This document presents the results of an experiment and simulation investigating the properties of turbulent jets. Laser-induced fluorescence and particle image velocimetry were used to capture experimental data, while MOBILE software was used to simulate jets. Inlet velocity, virtual origin, and other properties were estimated from the experimental and simulation data using image processing and analysis in MATLAB. The results from experiment and simulation were found to correspond reasonably while some error was present.


![Abstract
A moving fluids, turbulent jet entering a quiescent body of the same
fluids is modelled by both experiment and simulation. In experiment, data
is captured by camera and simulation images are produced by MOBILE.
MATLAB is used to analyse the data from experimental and simulation
in order to calculated the inlet velocity, virtual origin, attenuation con-
stant, dilution rate and Reynold’s number. The results on experimental
and simulation are shown to be corresponding to each other while some
error is present which affects the reliability of the results.
1 Introduction
Turbulent jets are fluid flows produced by a pressure drop through an orifice[1].
In this experiment, a moving fluid exits an orifice and enters a quiescent body
of the same fluid, the difference between the entering and ambient fluids create
a velocity shear which cause turbulence and mixing[2].
In this experiment, the jet formed by the Rhodamine dye solution, em-
anates from a small nozzle of width d whose axis is parallel with the z axis
which is shown in Figure 1. It then spreads out from the nozzle and forming
a well-defined boundary, which is highly turbulent in the inner layer and is
non-turbulent ambient fluid in the outer layer [3]. For the reason of flow visu-
alisation, the method of Laser Induced Fluorescence is used to investigate the
properties of the jet while the method of Particle Image Velocimetry is used to
obtain the instantaneous velocity in fluids, by adding the particles that acts as
a tracer which will glisten under laser light.
The aim of this report is to investigate the properties of turbulent jet includ-
ing virtual origin, spatial rate of dilution , entrainment velocity and Reynold’s
number and by comparing the results between the simulation, theoretical and
experimental.
Figure 1: An image illustrates the characteristic of jet [3]
3](https://image.slidesharecdn.com/6a0d7a5b-13bb-4899-9a28-0898f536a456-160705152855/85/Fluids-3-Report-es13906-rt13074-kp13594-3-320.jpg)













![4 Conclusion
The main objective of this investigation in this report is to compare the dif-
ferences between the theoretical results and the simulation and experimental
results under different aspects, including the inlet velocity, spread rate, virtual
origin, dilution rate and the entrainment velocity.
For the inlet velocity, it can be proved that both experimental and simula-
tion data shows the trend that a faster jets will give a smaller inlet angle. Some
error can be observed as the experimental data is expected to be larger.
The MOBILE simulations and experimental observations for the virtual ori-
gin appear to support one another and it also proved that the virtual source does
not change with velocity. However, ”truncation error” is present as the standard
iterative process used to solve parabolic equations relies on Taylor expansions of
differential equations, and frequently discount any terms of third order or higher.
Attenuation constant can be found according to Lambert Beer’s law. It
is found that the maximum error between the aC value to the mean value is
about 3.30%. Therefore, it is not required to know a independently as the error
is small. Moreover, both experimental and simulation results in dilution rate
are similar. However, because of the size of the nozzle, the peak value of the
graph from the experimental results appear to be sharper.
Particle Image Velocimetry is the method used to analysis the entrainment
velocity. However, due to certain degree of statistical noise in the results, which
makes the results are not reliable enough.
In conclusion, it shows that both simulation data and experimental data
support each other. However, apart from the truncation error mentioned above,
the error that existed in the experimental data can be explained by the poor
quality of image, human error in experiment and noise existed in the image. To
improve this, some opaque plastic sheet can be placed beside the glass tank to
avoid noise from the surrounding and nevertheless, ensure the gas bubbles are
wiped off from the glass tank in order to improve the quality of image.
