Chemical bond 2.ppt
•Download as PPT, PDF•
0 likes•26 views
This document discusses different types of intermolecular forces: 1) Dipole-dipole forces occur between polar molecules due to the attraction between opposite charges. 2) Ion-dipole forces occur between ions and polar molecules. 3) Dispersion forces, the weakest intermolecular forces, arise from temporary dipoles induced in nonpolar molecules. 4) Hydrogen bonding, a stronger intermolecular force, occurs between a hydrogen covalently bonded to F, O or N and another electronegative atom.
Report
Share
Report
Share
Recommended
Ionic Bonding

Ionic bonding occurs when there is a difference in electronegativity between two atoms. The atom with higher electronegativity attracts the electrons in the bond, becoming negatively charged (δ-), while the other atom loses electrons, becoming positively charged (δ+). This ionic attraction forms ionic compounds of cations and anions. Intermolecular forces include dipole-dipole interactions between polar molecules, ion-dipole interactions between ions and polar molecules, and hydrogen bonding between molecules containing hydrogen bonded to fluorine, oxygen, or nitrogen.
Chapter 6 Notes

Chemical bonds form between atoms to achieve more stable arrangements with lower potential energy. The main types of bonds are ionic, covalent, and metallic. Ionic bonds form between oppositely charged ions, covalent bonds form when atoms share electrons, and metallic bonds result from delocalized electrons being shared among many atoms in a metal. The degree of ionic or covalent character in a bond depends on the electronegativity difference between the atoms. Molecular compounds are made of molecules held together by covalent bonds, while ionic compounds are made of ionic bonds between cations and anions.
Chapter 6 

Chemical bonds form between atoms to achieve more stable arrangements with lower potential energy. The type of bonding depends on differences in electronegativity between atoms. Ionic bonds form between ions, covalent bonds involve shared electron pairs, and metallic bonds result from delocalized electrons shared among many atoms in a lattice. Molecular geometry and intermolecular forces also influence molecular properties.
4-4 Ionic & Covalent Bonding

Ionic bonds form when electrons are transferred between metals and nonmetals, resulting in oppositely charged ions. Covalent bonds form when nonmetals share electrons in order to achieve a stable electron configuration. Ionic compounds have high melting points and conduct electricity well when melted but not solid, while covalent compounds have low melting points and generally do not conduct electricity or dissolve in water well.
Chapter 8, section 8.4 (1)

This document discusses bond polarity and molecular polarity. It explains that polar covalent bonds form when electrons are shared unequally between atoms of different electronegativity. The more electronegative atom attracts the electrons more strongly, acquiring a partial negative charge, while the other atom acquires a partial positive charge. Whether a bond is nonpolar, moderately polar, or ionic depends on the electronegativity difference between the atoms. Polar bonds can result in polar molecules if the bond polarities do not cancel out. Intermolecular forces like dipole interactions and dispersion forces are responsible for molecular phase changes and arise from attractions between slightly positively and negatively charged regions on different molecules. Hydrogen bonds are especially strong dipole interactions that
Chapter 8 Powerpoint.pptx

This document provides information about molecular and ionic compounds, including:
- Molecular compounds are formed by covalent bonds between nonmetal atoms, while ionic compounds involve metal and nonmetal atoms bonded by ionic bonds.
- Molecular formulas show the actual number and type of atoms in a molecule, while ionic formulas use the lowest whole number ratio.
- Covalent bonds are represented by electron dot structures that show how atoms share electrons to achieve stable configurations. Multiple and coordinate covalent bonds are also discussed.
- Polarity arises in polar covalent bonds due to unequal electron sharing. Polar molecules have dipole moments while intermolecular forces include hydrogen bonding, dipole-dipole interactions, and
Polarity and Intermolecular Forces.ppt

This document discusses polarity and intermolecular forces. It defines electronegativity and explains how differences in electronegativity between atoms result in polar covalent bonds. Dipole moments are used to represent bond polarity. Intermolecular forces, including dipole-dipole interactions, hydrogen bonding, and London dispersion forces are described. These intermolecular forces influence important properties like melting and boiling points.
ECA. Polarity and Intermolecular Forces.ppt

This document discusses polarity and intermolecular forces. It defines electronegativity and explains how differences in electronegativity between atoms result in polar covalent bonds. Dipole moments are used to represent bond polarity. Intermolecular forces, including dipole-dipole interactions, hydrogen bonding, and London dispersion forces are described. These intermolecular forces influence important properties like melting and boiling points.
Recommended
Ionic Bonding

