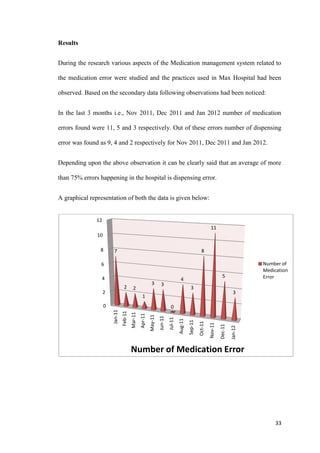
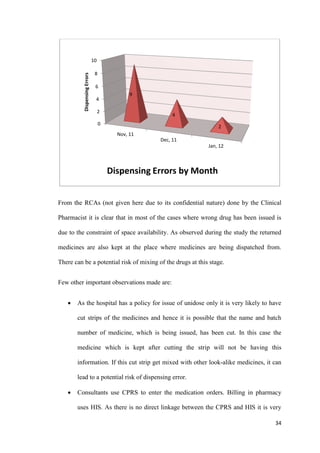
This document discusses medication errors that can occur in hospitals. It defines medication errors as any error in prescribing, dispensing, or administering drugs, regardless of whether harm occurs. Medication errors are a major cause of preventable patient harm. The document classifies medication errors as mistakes, slips, or lapses, depending on where the error occurs in the medication use process. It also discusses different ways medication errors have been estimated to cause deaths in other countries to highlight the significant impact of these errors.