References
[1] E. J .List, 1982 Turbulent Jets And Plumes [Online], California Institute of
Technology
Available from: http://www.annualreviews.org/doi/pdf/10.1146/
annurev.fl.14.010182.001201 [Accessed 21st March 2016]
[2] Cushman-Roisin, B.C.R. , 2014 Environmental Fluid Mechanics [Online],
Thayer School of Engineering, Dartmouth College
Available from: https://engineering.dartmouth.edu/~d30345d/books/
EFM/chap9.pdf [Accessed 21st March 2016]
22](https://image.slidesharecdn.com/6a0d7a5b-13bb-4899-9a28-0898f536a456-160705152855/85/Fluids-3-Report-es13906-rt13074-kp13594-22-320.jpg)
![[3] Lawrie, A.G.W, 2016 Submerged Turbulent Jet [Online], University of
Bristol
Available from: https://www.ole.bris.ac.uk/bbcswebdav/
pid-2395821-dt-content-rid-6510483_2/courses/MENG30001_2015/
jet_similarity%282%29.pdf [Accessed 21st March 2016]
5 Appendix
5.1 MOBILE input
begin();
usempi:=assign(ON);
igmx:=assign(2);
jgmx:=assign(igmx);
kgmx:=assign(igmx*2);
tnx:=assign(256);
tny:=assign(tnx);
tnz:=assign(tnx*2);
xmax:=assign(1.0);
ymax:=assign(xmax);
zmax:=assign(xmax*2);
mgrids:=assign(7);
cgrids:=assign(1);
scorder:=assign(LONEDENSITY);
hsorder:=assign(SECOND);
nscalars:=assign(2);
nheights:=assign(4);
writestrat:=assign(OFF);
writescalars:=assign(OFF);
#istop:=assign(20);
iprint:=assign(1000);
#tprint:=assign(0.50);
tstop:=assign(200);
tderive:=assign(0.25);
#iderive:=assign(180);
postmem:=assign(DYNAMIC);
dtmod:=assign(OFF);
divtol:=assign(2.4e-2);
petol:=assign(1e-4);
pdtol:=assign(1e-12);
pctol:=assign(1e-12);
#
topbc:=assign(FORCEDINFLOW);
botbc:=assign(INDUCEDOUTFLOW);
leftbc:=assign(SLIPWALL);
rightbc:=assign(SLIPWALL);
frontbc:=assign(SLIPWALL);
backbc:=assign(SLIPWALL);
bcprofile:=assign(TOPHAT);
23](https://image.slidesharecdn.com/6a0d7a5b-13bb-4899-9a28-0898f536a456-160705152855/85/Fluids-3-Report-es13906-rt13074-kp13594-23-320.jpg)
![writebounds:=assign(ON);
writebcprofile:=assign(ON);
writeppert:=assign(OFF);
readppert:=assign(OFF);
bcbaseflow:=assign(-0.4);
writebcmean:=assign(ON);
bcturbamp:=assign(0.0);
bcturbvel:=assign(0.0);
bccoflowvel:=assign(0.0);
bccoflowrad:=assign(0.05);
#
#readchkfile:=assign(ON);
#istart:=assign(100);
#tstart:=assign(9.091178);
tlimit:=assign(4);
readtlimfile:=assign(ON);
end();
5.2 The MATLAB code
5.2.1 Section 3.1 - 3.2, 3.5-3.6
function timeaverage2(list)
%demonstration of time−average processing
nfiles=size(list); %find the size of the list passed in
nfiles=nfiles(2); % assumes 2D matrix, so take second element
for i=1:nfiles
%this will need editing for simulation data
infile=sprintf('DSC %04d.JPG',list(i));
outfile=sprintf('bw %04d.bmp',list(i));
imdata=imread(infile); % read image from file
% image(imdata); % output image to screen
imdata=double(imdata);
sz=size(imdata); % find the size of the image
imbw=zeros(sz(1),sz(2)); % set up a new B&W image array of the same size
imbw=sqrt((imdata(:,:,1).*imdata(:,:,1))+(imdata(:,:,2).*imdata(:,:,2))+(imdata(:,:,3).*imdata(:,:,
mx=max(max(imbw)); %find max value
mn=min(min(imbw)); %find min value
if (mx−mn)>0 %protect in case image is all zeros
imbw=(imbw−mn)/(mx−mn); %normalise to [0,1]
end
% image(imbw) %plot the scalar image in matlab colour scheme
if i==1
immean=imbw; % initialise the cumulative count to zero
else
immean=immean+imbw; %add one image each time.