Ionic bonding occurs when there is a difference in electronegativity between two atoms. The atom with higher electronegativity attracts the electrons in the bond, becoming negatively charged (δ-), while the other atom loses electrons, becoming positively charged (δ+). This ionic attraction forms ionic compounds of cations and anions. Intermolecular forces include dipole-dipole interactions between polar molecules, ion-dipole interactions between ions and polar molecules, and hydrogen bonding between molecules containing hydrogen bonded to fluorine, oxygen, or nitrogen.
Chapter 6 Notes

Chemical bonds form between atoms to achieve more stable arrangements with lower potential energy. The main types of bonds are ionic, covalent, and metallic. Ionic bonds form between oppositely charged ions, covalent bonds form when atoms share electrons, and metallic bonds result from delocalized electrons being shared among many atoms in a metal. The degree of ionic or covalent character in a bond depends on the electronegativity difference between the atoms. Molecular compounds are made of molecules held together by covalent bonds, while ionic compounds are made of ionic bonds between cations and anions.
Chapter 6 

Chemical bonds form between atoms to achieve more stable arrangements with lower potential energy. The type of bonding depends on differences in electronegativity between atoms. Ionic bonds form between ions, covalent bonds involve shared electron pairs, and metallic bonds result from delocalized electrons shared among many atoms in a lattice. Molecular geometry and intermolecular forces also influence molecular properties.
4-4 Ionic & Covalent Bonding

Ionic bonds form when electrons are transferred between metals and nonmetals, resulting in oppositely charged ions. Covalent bonds form when nonmetals share electrons in order to achieve a stable electron configuration. Ionic compounds have high melting points and conduct electricity well when melted but not solid, while covalent compounds have low melting points and generally do not conduct electricity or dissolve in water well.
Chapter 8, section 8.4 (1)

This document discusses bond polarity and molecular polarity. It explains that polar covalent bonds form when electrons are shared unequally between atoms of different electronegativity. The more electronegative atom attracts the electrons more strongly, acquiring a partial negative charge, while the other atom acquires a partial positive charge. Whether a bond is nonpolar, moderately polar, or ionic depends on the electronegativity difference between the atoms. Polar bonds can result in polar molecules if the bond polarities do not cancel out. Intermolecular forces like dipole interactions and dispersion forces are responsible for molecular phase changes and arise from attractions between slightly positively and negatively charged regions on different molecules. Hydrogen bonds are especially strong dipole interactions that
Chapter 8 Powerpoint.pptx

This document provides information about molecular and ionic compounds, including:
- Molecular compounds are formed by covalent bonds between nonmetal atoms, while ionic compounds involve metal and nonmetal atoms bonded by ionic bonds.
- Molecular formulas show the actual number and type of atoms in a molecule, while ionic formulas use the lowest whole number ratio.
- Covalent bonds are represented by electron dot structures that show how atoms share electrons to achieve stable configurations. Multiple and coordinate covalent bonds are also discussed.
- Polarity arises in polar covalent bonds due to unequal electron sharing. Polar molecules have dipole moments while intermolecular forces include hydrogen bonding, dipole-dipole interactions, and
Polarity and Intermolecular Forces.ppt

This document discusses polarity and intermolecular forces. It defines electronegativity and explains how differences in electronegativity between atoms result in polar covalent bonds. Dipole moments are used to represent bond polarity. Intermolecular forces, including dipole-dipole interactions, hydrogen bonding, and London dispersion forces are described. These intermolecular forces influence important properties like melting and boiling points.
ECA. Polarity and Intermolecular Forces.ppt

This document discusses polarity and intermolecular forces. It defines electronegativity and explains how differences in electronegativity between atoms result in polar covalent bonds. Dipole moments are used to represent bond polarity. Intermolecular forces, including dipole-dipole interactions, hydrogen bonding, and London dispersion forces are described. These intermolecular forces influence important properties like melting and boiling points.
Polarity and Intermolecular Forces...ppt

This document discusses polarity and intermolecular forces. It defines electronegativity and explains how differences in electronegativity between atoms result in polar covalent bonds. Dipole moments are used to represent bond polarity. Intermolecular forces, including dipole-dipole interactions, hydrogen bonding, and London dispersion forces are described. These intermolecular forces influence important properties like melting and boiling points.
Chemical bonding theories, 10(1) 

Theories about how chemical bonds are formed. Covalent bond, multi covalent bonds. How to write Lewis Structures
bonding_regents_chem.ppt

