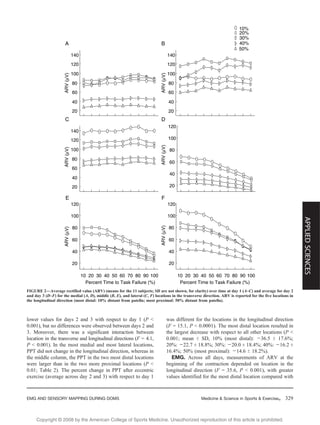
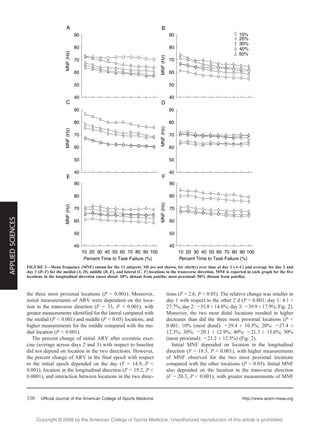
This study investigated changes in sensory and electromyography (EMG) measurements over different regions of the quadriceps muscle before and after eccentric exercise intended to induce delayed-onset muscle soreness (DOMS). Surface EMG signals and pressure pain thresholds were measured at 15 locations on the quadriceps during sustained isometric contractions and at rest. After eccentric exercise, time to failure during contractions, EMG amplitude, and pain thresholds all decreased significantly and were lowest in the distal region of the quadriceps. This suggests DOMS manifestations vary by muscle region, likely due to differences in fiber morphology and architecture. The distal quadriceps may be more susceptible to further injury after eccentric exercise.