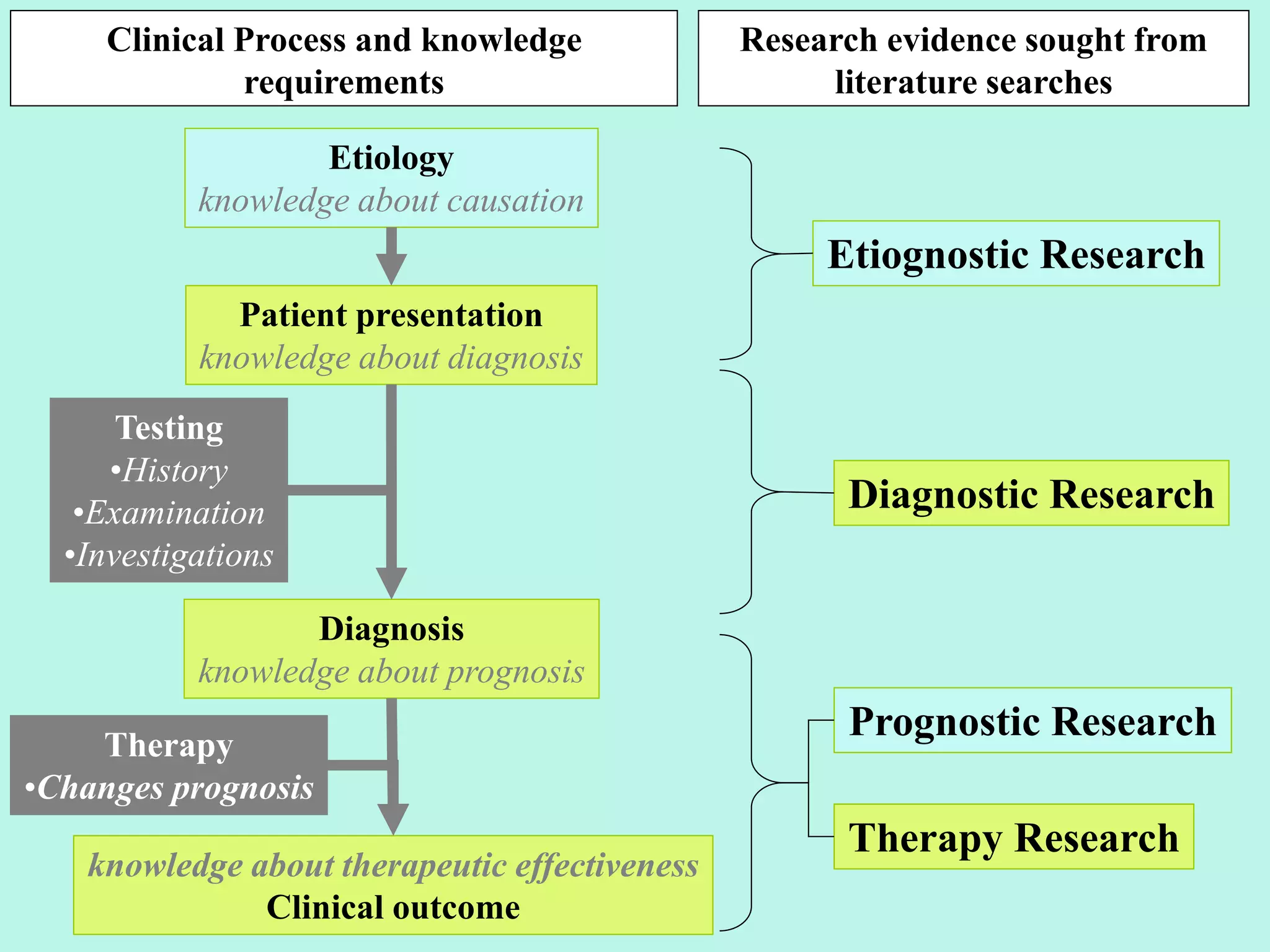
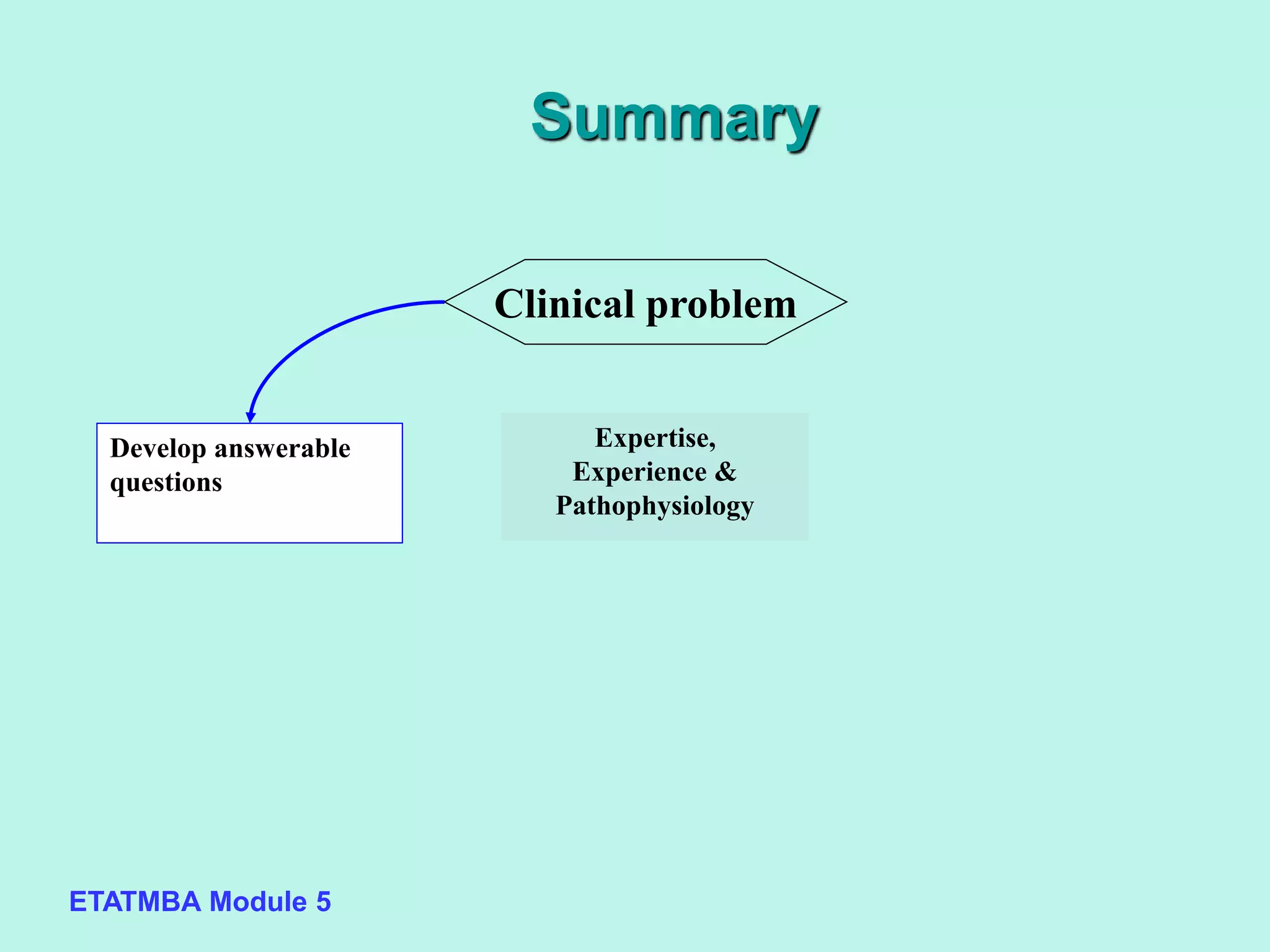
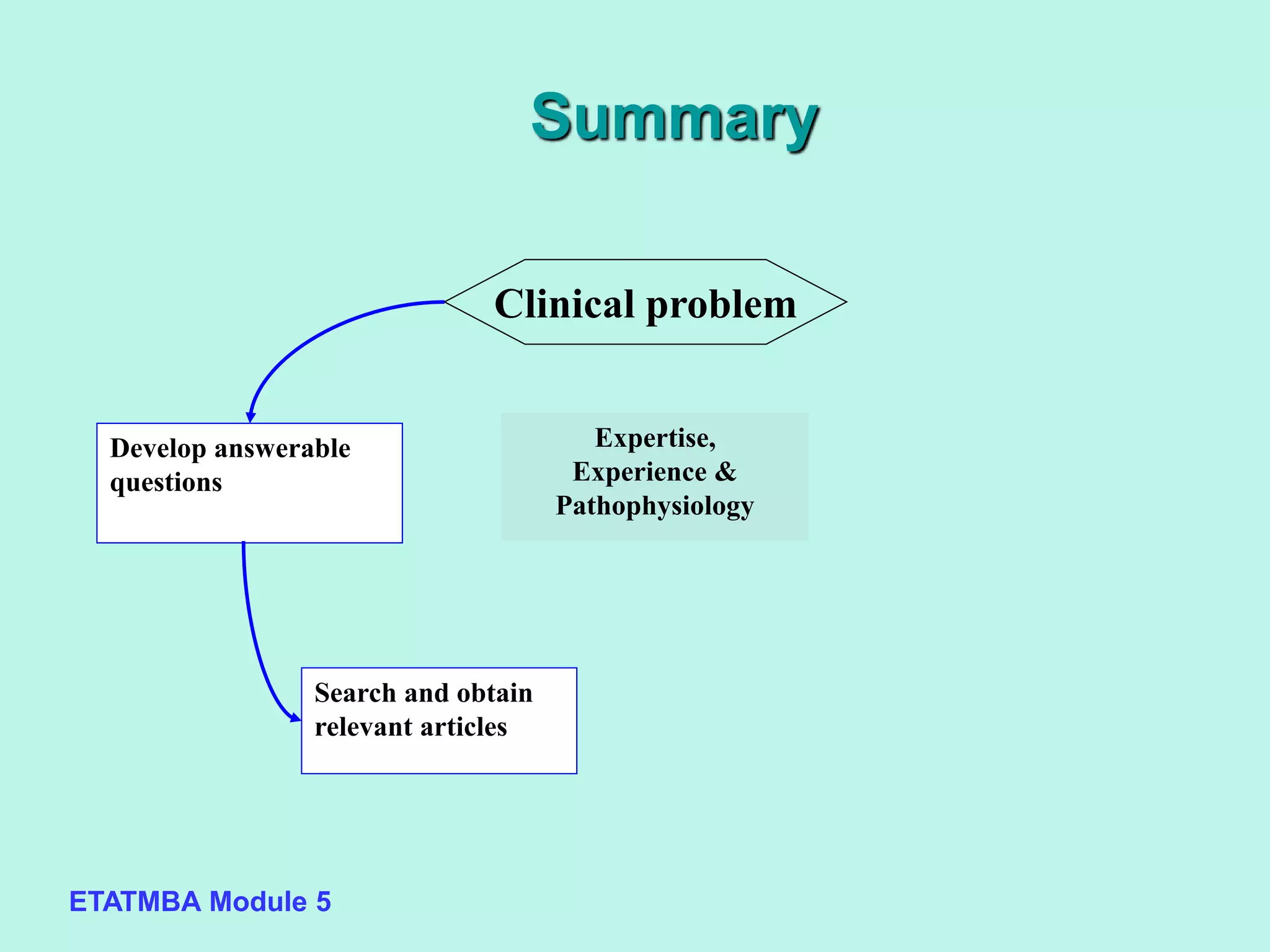
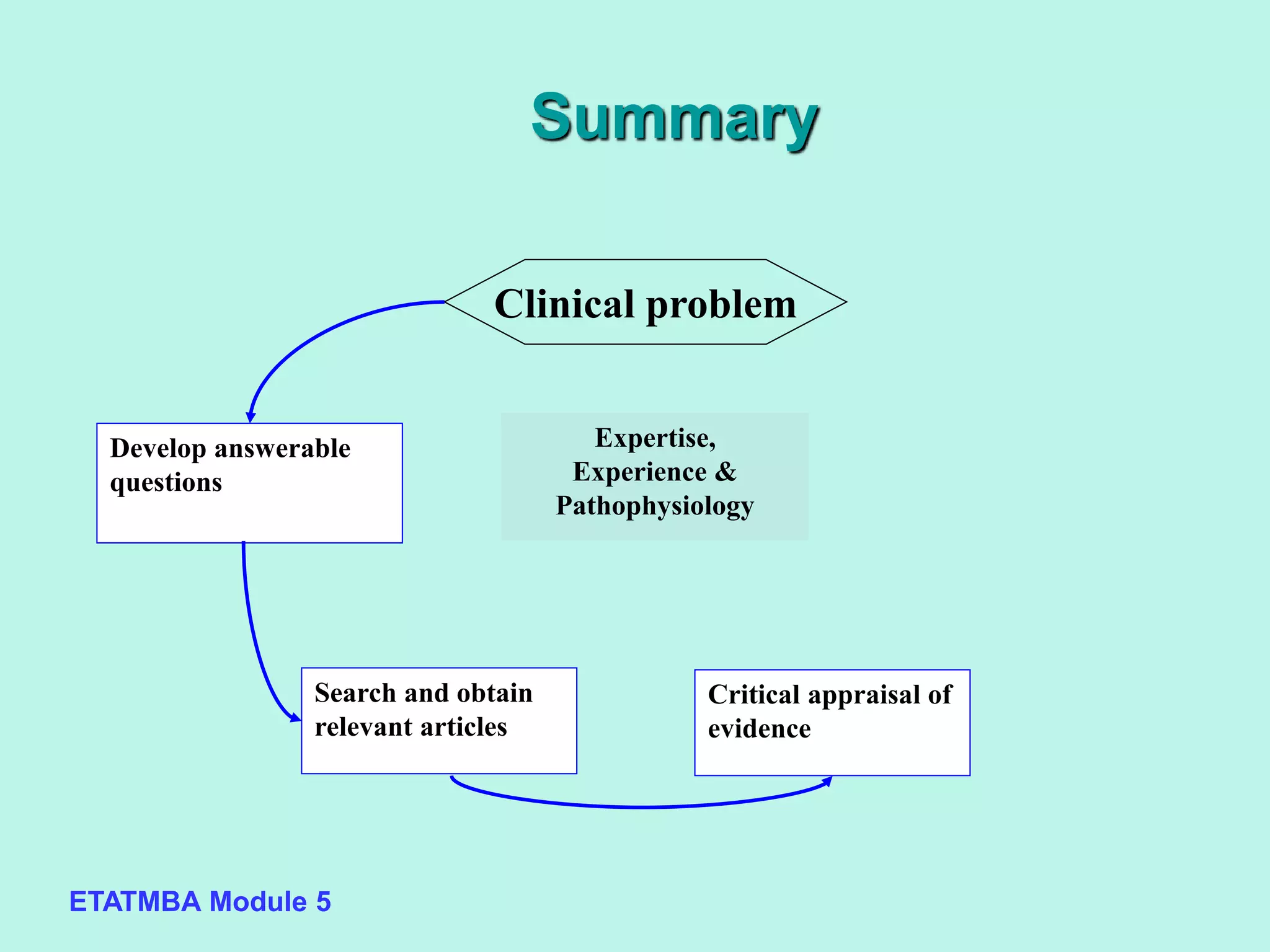