This document defines chemical bonding and describes the three main types of bonds: metallic, ionic, and covalent. Metallic bonds form a crystalline lattice structure with freely moving electrons. Ionic bonds form when ions with opposite charges are attracted to each other via electrostatic forces. Covalent bonds form when atoms share electrons to achieve stable electron configurations. The type of bonding determines various physical properties like melting point, hardness, and conductivity.
4-2 Ionic & Covalent Bonding

The document summarizes the key differences between ionic and covalent bonding. Ionic bonds form when a metal transfers electrons to a nonmetal, creating oppositely charged ions. Covalent bonds form when nonmetals share electrons to obtain a full outer shell. Ionic compounds have high melting points, are brittle solids, and dissolve well in water, while covalent compounds have lower melting points, are soft and pliable, and are generally insoluble in water.
Lecture 8.4c- Intermolecular Forces

1) Ammonia (NH3) is a polar molecule that has tetrahedral shape due to hydrogen bonding between nitrogen and hydrogen atoms.
2) Intermolecular forces include dipole-dipole interactions between polar molecules and London dispersion forces between all molecules.
3) Hydrogen bonding is a strong type of dipole-dipole interaction that occurs between molecules containing hydrogen bonded to nitrogen, oxygen, or fluorine.
Chapter 8 covalent bonding

Covalent bonds form when two nonmetal atoms share one or more pairs of valence electrons to achieve a noble gas configuration. This sharing of electrons results in the formation of molecules with lower potential energy than the individual atoms. Covalent bonds can be either polar or nonpolar depending on whether the electrons are shared equally or unequally between the bonded atoms. Multiple bonds are also possible where two or more pairs of electrons are shared, such as double and triple bonds.
Chem 2 - Intermolecular Forces & Phases of Matter II 

1) There are several types of intermolecular forces ranging from strongest to weakest - hydrogen bonding, dipole-dipole interactions, dipole-induced dipole interactions, and dispersion forces.
2) Hydrogen bonding is the strongest and involves the electrostatic attraction between a hydrogen atom covalently bonded to a highly electronegative atom like N, O, or F, and another electronegative atom's lone pair of electrons.
3) Dipole-dipole interactions occur between polar molecules that have partially positive and negative ends that electrostatically attract each other, while dipole-induced dipole interactions involve a polar molecule inducing a dipole in a neighboring nonpolar molecule.
Subject: Chemical Bonding in physics....

This document discusses different types of chemical bonds:
- Ionic bonds form between metals and nonmetals through the transfer of electrons to create positively and negatively charged ions. Covalent bonds form between nonmetals through the sharing of electron pairs to create molecular compounds. Metallic bonds involve the pooling of electrons between metal atoms. Polar bonds occur when electrons are shared unevenly between atoms. Understanding chemical bonds is important because they join atoms in the materials used in everyday life.
Chemical%20Bonds.pptx

This document provides a summary of a lecture on inorganic chemistry. It discusses chemical bonds, including the definition of a chemical bond as a force that holds atoms together in a stable molecule. It describes four primary types of chemical bonds: ionic, covalent, coordinate covalent, and metallic. Covalent bonds are formed by the sharing of electron pairs between atoms. Coordinate covalent or dative bonds occur when both electrons in the shared pair come from one atom. Examples of compounds containing different bond types are also provided.
Ionic bond,covalent bond and hydrogen bond

Ionic bond,covalent bond and hydrogen bond . we can see from this slide the definition and mechanism of those chemical bonding eaily
Lesson 2 Intermolecular Forces.pptx

This document defines and differentiates the main types of intermolecular forces: dipole-dipole forces, London dispersion forces, ion-dipole forces, and hydrogen bonding. It provides examples of polar and nonpolar molecules, and describes the characteristics of each type of intermolecular force, including their relative strengths and the molecules between which they typically occur.
Key SI #6

Intermolecular forces exist between molecules, while intramolecular forces exist within molecules. Melting points increase as intermolecular forces strengthen. Polar molecules have partial charges and can form ion-dipole attractions. The main intermolecular forces are London dispersion, dipole-dipole, and hydrogen bonding. Ionic and covalent bonds are stronger than any intermolecular force within a molecule. A flow chart determines the dominant intermolecular force based on whether ions or polar molecules are present.
Chemical bonds

