The document contains a 4 question chemistry assessment with the following key details:
1) Rate of a chemical reaction can be determined by measuring the volume of gas liberated per unit time.
2) A table shows the volume of oxygen gas liberated over time intervals when manganese (IV) oxide is added to hydrogen peroxide. The rate of decomposition decreases with time and the total volume is 54 cm3.
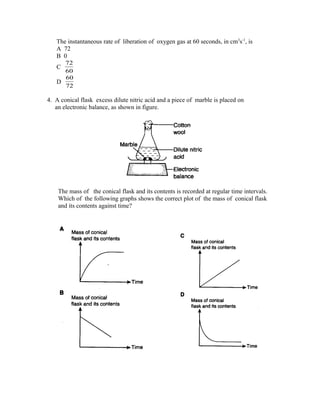
3) A graph shows the volume of oxygen gas liberated over time when manganese (IV) oxide is added as a catalyst to sodium chlorate solution. The instantaneous rate at 60 seconds is 72 cm3/s.