end
immono=zeros(sz(1),sz(2),3);
immono(:,:,1)=imbw; %use the same value for Red
immono(:,:,2)=imbw; % Green
immono(:,:,3)=imbw; % Blue
imwrite(immono,outfile,'bmp'); %write out new greyscale file
% image(immono)
end
immean=immean/nfiles; %take the average intensity (outside the loop)
24](https://image.slidesharecdn.com/6a0d7a5b-13bb-4899-9a28-0898f536a456-160705152855/85/Fluids-3-Report-es13906-rt13074-kp13594-24-320.jpg)

![X=[ones(1,ny); xmoment]';
Y=yidx';
A1=(X'*Y);
A2=(X'*X);
A=inv(A2)*A1;
hold on
b=A(2,1);
a=A(1,1);
xfit=((yidx−a)/b);
plot(xfit,yidx);
axis([0 100 0 600])
legend('Actual jet shape','Least−squares mean shape')
xlabel('Axial depth')
ylabel('Radial distance')
text(20,300,['a=',num2str(a)])
text(20,250,['b=',num2str(b)])
figure(2);
background=imread('background.png');
image(background)
colormap(gray)
% Converting back to image space
X=[0:1:200];
X0=1;
X1=X−24;
hold on
d=−b;
c=930;
Y=c+(d*X1);
Y0=Y(1,1);
theta=atan(200/(2570−Y0));
thetad=theta*(180/pi);
plot(X,Y,'−b',X0,Y0,'or')
legend('Jet profile','Virtual jet origin')
text(300,500,['Jet angle is ',num2str(thetad),' degrees'],'color','red')
% Isolate an image of just the right hand side of the jet
immean2=imread('exp mean.bmp');
immean2=double(immean2);
immean2=immean2(709:2570,1800:3030);
sz=size(immean2);
% Find five depths within the jet
dz=sz(1)/10;
xmax=sz(2);
Z=round([2*dz 3*dz 4*dz 5*dz 6*dz]);
X2=[1:1:xmax];
% 2090 pixels = 250 mm
% 1 pixel = 0.12 mm
D=Z*0.12;
RZ=X2*0.12;
26](https://image.slidesharecdn.com/6a0d7a5b-13bb-4899-9a28-0898f536a456-160705152855/85/Fluids-3-Report-es13906-rt13074-kp13594-26-320.jpg)
![% Take five slices out of the concentration field
C1=immean2(Z(1,1),:);
C2=immean2(Z(1,2),:);
C3=immean2(Z(1,3),:);
C4=immean2(Z(1,4),:);
C5=immean2(Z(1,5),:);
% Plot concentration profiles
figure(3)
plot(RZ,C1,RZ,C2,RZ,C3,RZ,C4,RZ,C5)
legend(['z=',num2str(D(1,1)),'mm'],['z=',num2str(D(1,2)),'mm'],...
['z=',num2str(D(1,3)),'mm'],['z=',num2str(D(1,4)),'mm'],...