This document discusses three types of chemical bonds: metallic bonds, ionic bonds, and covalent bonds. It then describes different types of covalent bonds including nonpolar, polar, and coordinated covalent bonds. Finally, it discusses three types of intermolecular forces: dispersion forces, dipole-dipole forces, and hydrogen bonding. Dispersion forces exist between all molecules due to temporary dipoles, while dipole-dipole forces exist between polar molecules due to attraction of partial charges. Hydrogen bonding is a strong form of dipole-dipole force between a hydrogen atom covalently bonded to an electronegative atom like N, O, or F.
Chapter12chemicalbondsandmixtures 150222095853-conversion-gate02

This chapter discusses chemical bonds and mixtures. It introduces electron-dot structures to show valence electrons and how they are involved in bonding. Ionic bonds form when ions with opposite charges are attracted to each other. Covalent bonds form when atoms share electrons. Polar covalent bonds result when electrons are shared unevenly. Molecular polarity arises if the polar bonds in a molecule do not cancel out. Most materials are mixtures that can be separated into pure substances. Solutions are homogeneous mixtures where a solute dissolves evenly throughout a solvent. Concentration, molarity, and solubility are measures used to describe solutions.
Chapter 12 chemical bonds and mixtures

This chapter discusses chemical bonds and mixtures. It introduces electron-dot structures to show valence electrons and how they are involved in bonding. Ionic bonds form when ions with opposite charges are attracted to each other. Covalent bonds form when atoms share electrons. Polar covalent bonds result when electrons are shared unevenly. Molecular polarity arises if the polar bonds in a molecule do not cancel out. Most materials are mixtures that can be separated into pure substances. Solutions are homogeneous mixtures where one substance dissolves evenly throughout another. Concentration, molarity, and solubility are measures used to describe solutions.
Chapter12chemicalbondsandmixtures 150222095853-conversion-gate02

This chapter discusses chemical bonds and mixtures. It introduces electron-dot structures to show valence electrons and how they are involved in bonding. Ionic bonds form when ions with opposite charges are attracted to each other. Covalent bonds form when atoms share electrons. Polar covalent bonds result when electrons are shared unevenly. Molecular polarity arises if the polar bonds in a molecule do not cancel out. Most materials are mixtures that can be separated into pure substances. Solutions are homogeneous mixtures where one substance dissolves evenly throughout another. Concentration, molarity, and solubility are discussed in relation to solutions.
Chapter6 chemicalbonding-100707021031-phpapp01

1) Chemical bonds form between atoms as a way for atoms to decrease their potential energy and become more stable.
2) There are three main types of chemical bonds: ionic bonds, which form between ions and are electrostatic in nature; covalent bonds, which form through the sharing of electron pairs between atoms; and metallic bonds, which form between positive metal ions and delocalized electrons in metals.
3) Ionic compounds consist of positive and negative ions that are attracted to one another via ionic bonding. The ions arrange themselves in repeating three-dimensional patterns called crystal lattices to minimize potential energy.
Chemical Bond MANIK

The chemical Bond: Electronic concept of valency. Different types of chemical bond e.g. ionic, covalent, coordinate covalent metallic, dipole, hydrogen bond etc. Theories of covalent bonding and hybridization.
Chemical bonding 

There are four main types of chemical bonds: ionic bonds, covalent bonds, metallic bonds, and hydrogen bonds. Ionic bonds form through the transfer of electrons from one atom to another, creating oppositely charged ions. Covalent bonds form through the sharing of electron pairs between atoms. Metallic bonds arise from the electrostatic attraction between positively charged metal ions and delocalized electrons. Hydrogen bonds result from the attraction between hydrogen atoms covalently bonded to electronegative atoms like nitrogen or oxygen, and another electronegative atom. Chemical bonds form through these interactions to hold groups of atoms together in molecules, ions, crystals and other stable species.
Bonds

A bond is formed when two or more atoms join together through sharing or exchanging electrons to achieve stability. A molecule is two or more bonded atoms, and a substance can be either a pure element or compound made of two or more elements. There are two main types of bonds: covalent bonds formed by sharing electrons between nonmetals and ionic bonds formed by a transfer of electrons between a metal and nonmetal. The periodic table arranges elements and can provide information about their valence electrons and reactivity through bonding properties.
BÀI TẬP BỔ TRỢ TIẾNG ANH LỚP 9 CẢ NĂM - GLOBAL SUCCESS - NĂM HỌC 2024-2025 - ...

