise you the following – Plagiarism free, always on Time, outstanding customer support, written to Standard, Unlimited Revisions support and High-quality Subject Matter Experts.
Learn More: https://bit.ly/38jAbCT
Contact us:
Web: https://pubrica.com/
Blog: https://pubrica.com/academy/
Email: sales@pubrica.com
WhatsApp : +91 9884350006
United Kingdom : +44-1143520021
A research study Writing a Systematic Review in Clinical Research – Pubrica

A systematic review summarises the findings of precisely organized healthcare research (controlled trials) and gives a high degree of evidence on the efficacy of healthcare interventions. The evidence may be used to make decisions and guide healthcare recommendations.
Reference: https://bit.ly/3morikF
For our services: https://pubrica.com/services/research-services/systematic-review/
Why Pubrica:
When you order our services, We promise you the following – Plagiarism free | always on Time | 24*7 customer support | Written to international Standard | Unlimited Revisions support | Medical writing Expert | Publication Support | Biostatistical experts | High-quality Subject Matter Experts.
Contact us:
Web: https://pubrica.com/
Blog: https://pubrica.com/academy/
Email: sales@pubrica.com
WhatsApp : +91 9884350006
United Kingdom: +44-1618186353
A research study writing a systematic review in clinical research – pubrica

A systematic review summarises the findings of precisely organized healthcare research (controlled trials) and gives a high degree of evidence on the efficacy of healthcare interventions. The evidence may be used to make decisions and guide healthcare recommendations.
Reference: https://bit.ly/3morikF
For our services: https://pubrica.com/services/research-services/systematic-review/
Why Pubrica:
When you order our services, We promise you the following – Plagiarism free | always on Time | 24*7 customer support | Written to international Standard | Unlimited Revisions support | Medical writing Expert | Publication Support | Biostatistical experts | High-quality Subject Matter Experts.
Contact us:
Web: https://pubrica.com/
Blog: https://pubrica.com/academy/
Email: sales@pubrica.com
WhatsApp : +91 9884350006
United Kingdom: +44-1618186353
Recommended
An Introduction to writing an Systematic review of literature - Scientific re...

A systematic review is a type of literature review that uses a systematic method to collect Secondary data in which there is a comprehensive search for relevant studies on a particular topic, and those identified are then evaluated and synthesized according to a predetermined and explicit method.
This framework especially is essential for early career researchers and medical students to enhance their writing knowledge on the systematic review of the literature.
Why Pubrica:
When you order our services, we promise you the following – Plagiarism free, always on Time, outstanding customer support, written to Standard, Unlimited Revisions support and High-quality Subject Matter Experts.
Learn More: https://bit.ly/38jAbCT
Contact us:
Web: https://pubrica.com/
Blog: https://pubrica.com/academy/
Email: sales@pubrica.com
WhatsApp : +91 9884350006
United Kingdom : +44-1143520021
A research study Writing a Systematic Review in Clinical Research – Pubrica

A systematic review summarises the findings of precisely organized healthcare research (controlled trials) and gives a high degree of evidence on the efficacy of healthcare interventions. The evidence may be used to make decisions and guide healthcare recommendations.
Reference: https://bit.ly/3morikF
For our services: https://pubrica.com/services/research-services/systematic-review/
Why Pubrica:
When you order our services, We promise you the following – Plagiarism free | always on Time | 24*7 customer support | Written to international Standard | Unlimited Revisions support | Medical writing Expert | Publication Support | Biostatistical experts | High-quality Subject Matter Experts.
Contact us:
Web: https://pubrica.com/
Blog: https://pubrica.com/academy/
Email: sales@pubrica.com
WhatsApp : +91 9884350006
United Kingdom: +44-1618186353
A research study writing a systematic review in clinical research – pubrica

A systematic review summarises the findings of precisely organized healthcare research (controlled trials) and gives a high degree of evidence on the efficacy of healthcare interventions. The evidence may be used to make decisions and guide healthcare recommendations.
Reference: https://bit.ly/3morikF
For our services: https://pubrica.com/services/research-services/systematic-review/
Why Pubrica:
When you order our services, We promise you the following – Plagiarism free | always on Time | 24*7 customer support | Written to international Standard | Unlimited Revisions support | Medical writing Expert | Publication Support | Biostatistical experts | High-quality Subject Matter Experts.
Contact us:
Web: https://pubrica.com/
Blog: https://pubrica.com/academy/
Email: sales@pubrica.com
WhatsApp : +91 9884350006
United Kingdom: +44-1618186353
Developing a Systematic Review Protocol

A systematic review (SR) is a rigorous and organized method to synthesize
the evidence from multiple studies on a particular research question or topic.
The purpose of a systematic review is to identify, appraise, and summarize all
available evidence relevant to a specific research question in a transparent
and replicable manner.
It aims to provide a comprehensive overview of academic literature
concerning a particular research question of topic.
This presentation explores the steps nee
DMID Interventional Protocol TemplateVersion 2.028 April 2005