['z=',num2str(D(1,5)),'mm'])
xlabel('r(z)')
ylabel('C(z)')
% Estimate radii
R=D*tan(theta);
% Normalise concentration profiles to check for similarity
RN1=RZ/R(1,1);
RN2=RZ/R(1,2);
RN3=RZ/R(1,3);
RN4=RZ/R(1,4);
RN5=RZ/R(1,5);
CN1=C1/max(C1);
CN2=C2/max(C2);
CN3=C3/max(C3);
CN4=C4/max(C4);
CN5=C5/max(C5);
figure(4)
plot(RN1,CN1,RN2,CN2,RN3,CN3,RN4,CN4,RN5,CN5)
legend(['z=',num2str(D(1,1)),'mm'],['z=',num2str(D(1,2)),'mm'],...
['z=',num2str(D(1,3)),'mm'],['z=',num2str(D(1,4)),'mm'],...
['z=',num2str(D(1,5)),'mm'])
axis([0 1 0 1])
xlabel('r(z)/r')
ylabel('C(z)/C0')
end
5.2.2 Section 3.3 , 3.4
function attenuation()
clc
clear
filemin=1; %put your own favourite file number in here
filemax=3;
ifile=0;
xlaser=32; %source position
zlaser=0; %source position
divergence=120; %spread angle of laser sheet
nrays=121; %refinement of ray casting
raylength=2000; %how many increments along ray
dzray=1; % vertical increment of ray on grid
a=−0.0001; %attenuation cofficient proportional to concentration
b=−0.00043; %background level of attenuation
27](https://image.slidesharecdn.com/6a0d7a5b-13bb-4899-9a28-0898f536a456-160705152855/85/Fluids-3-Report-es13906-rt13074-kp13594-27-320.jpg)


![pray(ir,:)=ptmp; %update final intensity array
end
figure (1)
contour(zray,xray,pray);
contour(scal);
pray mean(i,:)=mean(pray,2); % separate three intensity
%section 3.4 dilution rate
% figure(2)
% contour(zray,xray,pray,100);
% hold on
% contour(scal);
% hold off
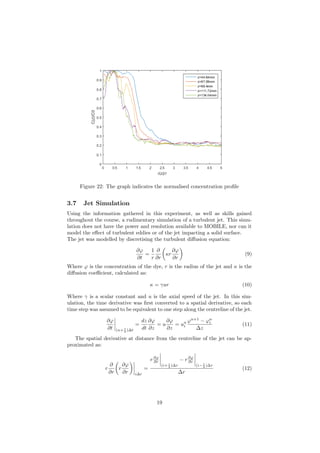
% title('Graph of jet and position of laser ray')
% xlabel('Distance jet travels in z direction')
% ylabel('Radius of jet in x direcion')
% set(gcf,'color','white');
end
end
%section 3.3
% Attenuation constant
%%%%%%%%%%%%%%%%%%%%%%%%%%%%%%%%%%%
figure(1)
s=[1:2000];
s=s';
%1st
y1=(pray mean(1,:))';
%2nd
y2=(pray mean(2,:))';
%3rd
y3=(pray mean(3,:))';
%taking natural log to the intensity
y1=log(y1);
y2=log(y2);
y3=log(y3);
%plotting graph from the raw data
figure (1)
f1= plot(s,y1,'b','LineWidth', 2);
f1 r= get(f1,'YData');
hold on
f2=plot(s,y2,'r','LineWidth', 2);
f2 r= get(f2,'YData');
f3= plot(s,y3,'g','LineWidth', 2);
f3 r= get(f3,'YData');
title('raw')
%plotting graph for the best fit
figure (2)
p=polyfit(s,y1,1);
f1 = polyval(p,s);
plot(s,f1,'b','LineWidth', 2)
hold on
p2=polyfit(s,y2,1);
f2 = polyval(p2,s);
plot(s,f2,'r','LineWidth', 2)
p3=polyfit(s,y3,1);
f3 = polyval(p3,s);
plot(s,f3,'g','LineWidth', 2)
xlabel('Distance (pixel)')
30](https://image.slidesharecdn.com/6a0d7a5b-13bb-4899-9a28-0898f536a456-160705152855/85/Fluids-3-Report-es13906-rt13074-kp13594-30-320.jpg)