BÀI TẬP BỔ TRỢ TIẾNG ANH LỚP 9 CẢ NĂM - GLOBAL SUCCESS - NĂM HỌC 2024-2025 - ...Nguyen Thanh Tu Collection
https://app.box.com/s/tacvl9ekroe9hqupdnjruiypvm9rdaneMore Related Content
Similar to Chemical bond 2.ppt
Polarity and Intermolecular Forces...ppt

This document discusses polarity and intermolecular forces. It defines electronegativity and explains how differences in electronegativity between atoms result in polar covalent bonds. Dipole moments are used to represent bond polarity. Intermolecular forces, including dipole-dipole interactions, hydrogen bonding, and London dispersion forces are described. These intermolecular forces influence important properties like melting and boiling points.
Chemical bonding theories, 10(1) 

Theories about how chemical bonds are formed. Covalent bond, multi covalent bonds. How to write Lewis Structures
bonding_regents_chem.ppt

This document defines chemical bonding and describes the three main types of bonds: metallic, ionic, and covalent. Metallic bonds form a crystalline lattice structure with freely moving electrons. Ionic bonds form when ions with opposite charges are attracted to each other via electrostatic forces. Covalent bonds form when atoms share electrons to achieve stable electron configurations. The type of bonding determines various physical properties like melting point, hardness, and conductivity.
4-2 Ionic & Covalent Bonding

The document summarizes the key differences between ionic and covalent bonding. Ionic bonds form when a metal transfers electrons to a nonmetal, creating oppositely charged ions. Covalent bonds form when nonmetals share electrons to obtain a full outer shell. Ionic compounds have high melting points, are brittle solids, and dissolve well in water, while covalent compounds have lower melting points, are soft and pliable, and are generally insoluble in water.
Lecture 8.4c- Intermolecular Forces

1) Ammonia (NH3) is a polar molecule that has tetrahedral shape due to hydrogen bonding between nitrogen and hydrogen atoms.
2) Intermolecular forces include dipole-dipole interactions between polar molecules and London dispersion forces between all molecules.
3) Hydrogen bonding is a strong type of dipole-dipole interaction that occurs between molecules containing hydrogen bonded to nitrogen, oxygen, or fluorine.
Chapter 8 covalent bonding

Covalent bonds form when two nonmetal atoms share one or more pairs of valence electrons to achieve a noble gas configuration. This sharing of electrons results in the formation of molecules with lower potential energy than the individual atoms. Covalent bonds can be either polar or nonpolar depending on whether the electrons are shared equally or unequally between the bonded atoms. Multiple bonds are also possible where two or more pairs of electrons are shared, such as double and triple bonds.
Chem 2 - Intermolecular Forces & Phases of Matter II 

1) There are several types of intermolecular forces ranging from strongest to weakest - hydrogen bonding, dipole-dipole interactions, dipole-induced dipole interactions, and dispersion forces.
2) Hydrogen bonding is the strongest and involves the electrostatic attraction between a hydrogen atom covalently bonded to a highly electronegative atom like N, O, or F, and another electronegative atom's lone pair of electrons.
3) Dipole-dipole interactions occur between polar molecules that have partially positive and negative ends that electrostatically attract each other, while dipole-induced dipole interactions involve a polar molecule inducing a dipole in a neighboring nonpolar molecule.
Subject: Chemical Bonding in physics....

This document discusses different types of chemical bonds:
- Ionic bonds form between metals and nonmetals through the transfer of electrons to create positively and negatively charged ions. Covalent bonds form between nonmetals through the sharing of electron pairs to create molecular compounds. Metallic bonds involve the pooling of electrons between metal atoms. Polar bonds occur when electrons are shared unevenly between atoms. Understanding chemical bonds is important because they join atoms in the materials used in everyday life.
Chemical%20Bonds.pptx

This document provides a summary of a lecture on inorganic chemistry. It discusses chemical bonds, including the definition of a chemical bond as a force that holds atoms together in a stable molecule. It describes four primary types of chemical bonds: ionic, covalent, coordinate covalent, and metallic. Covalent bonds are formed by the sharing of electron pairs between atoms. Coordinate covalent or dative bonds occur when both electrons in the shared pair come from one atom. Examples of compounds containing different bond types are also provided.
Ionic bond,covalent bond and hydrogen bond

Ionic bond,covalent bond and hydrogen bond . we can see from this slide the definition and mechanism of those chemical bonding eaily
Lesson 2 Intermolecular Forces.pptx

This document defines and differentiates the main types of intermolecular forces: dipole-dipole forces, London dispersion forces, ion-dipole forces, and hydrogen bonding. It provides examples of polar and nonpolar molecules, and describes the characteristics of each type of intermolecular force, including their relative strengths and the molecules between which they typically occur.
Key SI #6