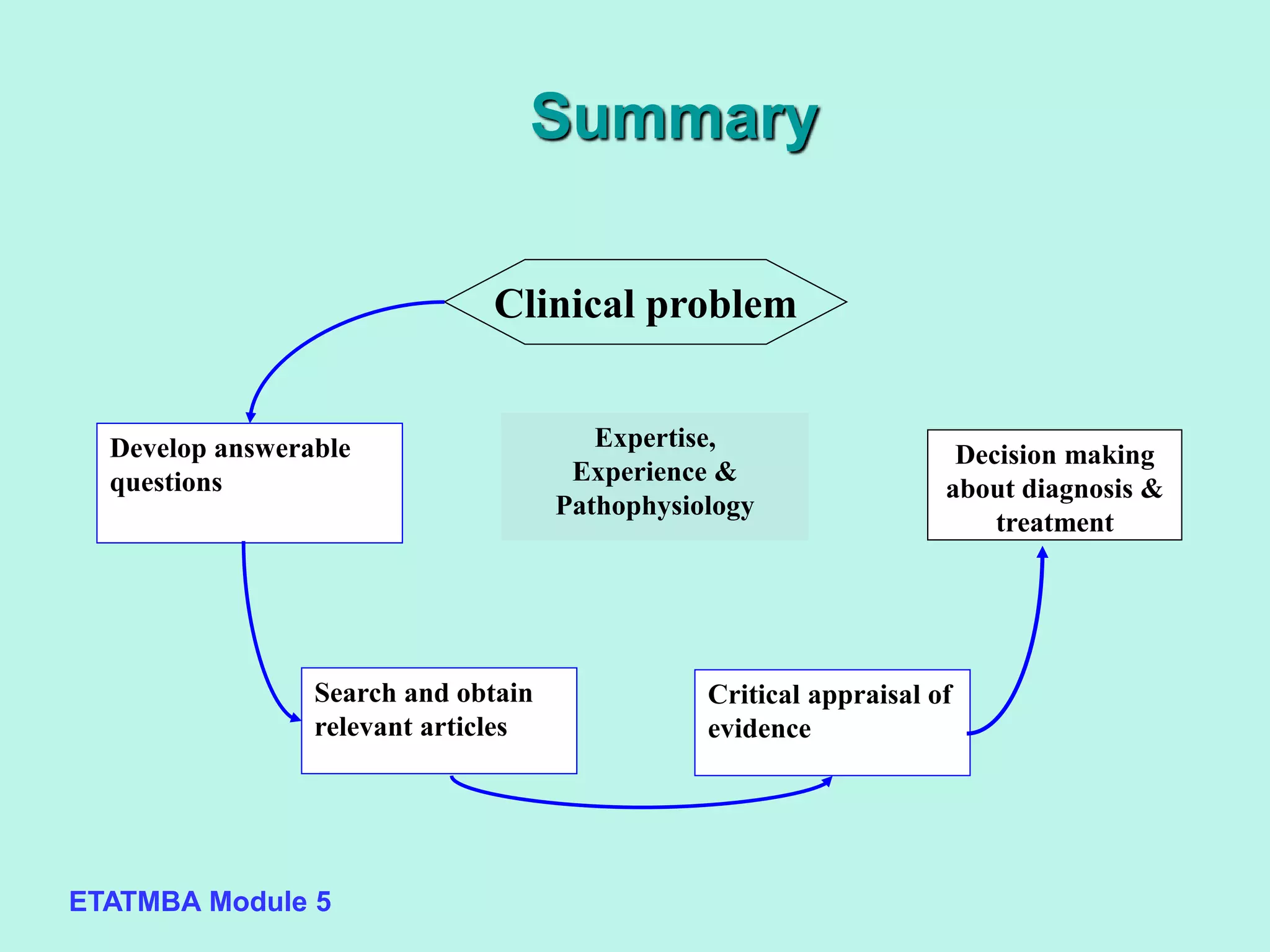
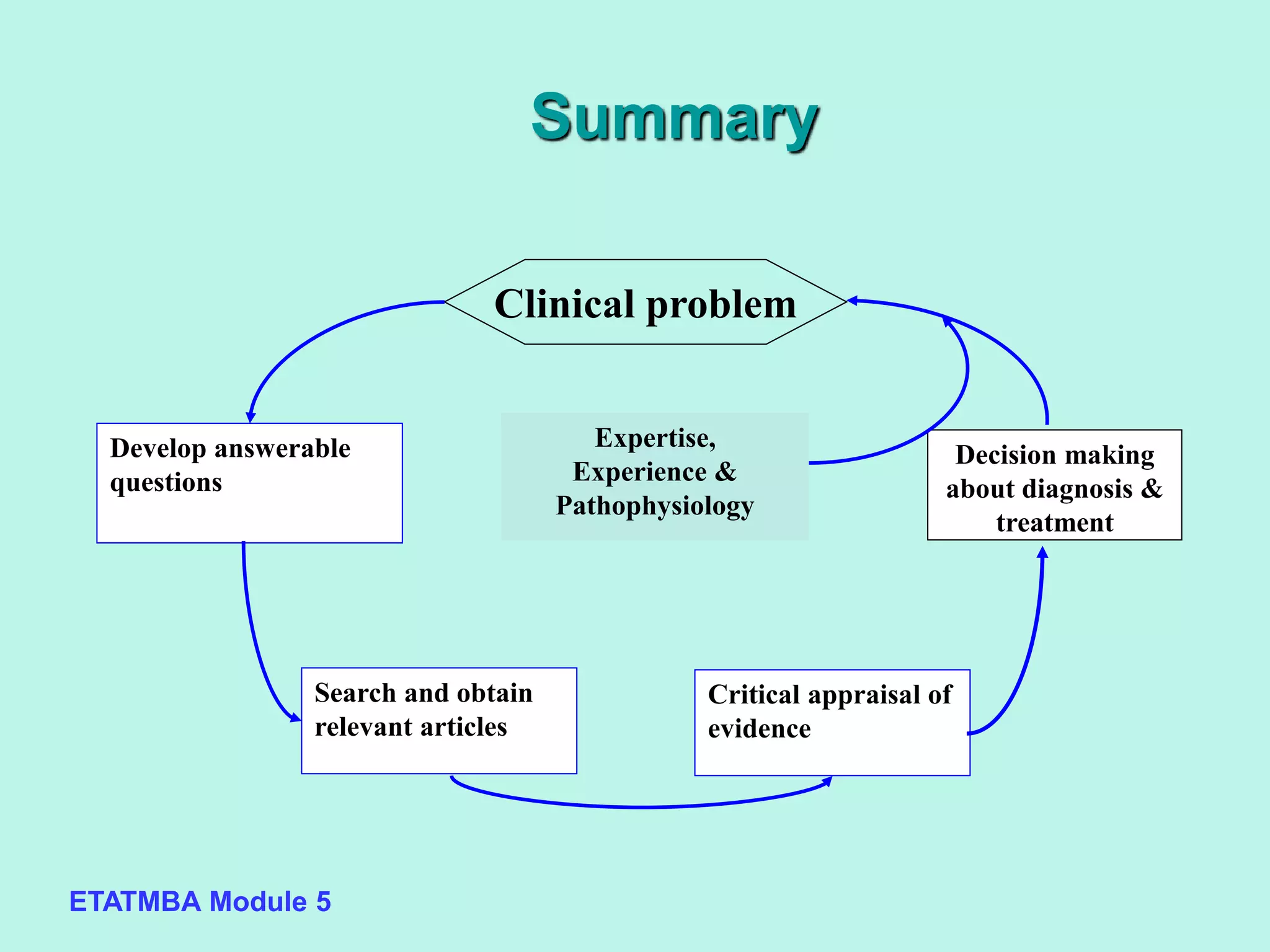
This document discusses evidence-based medicine (EBM) and outlines the key steps in applying an EBM approach to clinical practice. It begins by contrasting the old paradigm of unsystematic clinical experience with the new EBM paradigm. The 5 key steps of EBM are then summarized as: 1) developing answerable clinical questions, 2) searching for and obtaining relevant evidence, 3) critically appraising the evidence, 4) using clinical expertise and patient preferences to apply evidence to individual patients, and 5) evaluating outcomes. The document provides guidance on formulating answerable clinical questions, identifying appropriate study designs, critically appraising evidence, and integrating evidence with expertise and patient values in clinical decision making.