DMID Interventional Protocol Template Version 2.0
28 April 2005
Protocol Title Version number and date
GENERAL INSTRUCTIONS – delete this box from the submitted Protocol
This template is for students in the Virginia University of Lynchburg Doctor of Healthcare Administration Research Practicum course who are preparing a detailed protocol for a study involving human subjects. Depending on the nature of what you are doing, some sections may not be applicable to your research. If a section is not applicable, delete. You may delete subsections that are not applicable. The full research protocol must be uploaded to Moodle to be considered complete. This includes the IRB Application with research protocol, Informed Consent Document (s), Recruitment Collateral, and any other supporting documentation. Applications with ANY missing elements will be considered incomplete and will be graded accordingly.
Use this template to create a study protocol as follows:
· Red text represents instructions to you – to be deleted from the final version
· Blue text represents guidance on suggested content – to be edited and changed to black or replaced with black in the final version.
· Black text represents text that should ordinarily be incorporated as-is, if applicable
Note that the table of contents is automatically included, so do not change the content or formatting of the headings. Be sure to right click on the table of contents and select “Update field” before saving the protocol and uploading it to Moodle. As always, make sure to proofread the document before submission.
Please make sure to complete the header on this page with the protocol title and version number and date.
The submitted protocol should have no red or blue text (including the header and instruction boxes like this one). The submitted protocol should have no spelling or grammar errors. All references MUST be in APA 7 format. PROTOCOL TITLEProtocol Version Number: CompleteProtocol Version Date: day, month, year [Include if there is an external funder; otherwise, delete heading] Funding Mechanism: organization and grant or contract #[Include if there is industry support; otherwise, delete heading] Industry Support provided by: name of industryPrincipal Investigator: name Phone: Complete E-mail: Complete[Include if the study has a medical monitor; otherwise, delete heading] Medical Monitor: name
Table of Contents
1List of Abbreviations4
2Protocol Summary4
3Background/Rationale & Purpose5
3.1Background Information5
3.2Rationale and Purpose5
4Objectives5
4.1Study Objectives5
4.2Study Outcome Measures6
4.2.1Primary Outcome Measures6
4.2.2Secondary Outcome Measures6
5Study Design6
6Potential Risks and Benefits7
6.1Risks7
6.2Potential Benefits8
6.3Analysis of Risks in Relation to Benefits8
7Study Subject Selection8
7.1Subject Inclusion Criteria8
7.2Subject Exclusion Criteria8
7.3Recruitment Methods9
7.4Compensation for Participation in Research Activities9
7.5Withdrawal of Pa ...
Systematic review.pptx

Pubrica's team of researchers and authors develop Scientific and medical research papers that can act as an indispensable tool to the practitioner/authors. Here is how we help.
Scientific Report Writing.pdf

Basic Understanding about Scientific Report Writings
Email at
razzaque.sarker@gmail.com
How to write a research paper before you start by Dr. R. Narayanasamy, Retd. ...

This ppt is uploaded on behalf of Dr.R.Narayanasamy, Retd Prof, Production Engg, NIT Trichy
protocol writing in clinical research 

protocol designing in clinical research - introduction, objectives, contents .....
Critique Template for a Qualitative StudyCritiquing Qualit.docx

Critique Template for a Qualitative Study
Critiquing Qualitative
What is a critique? Simply stated, a critique is a critical analysis undertaken for some purpose. Nurses critique research for three main reasons: to improve their practice, to broaden their understanding, and to provide a base for the conduct of a study.
When the purpose is to improve practice, nurses must give special consideration to questions such as these:
Are the research findings appropriate to my practice setting and situation?
What further research or pilot studies need to be done, if any, before incorporating findings into practice to assure both safety and effectiveness?
How might a proposed change in practice trigger changes in other aspects of practice?
To help you synthesize your learning throughout this course and prepare you to utilize research in your practice, you will be critiquing a qualitative, quantitative, or mixed-methods research study of your choice.
If the article is unavailable in a full-text version through the Walden University Library, you must
e-mail the article as a PDF or Word attachment to your Instructor.
QUALITATIVE RESEARCH CRITIQUE
Research Issue and Purpose
What is the research question or issue of the referenced study? What is its purpose? (Sometimes ONLY the purpose is stated clearly and the question must be inferred from the introductory discussion of the purpose.)
Researcher Pre-understandings
Does the article include a discussion of the researcher’s pre-understandings? What does the article disclose about the researcher’s professional and personal perspectives on the research problem?
Literature Review
What is the quality of the literature review? Is the literature review current, relevant? Is there evidence that the author critiqued the literature or merely reported it without critique? Is there an integrated summary of the current knowledge base regarding the research problem, or does the literature review contain opinion or anecdotal articles without any synthesis or summary of the whole? (Sometimes the literature review is incorporated into the introductory section without being explicitly identified.)
Theoretical or Conceptual Framework
Is a theoretical or conceptual framework identified? If so, what is it? Is it a nursing framework or one drawn from another discipline? (Sometimes there is no explicitly identified theoretical or conceptual framework; in addition, many “nursing” research studies draw on a “borrowed” framework, e.g., stress, medical pathology, etc.)
Participants
Who were the participants? Is the setting or study group adequately described? Is the setting appropriate for the research question? What type of sampling strategy was used? Was it appropriate? Was the sample size adequate? Did the researcher stipulate that information redundancy was achieved?
Protection of Human Research Participants
What steps were taken to protect human research subjects?
.
Research ProposalThe written research proposal will be created b.docx