Intermolecular forces exist between molecules, while intramolecular forces exist within molecules. Melting points increase as intermolecular forces strengthen. Polar molecules have partial charges and can form ion-dipole attractions. The main intermolecular forces are London dispersion, dipole-dipole, and hydrogen bonding. Ionic and covalent bonds are stronger than any intermolecular force within a molecule. A flow chart determines the dominant intermolecular force based on whether ions or polar molecules are present.
Chemical bonds

This document discusses three types of chemical bonds: metallic bonds, ionic bonds, and covalent bonds. It then describes different types of covalent bonds including nonpolar, polar, and coordinated covalent bonds. Finally, it discusses three types of intermolecular forces: dispersion forces, dipole-dipole forces, and hydrogen bonding. Dispersion forces exist between all molecules due to temporary dipoles, while dipole-dipole forces exist between polar molecules due to attraction of partial charges. Hydrogen bonding is a strong form of dipole-dipole force between a hydrogen atom covalently bonded to an electronegative atom like N, O, or F.
Chapter12chemicalbondsandmixtures 150222095853-conversion-gate02

This chapter discusses chemical bonds and mixtures. It introduces electron-dot structures to show valence electrons and how they are involved in bonding. Ionic bonds form when ions with opposite charges are attracted to each other. Covalent bonds form when atoms share electrons. Polar covalent bonds result when electrons are shared unevenly. Molecular polarity arises if the polar bonds in a molecule do not cancel out. Most materials are mixtures that can be separated into pure substances. Solutions are homogeneous mixtures where a solute dissolves evenly throughout a solvent. Concentration, molarity, and solubility are measures used to describe solutions.
Chapter 12 chemical bonds and mixtures

This chapter discusses chemical bonds and mixtures. It introduces electron-dot structures to show valence electrons and how they are involved in bonding. Ionic bonds form when ions with opposite charges are attracted to each other. Covalent bonds form when atoms share electrons. Polar covalent bonds result when electrons are shared unevenly. Molecular polarity arises if the polar bonds in a molecule do not cancel out. Most materials are mixtures that can be separated into pure substances. Solutions are homogeneous mixtures where one substance dissolves evenly throughout another. Concentration, molarity, and solubility are measures used to describe solutions.
Chapter12chemicalbondsandmixtures 150222095853-conversion-gate02

This chapter discusses chemical bonds and mixtures. It introduces electron-dot structures to show valence electrons and how they are involved in bonding. Ionic bonds form when ions with opposite charges are attracted to each other. Covalent bonds form when atoms share electrons. Polar covalent bonds result when electrons are shared unevenly. Molecular polarity arises if the polar bonds in a molecule do not cancel out. Most materials are mixtures that can be separated into pure substances. Solutions are homogeneous mixtures where one substance dissolves evenly throughout another. Concentration, molarity, and solubility are discussed in relation to solutions.
Chapter6 chemicalbonding-100707021031-phpapp01

1) Chemical bonds form between atoms as a way for atoms to decrease their potential energy and become more stable.
2) There are three main types of chemical bonds: ionic bonds, which form between ions and are electrostatic in nature; covalent bonds, which form through the sharing of electron pairs between atoms; and metallic bonds, which form between positive metal ions and delocalized electrons in metals.
3) Ionic compounds consist of positive and negative ions that are attracted to one another via ionic bonding. The ions arrange themselves in repeating three-dimensional patterns called crystal lattices to minimize potential energy.
Chemical Bond MANIK

The chemical Bond: Electronic concept of valency. Different types of chemical bond e.g. ionic, covalent, coordinate covalent metallic, dipole, hydrogen bond etc. Theories of covalent bonding and hybridization.
Chemical bonding 

There are four main types of chemical bonds: ionic bonds, covalent bonds, metallic bonds, and hydrogen bonds. Ionic bonds form through the transfer of electrons from one atom to another, creating oppositely charged ions. Covalent bonds form through the sharing of electron pairs between atoms. Metallic bonds arise from the electrostatic attraction between positively charged metal ions and delocalized electrons. Hydrogen bonds result from the attraction between hydrogen atoms covalently bonded to electronegative atoms like nitrogen or oxygen, and another electronegative atom. Chemical bonds form through these interactions to hold groups of atoms together in molecules, ions, crystals and other stable species.
Bonds

A bond is formed when two or more atoms join together through sharing or exchanging electrons to achieve stability. A molecule is two or more bonded atoms, and a substance can be either a pure element or compound made of two or more elements. There are two main types of bonds: covalent bonds formed by sharing electrons between nonmetals and ionic bonds formed by a transfer of electrons between a metal and nonmetal. The periodic table arranges elements and can provide information about their valence electrons and reactivity through bonding properties.
Similar to Chemical bond 2.ppt (20)
Chem 2 - Intermolecular Forces & Phases of Matter II 

