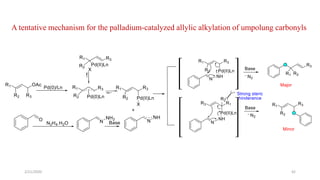
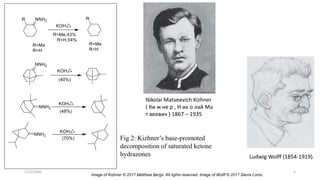
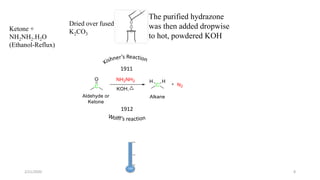
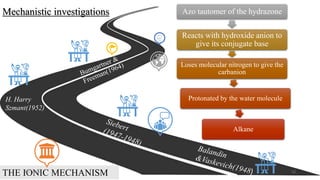
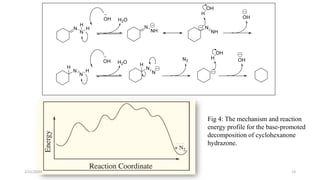
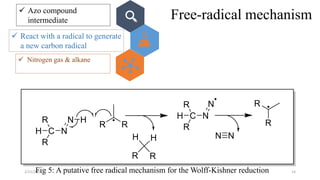
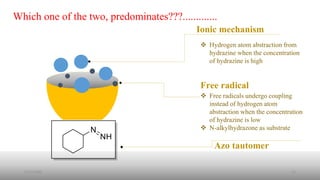
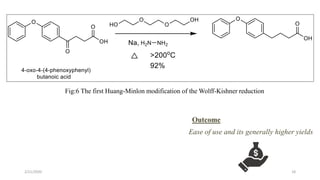
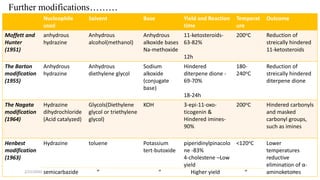
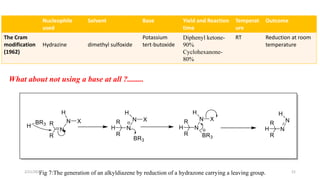
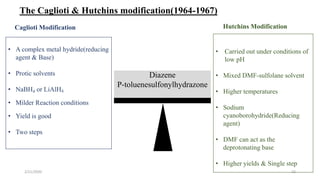
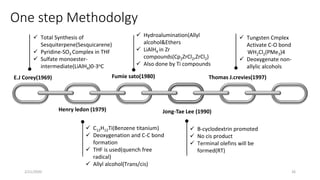
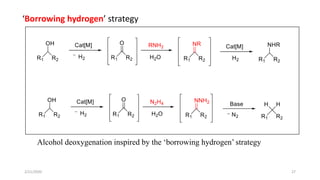
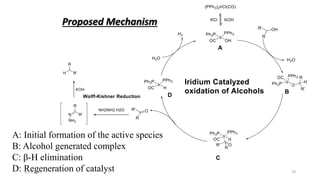
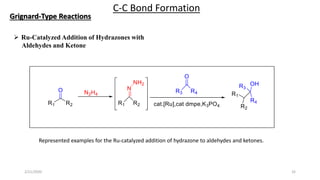
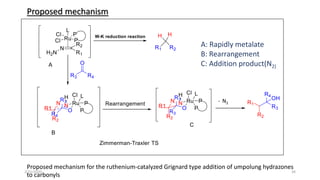
The document summarizes the history and developments of the Wolff-Kishner reduction reaction. It describes how Nikolai Kizhner and Ludwig Wolff independently discovered the reaction in 1912 for carbonyl deoxygenation. Over time, various modifications were made to reduce harsh reaction conditions and improve yields, including those by Huang-Minlon, Barton, Nagata, Henbest, Cram, and Caglioti. The reaction mechanism was investigated and both ionic and free radical pathways were proposed. The Wolff-Kishner reduction has been applied in natural product synthesis and the development of new one-step alcohol deoxygenation methods continues to be an area of active research.