Research Proposal
The written research proposal will be created based on a narrowly defined aspect of the topic selected in Week One. A research proposal is a detailed plan for a specific study to be conducted at a future time. It is the document which potential researchers typically submit to an institutional review board (IRB) for ethical review and approval, and/or to funding agencies to secure financial support for a research effort. Because it is a plan for research which has not yet been conducted, the Methods section should be written in the future tense and should not contain any hypothetical results. The paper must address all of the components required in the Methods section of a research proposal. The following actions must be completed.
State the research question and/or hypothesis.
Briefly compare the characteristics of the major research paradigms used in previous studies on the chosen topic. Introduce the specific approach (qualitative, quantitative, or mixed methods), research design, sampling strategy, data collection procedures, and data analysis techniques to be used in this study. Provide a short explanation as to why the selected procedures are more feasible than other alternatives for the research topic.
Describe any relevant variables, measures, and statistical tests.
Apply ethical principles and professional standards to the proposed psychological research. Provide an analysis of any ethical issues that may arise and explain how these issues will be resolved.
The following headings for the required sections and subsections must appear in the paper. In accordance with APA style, all references listed must be cited in the text of the paper.
Introduction
Introduce the research topic, explain why it is important, and present an appropriately and narrowly defined research question and/or hypothesis.
Literature Review
Evaluate the published research on the chosen topic including a minimum of three peer-reviewed articles. Summarize the current state of knowledge on the topic, making reference to the findings of previous research studies. Briefly mention the research methods that have previously been used to study the topic. State whether the proposed study is a replication of a previous study or a new approach employing methods that have not been used before. Be sure to properly cite all sources in APA style.
Methods
Design –
Create a feasible research design that incorporates appropriate methods to address the topic. Indicate whether the approach of the proposed study is qualitative, quantitative, or mixed methods. Identify the specific research design, and indicate whether it is experimental or non-experimental. Evaluate the chosen design and explain why this design is feasible and appropriate for the topic and how it will provide the information needed to answer the research question. Cite sources on research methodology to support these choices. Include a minimum of two peer-reviewed sources.
Participants –
Identify and.
More Related Content
Similar to Handout-Structure-of-med-res-paper-RG-checklists_FINAL.pdf
Developing a Systematic Review Protocol

A systematic review (SR) is a rigorous and organized method to synthesize
the evidence from multiple studies on a particular research question or topic.
The purpose of a systematic review is to identify, appraise, and summarize all
available evidence relevant to a specific research question in a transparent
and replicable manner.
It aims to provide a comprehensive overview of academic literature
concerning a particular research question of topic.
This presentation explores the steps nee
DMID Interventional Protocol TemplateVersion 2.028 April 2005

DMID Interventional Protocol Template Version 2.0
28 April 2005
Protocol Title Version number and date
GENERAL INSTRUCTIONS – delete this box from the submitted Protocol
This template is for students in the Virginia University of Lynchburg Doctor of Healthcare Administration Research Practicum course who are preparing a detailed protocol for a study involving human subjects. Depending on the nature of what you are doing, some sections may not be applicable to your research. If a section is not applicable, delete. You may delete subsections that are not applicable. The full research protocol must be uploaded to Moodle to be considered complete. This includes the IRB Application with research protocol, Informed Consent Document (s), Recruitment Collateral, and any other supporting documentation. Applications with ANY missing elements will be considered incomplete and will be graded accordingly.
Use this template to create a study protocol as follows:
· Red text represents instructions to you – to be deleted from the final version
· Blue text represents guidance on suggested content – to be edited and changed to black or replaced with black in the final version.
· Black text represents text that should ordinarily be incorporated as-is, if applicable
Note that the table of contents is automatically included, so do not change the content or formatting of the headings. Be sure to right click on the table of contents and select “Update field” before saving the protocol and uploading it to Moodle. As always, make sure to proofread the document before submission.
Please make sure to complete the header on this page with the protocol title and version number and date.
The submitted protocol should have no red or blue text (including the header and instruction boxes like this one). The submitted protocol should have no spelling or grammar errors. All references MUST be in APA 7 format. PROTOCOL TITLEProtocol Version Number: CompleteProtocol Version Date: day, month, year [Include if there is an external funder; otherwise, delete heading] Funding Mechanism: organization and grant or contract #[Include if there is industry support; otherwise, delete heading] Industry Support provided by: name of industryPrincipal Investigator: name Phone: Complete E-mail: Complete[Include if the study has a medical monitor; otherwise, delete heading] Medical Monitor: name
Table of Contents
1List of Abbreviations4
2Protocol Summary4
3Background/Rationale & Purpose5
3.1Background Information5
3.2Rationale and Purpose5
4Objectives5
4.1Study Objectives5
4.2Study Outcome Measures6
4.2.1Primary Outcome Measures6
4.2.2Secondary Outcome Measures6
5Study Design6
6Potential Risks and Benefits7
6.1Risks7
6.2Potential Benefits8
6.3Analysis of Risks in Relation to Benefits8
7Study Subject Selection8
7.1Subject Inclusion Criteria8
7.2Subject Exclusion Criteria8
7.3Recruitment Methods9
7.4Compensation for Participation in Research Activities9
7.5Withdrawal of Pa ...
Systematic review.pptx