Chem 2 - Intermolecular Forces & Phases of Matter II
Chapter12chemicalbondsandmixtures 150222095853-conversion-gate02

Chapter12chemicalbondsandmixtures 150222095853-conversion-gate02
Chapter12chemicalbondsandmixtures 150222095853-conversion-gate02

Chapter12chemicalbondsandmixtures 150222095853-conversion-gate02
Recently uploaded
BÀI TẬP BỔ TRỢ TIẾNG ANH LỚP 9 CẢ NĂM - GLOBAL SUCCESS - NĂM HỌC 2024-2025 - ...

BÀI TẬP BỔ TRỢ TIẾNG ANH LỚP 9 CẢ NĂM - GLOBAL SUCCESS - NĂM HỌC 2024-2025 - ...Nguyen Thanh Tu Collection
https://app.box.com/s/tacvl9ekroe9hqupdnjruiypvm9rdaneHow to Make a Field Mandatory in Odoo 17

In Odoo, making a field required can be done through both Python code and XML views. When you set the required attribute to True in Python code, it makes the field required across all views where it's used. Conversely, when you set the required attribute in XML views, it makes the field required only in the context of that particular view.
How to Manage Your Lost Opportunities in Odoo 17 CRM

Odoo 17 CRM allows us to track why we lose sales opportunities with "Lost Reasons." This helps analyze our sales process and identify areas for improvement. Here's how to configure lost reasons in Odoo 17 CRM
PCOS corelations and management through Ayurveda.

This presentation includes basic of PCOS their pathology and treatment and also Ayurveda correlation of PCOS and Ayurvedic line of treatment mentioned in classics.
The Diamonds of 2023-2024 in the IGRA collection

A review of the growth of the Israel Genealogy Research Association Database Collection for the last 12 months. Our collection is now passed the 3 million mark and still growing. See which archives have contributed the most. See the different types of records we have, and which years have had records added. You can also see what we have for the future.
Pollock and Snow "DEIA in the Scholarly Landscape, Session One: Setting Expec...

Pollock and Snow "DEIA in the Scholarly Landscape, Session One: Setting Expec...National Information Standards Organization (NISO)
This presentation was provided by Steph Pollock of The American Psychological Association’s Journals Program, and Damita Snow, of The American Society of Civil Engineers (ASCE), for the initial session of NISO's 2024 Training Series "DEIA in the Scholarly Landscape." Session One: 'Setting Expectations: a DEIA Primer,' was held June 6, 2024.Leveraging Generative AI to Drive Nonprofit Innovation

In this webinar, participants learned how to utilize Generative AI to streamline operations and elevate member engagement. Amazon Web Service experts provided a customer specific use cases and dived into low/no-code tools that are quick and easy to deploy through Amazon Web Service (AWS.)
Hindi varnamala | hindi alphabet PPT.pdf

हिंदी वर्णमाला पीपीटी, hindi alphabet PPT presentation, hindi varnamala PPT, Hindi Varnamala pdf, हिंदी स्वर, हिंदी व्यंजन, sikhiye hindi varnmala, dr. mulla adam ali, hindi language and literature, hindi alphabet with drawing, hindi alphabet pdf, hindi varnamala for childrens, hindi language, hindi varnamala practice for kids, https://www.drmullaadamali.com
Your Skill Boost Masterclass: Strategies for Effective Upskilling

Your Skill Boost Masterclass: Strategies for Effective UpskillingExcellence Foundation for South Sudan
Strategies for Effective Upskilling is a presentation by Chinwendu Peace in a Your Skill Boost Masterclass organisation by the Excellence Foundation for South Sudan on 08th and 09th June 2024 from 1 PM to 3 PM on each day.Pengantar Penggunaan Flutter - Dart programming language1.pptx

Pengantar Penggunaan Flutter - Dart programming language1.pptx
How to Setup Warehouse & Location in Odoo 17 Inventory

In this slide, we'll explore how to set up warehouses and locations in Odoo 17 Inventory. This will help us manage our stock effectively, track inventory levels, and streamline warehouse operations.
Traditional Musical Instruments of Arunachal Pradesh and Uttar Pradesh - RAYH...