![HISTORY AND EMERGENCE
Kizhner, Professor of Organic Chemistry at the Imperial Tomsk Technological Institute, in
Siberia(1912).Published his report in the Zhurnal Russkogo Fiziko-Khimicheskogo
Obshchestva [Journal of the Russian Physical-Chemical Society]
Wolff, working at the Chemical Institute of Jena University, published a variant of the
same reaction in Justus Liebigs Annalen der Chemie(1912)
The reaction became known as the Wolff reduction until January 10, 1913, when Wolff
acknowledged Kizhner’s priority for the discovery.
In the first disclosure, Kizhner reported the Four deoxygenations.
2/21/2020 4](https://image.slidesharecdn.com/anolddogwithnewtricks-200221105834/85/Enjoin-wolff-kishner-reduction-4-320.jpg)
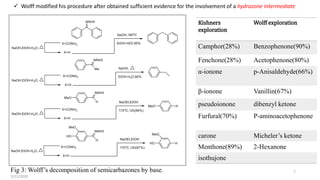
![Fig 2: Wolff’s first report of a semicarbazone decomposition
The first reaction reported by Wolff was the conversion of the quinone
monosemicarbazone into the phenol
Ironically, the paper was entitled, “Method for replacing the oxygen atom of ketones and
aldehydes by hydrogen. [First paper.]” No second paper appeared.
Wolff describes that the inspiration for his discovery was an observation by Johannes
Thiele and his student, Willy Barlow.
2/21/2020 6](https://image.slidesharecdn.com/anolddogwithnewtricks-200221105834/85/Enjoin-wolff-kishner-reduction-6-320.jpg)





![Bergmann and Orchin(1949)
Bartlett and
Knox(1939)
Sargent(1957)
Gates(1950)Rapoport(1960)
Zhilkibaev(2007)
Determination of the stereochemistry of the
Rauwolfia indole alkaloid, rauwolscine
Pelletier(1954)
3-Ethylacridine has been obtained by reduction of 3-
acetylacridine,reduction of 3-acetyl-9,10 dihydroacridine
thienyl ketones, as well
aldehydes
The reduction of the keto-
lactam to the lactam
Fieser(1948)
Stenhagen(1949)
Non-conjugated unsaturated linear keto-
esters and keto-amides to the straight-chain
carboxylic acid derivatives.
Cason(1949)
G.M. Badger(1948)
Branched-chain ketoacids(β,β-
Dialkyl-γ-ketoesters)
synthesis of the geometric
isomers of hexahydrochrysene
from the diketones
Preparation of the bornane-1-
carboxylic acid by reduction of the
2-bornanone-1-carboxylic acid
Synthesis of cyclobutane
from cyclobutanone
structural and stereochemical studies of the
diterpenes of Agathis australis
Fluorene-1-carboxylic acid from
reduction of the keto-acid
Enzell and Thomas(1965)
Roberts and Sauer(1949) Buu-Ho(1953)
Reduction of the geissoschizine
aldehyde to the methyl compound
His proofs of structure and
stereochemistry(veratrine)
The reduction of 3-aryl-2-azabicyclo[4.4.0]-decan-5-
ones gave good yields of the perhydroisoquinolines
Chatterjee and Prakash(1954)
Long-chain linear keto-acids to
the straight-chain carboxylic acids
2/21/2020 19](https://image.slidesharecdn.com/anolddogwithnewtricks-200221105834/85/Enjoin-wolff-kishner-reduction-19-320.jpg)


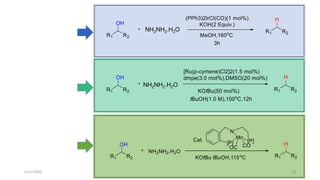
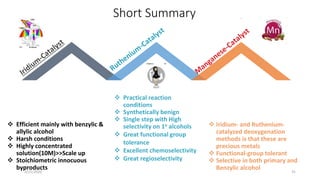


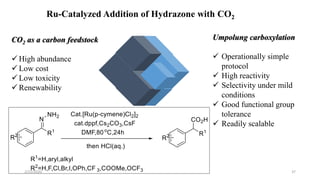
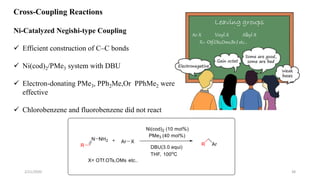
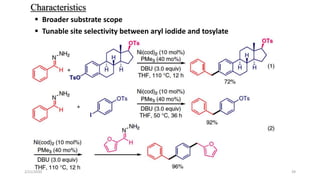
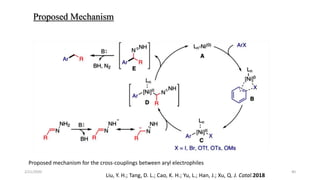
![Characteristics
[Ru(p-cymene)Cl2]2 providing the highest yield>>> The least expensive one
Favored by electron-rich phosphines(1,2-bis(dimethylphosphino)ethane (dmpe) and
tri(tert-butyl)-Phosphine
ligand-to-metal ratio(1:1)
K3PO4(Superior reactivity)
Cesium fluoride(CeF)>>>Additive
Removal of a chloride(Silver triflate)
Scope of Substrates
Aromatic aldehydes showed good reactivity
Halide substituents had a minor influence on the reactivity(Ortho-Least reactive)
Aromatic
Aliphatic
Heterocyclic
2/21/2020 36](https://image.slidesharecdn.com/anolddogwithnewtricks-200221105834/85/Enjoin-wolff-kishner-reduction-36-320.jpg)