Pubrica's team of researchers and authors develop Scientific and medical research papers that can act as an indispensable tool to the practitioner/authors. Here is how we help.
Scientific Report Writing.pdf

Basic Understanding about Scientific Report Writings
Email at
razzaque.sarker@gmail.com
How to write a research paper before you start by Dr. R. Narayanasamy, Retd. ...

This ppt is uploaded on behalf of Dr.R.Narayanasamy, Retd Prof, Production Engg, NIT Trichy
protocol writing in clinical research 

protocol designing in clinical research - introduction, objectives, contents .....
Critique Template for a Qualitative StudyCritiquing Qualit.docx

Critique Template for a Qualitative Study
Critiquing Qualitative
What is a critique? Simply stated, a critique is a critical analysis undertaken for some purpose. Nurses critique research for three main reasons: to improve their practice, to broaden their understanding, and to provide a base for the conduct of a study.
When the purpose is to improve practice, nurses must give special consideration to questions such as these:
Are the research findings appropriate to my practice setting and situation?
What further research or pilot studies need to be done, if any, before incorporating findings into practice to assure both safety and effectiveness?
How might a proposed change in practice trigger changes in other aspects of practice?
To help you synthesize your learning throughout this course and prepare you to utilize research in your practice, you will be critiquing a qualitative, quantitative, or mixed-methods research study of your choice.
If the article is unavailable in a full-text version through the Walden University Library, you must
e-mail the article as a PDF or Word attachment to your Instructor.
QUALITATIVE RESEARCH CRITIQUE
Research Issue and Purpose
What is the research question or issue of the referenced study? What is its purpose? (Sometimes ONLY the purpose is stated clearly and the question must be inferred from the introductory discussion of the purpose.)
Researcher Pre-understandings
Does the article include a discussion of the researcher’s pre-understandings? What does the article disclose about the researcher’s professional and personal perspectives on the research problem?
Literature Review
What is the quality of the literature review? Is the literature review current, relevant? Is there evidence that the author critiqued the literature or merely reported it without critique? Is there an integrated summary of the current knowledge base regarding the research problem, or does the literature review contain opinion or anecdotal articles without any synthesis or summary of the whole? (Sometimes the literature review is incorporated into the introductory section without being explicitly identified.)
Theoretical or Conceptual Framework
Is a theoretical or conceptual framework identified? If so, what is it? Is it a nursing framework or one drawn from another discipline? (Sometimes there is no explicitly identified theoretical or conceptual framework; in addition, many “nursing” research studies draw on a “borrowed” framework, e.g., stress, medical pathology, etc.)
Participants
Who were the participants? Is the setting or study group adequately described? Is the setting appropriate for the research question? What type of sampling strategy was used? Was it appropriate? Was the sample size adequate? Did the researcher stipulate that information redundancy was achieved?
Protection of Human Research Participants
What steps were taken to protect human research subjects?
.
Research ProposalThe written research proposal will be created b.docx