![ylabel('In (Intensity)')
title('In(Intensity) against distance')
% plotting the aC against distance, to show small fluctuation of the
% graphs
figure (3)
f1 diff=diff(f1 r);
f2 diff=diff(f2 r);
f3 diff=diff(f3 r);
s=[1:1999];
s=s';
subplot(3,1,1)
plot(s,f1 diff,'b')
hold on
f1 dmean 1=mean(f1 diff)
f1 dmean=zeros(size(s'));
f1 dmean(1,:)=f1 dmean 1;
plot(s,f1 dmean,'r')
% axis([ 0 2000 −0.010 0 ])
xlabel('distance (pixel)')
ylabel('aC 1')
title('aC 1 against distance')
hold off
subplot(3,1,2)
plot(s,f2 diff,'b')
hold on
f2 dmean 1=mean(f2 diff)
f2 dmean=zeros(size(s'));
f2 dmean(1,:)=f2 dmean 1;
plot(s,f2 dmean,'r')
%axis([ 0 2000 −0.010 0 ])
xlabel('distance (pixel)')
ylabel('aC 2')
title('aC 2 against distance')
hold off
subplot(3,1,3)
plot(s,f3 diff,'b')
hold on
f3 dmean 1=mean(f3 diff)
f3 dmean=zeros(size(s'));
f3 dmean(1,:)=f3 dmean 1;
plot(s,f3 dmean,'r')
%axis([ 0 2000 −0.010 0 ])
xlabel('distance (pixel)')
ylabel('aC 3')
title('aC 3 against distance')
hold off
end
%%%%%%%%%%%%%%%%%%%%%%%%%%%%%%%%%%%
%section 3.4
%%%%%%%%%%%%%%%%%%%%%%%%%%%%%%%%%%%
%
%1.Code for plot dilution rate for simulation
p rate= size(scal);
y = scal(p rate(1,1)/2,:)
x= 1:p rate(1,2);
figure(3)
plot(x,y)
xlabel('Distance in z direction')
ylabel('Relative concentration')
title('Dilution rate of centre of jet for 0.44m/s') set(gcf,'color','white');
hold on
%
31](https://image.slidesharecdn.com/6a0d7a5b-13bb-4899-9a28-0898f536a456-160705152855/85/Fluids-3-Report-es13906-rt13074-kp13594-31-320.jpg)
![%2.Code for plot dilution rate for experiment
%p23=scal(725,:);
%plot(p23);
%xlabel('Distance in z direction')
%ylabel('Relative concentration')
%title('Dilution rate of centre of jet for 0.44m/s')
%set(gcf,'color','white');
%
%Plot best fit line of dilution rate of simulation
%size(x);
%ones(size(x));
%x1=[ones(size(x));x];
%y1=y';
%A1=x1*y1;
%A2=x1*x1';
%A=inv(A2)*A1;
%b=A(2,1);
%a=A(1,1); %xfit=(y?a)/b; %plot(xfit,y) %
%3.Code for plot concentration Field for simulation
figure(4)
hold on
for i= 5:40:245;
y2= scal(:,i);
x2= 1:128;
plot(x2,y2);
xlabel('Distance along x direction (Radius of jet)')
ylabel('Relative concentration')
title('Concentration field for 0.44m/s')
set(gcf,'color','white');
end
%
%4.Code for plot concentration Field for experiment
%figure(4)
%hold on
%for i= 5:200:1401;
% y2= scal(:,i);
% x2= 1:1401;
%%%%%%%%%%%%%%%%%%%%%%%%%%%%%%%%%%%
32](https://image.slidesharecdn.com/6a0d7a5b-13bb-4899-9a28-0898f536a456-160705152855/85/Fluids-3-Report-es13906-rt13074-kp13594-32-320.jpg)