Research Proposal
The written research proposal will be created based on a narrowly defined aspect of the topic selected in Week One. A research proposal is a detailed plan for a specific study to be conducted at a future time. It is the document which potential researchers typically submit to an institutional review board (IRB) for ethical review and approval, and/or to funding agencies to secure financial support for a research effort. Because it is a plan for research which has not yet been conducted, the Methods section should be written in the future tense and should not contain any hypothetical results. The paper must address all of the components required in the Methods section of a research proposal. The following actions must be completed.
State the research question and/or hypothesis.
Briefly compare the characteristics of the major research paradigms used in previous studies on the chosen topic. Introduce the specific approach (qualitative, quantitative, or mixed methods), research design, sampling strategy, data collection procedures, and data analysis techniques to be used in this study. Provide a short explanation as to why the selected procedures are more feasible than other alternatives for the research topic.
Describe any relevant variables, measures, and statistical tests.
Apply ethical principles and professional standards to the proposed psychological research. Provide an analysis of any ethical issues that may arise and explain how these issues will be resolved.
The following headings for the required sections and subsections must appear in the paper. In accordance with APA style, all references listed must be cited in the text of the paper.
Introduction
Introduce the research topic, explain why it is important, and present an appropriately and narrowly defined research question and/or hypothesis.
Literature Review
Evaluate the published research on the chosen topic including a minimum of three peer-reviewed articles. Summarize the current state of knowledge on the topic, making reference to the findings of previous research studies. Briefly mention the research methods that have previously been used to study the topic. State whether the proposed study is a replication of a previous study or a new approach employing methods that have not been used before. Be sure to properly cite all sources in APA style.
Methods
Design –
Create a feasible research design that incorporates appropriate methods to address the topic. Indicate whether the approach of the proposed study is qualitative, quantitative, or mixed methods. Identify the specific research design, and indicate whether it is experimental or non-experimental. Evaluate the chosen design and explain why this design is feasible and appropriate for the topic and how it will provide the information needed to answer the research question. Cite sources on research methodology to support these choices. Include a minimum of two peer-reviewed sources.
Participants –
Identify and.
Similar to Handout-Structure-of-med-res-paper-RG-checklists_FINAL.pdf (20)
DMID Interventional Protocol TemplateVersion 2.028 April 2005

DMID Interventional Protocol TemplateVersion 2.028 April 2005
How to write a research paper before you start by Dr. R. Narayanasamy, Retd. ...

How to write a research paper before you start by Dr. R. Narayanasamy, Retd. ...
Critique Template for a Qualitative StudyCritiquing Qualit.docx

Critique Template for a Qualitative StudyCritiquing Qualit.docx
Research ProposalThe written research proposal will be created b.docx

Research ProposalThe written research proposal will be created b.docx
More from MOHAMMED YASER HUSSAIN
More from MOHAMMED YASER HUSSAIN (20)
2022-04-novartis-new-organizational-model-presentation.pdf

2022-04-novartis-new-organizational-model-presentation.pdf
omega-healthcare-new-investor-presentation-4q21-april-2022.pdf

omega-healthcare-new-investor-presentation-4q21-april-2022.pdf
HealthCare-Global-HCG-Q2FY18-Investor-Presentation.pdf

HealthCare-Global-HCG-Q2FY18-Investor-Presentation.pdf
gehc_2022_morgan_stanley_healthcare_conference_slides.pdf

gehc_2022_morgan_stanley_healthcare_conference_slides.pdf
Recently uploaded
Contact ME {89011**83002} Haridwar ℂall Girls By Full Service Call Girl In Ha...

Contact ME {89011**83002} Haridwar ℂall Girls By Full Service Call Girl In Haridwar ESCORTS💦
Demystifying-Gene-Editing-The-Promise-and-Peril-of-CRISPR.pdf

CRISPR-Cas9, a revolutionary gene-editing tool, holds immense potential to reshape medicine, agriculture, and our understanding of life. But like any powerful tool, it comes with ethical considerations.
Unveiling CRISPR: This naturally occurring bacterial defense system (crRNA & Cas9 protein) fights viruses. Scientists repurposed it for precise gene editing (correction, deletion, insertion) by targeting specific DNA sequences.
The Promise: CRISPR offers exciting possibilities:
Gene Therapy: Correcting genetic diseases like cystic fibrosis.
Agriculture: Engineering crops resistant to pests and harsh environments.
Research: Studying gene function to unlock new knowledge.
The Peril: Ethical concerns demand attention:
Off-target Effects: Unintended DNA edits can have unforeseen consequences.
Eugenics: Misusing CRISPR for designer babies raises social and ethical questions.
Equity: High costs could limit access to this potentially life-saving technology.
The Path Forward: Responsible development is crucial:
International Collaboration: Clear guidelines are needed for research and human trials.
Public Education: Open discussions ensure informed decisions about CRISPR.
Prioritize Safety and Ethics: Safety and ethical principles must be paramount.
CRISPR offers a powerful tool for a better future, but responsible development and addressing ethical concerns are essential. By prioritizing safety, fostering open dialogue, and ensuring equitable access, we can harness CRISPR's power for the benefit of all. (2998 characters)
Jaipur ❤cALL gIRLS 89O1183002 ❤ℂall Girls IN JaiPuR ESCORT SERVICE

Jaipur ❤cALL gIRLS 89O1183002 ❤ℂall Girls IN JaiPuR ESCORT SERVICE
Global launch of the Healthy Ageing and Prevention Index 2nd wave – alongside...

The Healthy Ageing and Prevention Index is an online tool created by ILC that ranks countries on six metrics including, life span, health span, work span, income, environmental performance, and happiness. The Index helps us understand how well countries have adapted to longevity and inform decision makers on what must be done to maximise the economic benefits that comes with living well for longer.
Alongside the 77th World Health Assembly in Geneva on 28 May 2024, we launched the second version of our Index, allowing us to track progress and give new insights into what needs to be done to keep populations healthier for longer.
The speakers included:
Professor Orazio Schillaci, Minister of Health, Italy
Dr Hans Groth, Chairman of the Board, World Demographic & Ageing Forum
Professor Ilona Kickbusch, Founder and Chair, Global Health Centre, Geneva Graduate Institute and co-chair, World Health Summit Council
Dr Natasha Azzopardi Muscat, Director, Country Health Policies and Systems Division, World Health Organisation EURO
Dr Marta Lomazzi, Executive Manager, World Federation of Public Health Associations
Dr Shyam Bishen, Head, Centre for Health and Healthcare and Member of the Executive Committee, World Economic Forum
Dr Karin Tegmark Wisell, Director General, Public Health Agency of Sweden
ICH Guidelines for Pharmacovigilance.pdf

The "ICH Guidelines for Pharmacovigilance" PDF provides a comprehensive overview of the International Council for Harmonisation of Technical Requirements for Pharmaceuticals for Human Use (ICH) guidelines related to pharmacovigilance. These guidelines aim to ensure that drugs are safe and effective for patients by monitoring and assessing adverse effects, ensuring proper reporting systems, and improving risk management practices. The document is essential for professionals in the pharmaceutical industry, regulatory authorities, and healthcare providers, offering detailed procedures and standards for pharmacovigilance activities to enhance drug safety and protect public health.
BOWEL ELIMINATION BY ANUSHRI SRIVASTAVA.pptx

Defecation
Normal defecation begins with movement in the left colon, moving stool toward the anus. When stool reaches the rectum, the distention causes relaxation of the internal sphincter and an awareness of the need to defecate. At the time of defecation, the external sphincter relaxes, and abdominal muscles contract, increasing intrarectal pressure and forcing the stool out
The Valsalva maneuver exerts pressure to expel faeces through a voluntary contraction of the abdominal muscles while maintaining forced expiration against a closed airway. Patients with cardiovascular disease, glaucoma, increased intracranial pressure, or a new surgical wound are at greater risk for cardiac dysrhythmias and elevated blood pressure with the Valsalva maneuver and need to avoid straining to pass the stool.
Normal defecation is painless, resulting in passage of soft, formed stool
CONSTIPATION
Constipation is a symptom, not a disease. Improper diet, reduced fluid intake, lack of exercise, and certain medications can cause constipation. For example, patients receiving opiates for pain after surgery often require a stool softener or laxative to prevent constipation. The signs of constipation include infrequent bowel movements (less than every 3 days), difficulty passing stools, excessive straining, inability to defecate at will, and hard feaces
IMPACTION
Fecal impaction results from unrelieved constipation. It is a collection of hardened feces wedged in the rectum that a person cannot expel. In cases of severe impaction the mass extends up into the sigmoid colon.
DIARRHEA
Diarrhea is an increase in the number of stools and the passage of liquid, unformed feces. It is associated with disorders affecting digestion, absorption, and secretion in the GI tract. Intestinal contents pass through the small and large intestine too quickly to allow for the usual absorption of fluid and nutrients. Irritation within the colon results in increased mucus secretion. As a result, feces become watery, and the patient is unable to control the urge to defecate. Normally an anal bag is safe and effective in long-term treatment of patients with fecal incontinence at home, in hospice, or in the hospital. Fecal incontinence is expensive and a potentially dangerous condition in terms of contamination and risk of skin ulceration
HEMORRHOIDS
Hemorrhoids are dilated, engorged veins in the lining of the rectum. They are either external or internal.
FLATULENCE
As gas accumulates in the lumen of the intestines, the bowel wall stretches and distends (flatulence). It is a common cause of abdominal fullness, pain, and cramping. Normally intestinal gas escapes through the mouth (belching) or the anus (passing of flatus)
FECAL INCONTINENCE
Fecal incontinence is the inability to control passage of feces and gas from the anus. Incontinence harms a patient’s body image
PREPARATION AND GIVING OF LAXATIVESACCORDING TO POTTER AND PERRY,
An enema is the instillation of a solution into the rectum and sig
Neuro Saphirex Cranial Brochure

Surgical navigation system introduced by Saphirex Surgiclas. Affordable, precise. Learn more: https://www.rxoom.com/
VVIP Dehradun Girls 9719300533 Heat-bake { Dehradun } Genteel ℂall Serviℂe By...

VVIP Dehradun Girls 9719300533 Heat-bake { Dehradun } Genteel ℂall Serviℂe By Our Agency
The Impact of Meeting: How It Can Change Your Life

The Impact of Meeting: How It Can Change Your Life
Haridwar ❤CALL Girls 🔝 89011★83002 🔝 ❤ℂall Girls IN Haridwar ESCORT SERVICE❤

Haridwar ❤CALL Girls 🔝 89011★83002 🔝 ❤ℂall Girls IN Haridwar ESCORT SERVICE❤
Empowering ACOs: Leveraging Quality Management Tools for MIPS and Beyond

Join us as we delve into the crucial realm of quality reporting for MSSP (Medicare Shared Savings Program) Accountable Care Organizations (ACOs).
In this session, we will explore how a robust quality management solution can empower your organization to meet regulatory requirements and improve processes for MIPS reporting and internal quality programs. Learn how our MeasureAble application enables compliance and fosters continuous improvement.
💘Ludhiana ℂall Girls 📞]][89011★83002][[ 📱 ❤ESCORTS service in Ludhiana💃💦Ludhi...![💘Ludhiana ℂall Girls 📞]][89011★83002][[ 📱 ❤ESCORTS service in Ludhiana💃💦Ludhi...](data:image/gif;base64,R0lGODlhAQABAIAAAAAAAP///yH5BAEAAAAALAAAAAABAAEAAAIBRAA7)
![💘Ludhiana ℂall Girls 📞]][89011★83002][[ 📱 ❤ESCORTS service in Ludhiana💃💦Ludhi...](data:image/gif;base64,R0lGODlhAQABAIAAAAAAAP///yH5BAEAAAAALAAAAAABAAEAAAIBRAA7)
💘Ludhiana ℂall Girls 📞]][89011★83002][[ 📱 ❤ESCORTS service in Ludhiana💃💦Ludhiana Call Girls Service ❤
Nursing Care of Client With Acute And Chronic Renal Failure.ppt

Nursing Care of Client With Acute And Chronic Renal Failure
Recently uploaded (20)
Contact ME {89011**83002} Haridwar ℂall Girls By Full Service Call Girl In Ha...

Contact ME {89011**83002} Haridwar ℂall Girls By Full Service Call Girl In Ha...
Demystifying-Gene-Editing-The-Promise-and-Peril-of-CRISPR.pdf

Demystifying-Gene-Editing-The-Promise-and-Peril-of-CRISPR.pdf
Jaipur ❤cALL gIRLS 89O1183002 ❤ℂall Girls IN JaiPuR ESCORT SERVICE

Jaipur ❤cALL gIRLS 89O1183002 ❤ℂall Girls IN JaiPuR ESCORT SERVICE
Global launch of the Healthy Ageing and Prevention Index 2nd wave – alongside...

Global launch of the Healthy Ageing and Prevention Index 2nd wave – alongside...
VVIP Dehradun Girls 9719300533 Heat-bake { Dehradun } Genteel ℂall Serviℂe By...

VVIP Dehradun Girls 9719300533 Heat-bake { Dehradun } Genteel ℂall Serviℂe By...
The Impact of Meeting: How It Can Change Your Life

The Impact of Meeting: How It Can Change Your Life
Surgery-Mini-OSCE-All-Past-Years-Questions-Modified.

Surgery-Mini-OSCE-All-Past-Years-Questions-Modified.
Haridwar ❤CALL Girls 🔝 89011★83002 🔝 ❤ℂall Girls IN Haridwar ESCORT SERVICE❤

Haridwar ❤CALL Girls 🔝 89011★83002 🔝 ❤ℂall Girls IN Haridwar ESCORT SERVICE❤
Empowering ACOs: Leveraging Quality Management Tools for MIPS and Beyond

Empowering ACOs: Leveraging Quality Management Tools for MIPS and Beyond
💘Ludhiana ℂall Girls 📞]][89011★83002][[ 📱 ❤ESCORTS service in Ludhiana💃💦Ludhi...![💘Ludhiana ℂall Girls 📞]][89011★83002][[ 📱 ❤ESCORTS service in Ludhiana💃💦Ludhi...](data:image/gif;base64,R0lGODlhAQABAIAAAAAAAP///yH5BAEAAAAALAAAAAABAAEAAAIBRAA7)
![💘Ludhiana ℂall Girls 📞]][89011★83002][[ 📱 ❤ESCORTS service in Ludhiana💃💦Ludhi...](data:image/gif;base64,R0lGODlhAQABAIAAAAAAAP///yH5BAEAAAAALAAAAAABAAEAAAIBRAA7)
💘Ludhiana ℂall Girls 📞]][89011★83002][[ 📱 ❤ESCORTS service in Ludhiana💃💦Ludhi...
Nursing Care of Client With Acute And Chronic Renal Failure.ppt

Nursing Care of Client With Acute And Chronic Renal Failure.ppt
Handout-Structure-of-med-res-paper-RG-checklists_FINAL.pdf
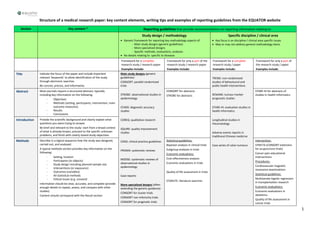
- 1. 1 Structure of a medical research paper: key content elements, writing tips and examples of reporting guidelines from the EQUATOR website Section Key content * Reporting guidelines that provide recommendations on reporting information relating to: Study design / methodology Generic framework for reporting key methodology aspects of: - Main study designs (generic guidelines) - More specialised designs - Specific methods, evaluations, analyses No details relating to specific to diseases Specific discipline / clinical area Key focus is on discipline / clinical area specific issues May or may not address general methodology items Framework for a complete research study / research paper Examples include: Framework for only a part of the research study / research paper Examples include: Framework for a complete research study / paper Examples include: Framework for only a part of the research study / paper Examples include: Title Indicate the focus of the paper and include important relevant ‘keywords’ to allow identification of the study through electronic searches. Be concise, precise, and informative. Main study designs (generic guidelines): CONSORT: parallel randomised trials STROBE: observational studies in epidemiology STARD: diagnostic accuracy studies COREQ: qualitative research SQUIRE: quality improvement studies COGS: clinical practice guidelines PRISMA: systematic reviews MOOSE: systematic reviews of observational studies in epidemiology Case reports More specialised designs (often extending the generic guidance): CONSORT for cluster trials CONSORT non-inferiority trials CONSORT for pragmatic trials TREND: non-randomised studies of behavioural and public health interventions REMARK: tumour marker prognostic studies STARE-HI: evaluation studies in health informatics Longitudinal studies in rheumatology Adverse events reports in traditional Chinese medicine Case series of colon tumours Abstract Most journals require a structured abstract, typically including key information on the following: - Objectives - Methods (setting, participants, intervention, main outcome measures) - Results - Conclusions CONSORT for abstracts STROBE for abstracts STARE-HI for abstracts of studies in health informatics Introduction Provide the scientific background and clearly explain what questions you were trying to answer. Be brief and relevant to the study: start from a broad context of what is already known, proceed to the specific unknown problems, and finish with clearly stated study objectives Methods Describe in a logical sequence how the study was designed, carried out, and analysed. A typical methods section provides key information on the following: - Setting, location - Participants (or objects) - Study design including planned sample size - Interventions (or exposures) - Outcomes (variables) - All statistical methods - Ethical issues (e.g. consent) Information should be clear, accurate, and complete (provide enough details to repeat, assess, and compare with other studies) Content should correspond with the Result section Statistical guidelines: Bayesian analysis in clinical trials Subgroup analyses in trials Economic evaluations: Cost-effectiveness analysis Economic evaluations in trials Quality of life assessment in trials STARLITE: literature searches Intervention: STRICTA (CONSORT extension for acupuncture trials) Cancer pain educational interventions Procedures: Cardiovascular magnetic resonance examinations Statistical guidelines: Multivariate logistic regression in transplantation research Economic evaluations: Economic evaluations in obstetrics Quality of life assessment in cancer trials
- 2. 2 Section Key content * Reporting guidelines that provide recommendations on reporting information relating to: Study design / methodology Generic framework for reporting key methodology aspects of: - Main study designs (generic guidelines) - More specialised designs - Specific methods, evaluations, analyses No details relating to specific to diseases Specific discipline / clinical area Key focus is on discipline / clinical area specific issues May or may not address general methodology items Framework for a complete research study / research paper Examples include: Framework for only a part of the research study / research paper Examples include: Framework for a complete research study / paper Examples include: Framework for only a part of the research study / paper Examples include: Results Report results of the investigations described in the Methods section (in same order) using text, tables, figures, and statistics Be as brief and clear as possible (but don’t leave out ‘disappointing’ results) A typical structure and chronology includes: - Description of study participants (if relevant, separately for important subgroups) - Presentation of answers to the main questions (starting with primary outcomes, then secondary outcomes, and any other analyses) Report on harms (adverse effects) as well as benefits, if relevant Outline any ways in which the study did not follow the prespecified plan. Pay special attention to presentation of data and results in tables and figures Statistical guidelines: Heterogeneity in trial treatment effects Missing data Figures, graphs Statistical results in case- control designs in neuropsychology Discussion Discuss what your findings mean and where they stand in the context of other studies A typical discussion section structure and chronology includes: - Brief presentation of the main findings - Assessment of study strengths and weaknesses - Comparison of findings with previous studies - Consideration of clinical and scientific implications - If relevant, suggestions for future research Structured discussion Research recommendations Conclusions This section is not always presented separately in a research article Any conclusions must be fully supported by the study findings Acknowledge ments State source of funding and any relevant conflict of interest Acknowledge any person who contributed to the study but who does not qualify as an author Conflict of Interest: ICMJE, WAME guidelines * Key content column lists basic general content of a primary research paper