Traditional Musical Instruments of Arunachal Pradesh and Uttar Pradesh
South African Journal of Science: Writing with integrity workshop (2024)

South African Journal of Science: Writing with integrity workshop (2024)Academy of Science of South Africa
A workshop hosted by the South African Journal of Science aimed at postgraduate students and early career researchers with little or no experience in writing and publishing journal articles.LAND USE LAND COVER AND NDVI OF MIRZAPUR DISTRICT, UP

This Dissertation explores the particular circumstances of Mirzapur, a region located in the
core of India. Mirzapur, with its varied terrains and abundant biodiversity, offers an optimal
environment for investigating the changes in vegetation cover dynamics. Our study utilizes
advanced technologies such as GIS (Geographic Information Systems) and Remote sensing to
analyze the transformations that have taken place over the course of a decade.
The complex relationship between human activities and the environment has been the focus
of extensive research and worry. As the global community grapples with swift urbanization,
population expansion, and economic progress, the effects on natural ecosystems are becoming
more evident. A crucial element of this impact is the alteration of vegetation cover, which plays a
significant role in maintaining the ecological equilibrium of our planet.Land serves as the foundation for all human activities and provides the necessary materials for
these activities. As the most crucial natural resource, its utilization by humans results in different
'Land uses,' which are determined by both human activities and the physical characteristics of the
land.
The utilization of land is impacted by human needs and environmental factors. In countries
like India, rapid population growth and the emphasis on extensive resource exploitation can lead
to significant land degradation, adversely affecting the region's land cover.
Therefore, human intervention has significantly influenced land use patterns over many
centuries, evolving its structure over time and space. In the present era, these changes have
accelerated due to factors such as agriculture and urbanization. Information regarding land use and
cover is essential for various planning and management tasks related to the Earth's surface,
providing crucial environmental data for scientific, resource management, policy purposes, and
diverse human activities.
Accurate understanding of land use and cover is imperative for the development planning
of any area. Consequently, a wide range of professionals, including earth system scientists, land
and water managers, and urban planners, are interested in obtaining data on land use and cover
changes, conversion trends, and other related patterns. The spatial dimensions of land use and
cover support policymakers and scientists in making well-informed decisions, as alterations in
these patterns indicate shifts in economic and social conditions. Monitoring such changes with the
help of Advanced technologies like Remote Sensing and Geographic Information Systems is
crucial for coordinated efforts across different administrative levels. Advanced technologies like
Remote Sensing and Geographic Information Systems
9
Changes in vegetation cover refer to variations in the distribution, composition, and overall
structure of plant communities across different temporal and spatial scales. These changes can
occur natural.
ANATOMY AND BIOMECHANICS OF HIP JOINT.pdf

it describes the bony anatomy including the femoral head , acetabulum, labrum . also discusses the capsule , ligaments . muscle that act on the hip joint and the range of motion are outlined. factors affecting hip joint stability and weight transmission through the joint are summarized.
Recently uploaded (20)
BÀI TẬP BỔ TRỢ TIẾNG ANH LỚP 9 CẢ NĂM - GLOBAL SUCCESS - NĂM HỌC 2024-2025 - ...

BÀI TẬP BỔ TRỢ TIẾNG ANH LỚP 9 CẢ NĂM - GLOBAL SUCCESS - NĂM HỌC 2024-2025 - ...
How to Manage Your Lost Opportunities in Odoo 17 CRM

How to Manage Your Lost Opportunities in Odoo 17 CRM
Pollock and Snow "DEIA in the Scholarly Landscape, Session One: Setting Expec...

Pollock and Snow "DEIA in the Scholarly Landscape, Session One: Setting Expec...
Leveraging Generative AI to Drive Nonprofit Innovation

Leveraging Generative AI to Drive Nonprofit Innovation
Your Skill Boost Masterclass: Strategies for Effective Upskilling

Your Skill Boost Masterclass: Strategies for Effective Upskilling
Pengantar Penggunaan Flutter - Dart programming language1.pptx

Pengantar Penggunaan Flutter - Dart programming language1.pptx
How to Setup Warehouse & Location in Odoo 17 Inventory

How to Setup Warehouse & Location in Odoo 17 Inventory
Traditional Musical Instruments of Arunachal Pradesh and Uttar Pradesh - RAYH...

Traditional Musical Instruments of Arunachal Pradesh and Uttar Pradesh - RAYH...
South African Journal of Science: Writing with integrity workshop (2024)

South African Journal of Science: Writing with integrity workshop (2024)
LAND USE LAND COVER AND NDVI OF MIRZAPUR DISTRICT, UP

LAND USE LAND COVER AND NDVI OF MIRZAPUR DISTRICT, UP
Chemical bond 2.ppt
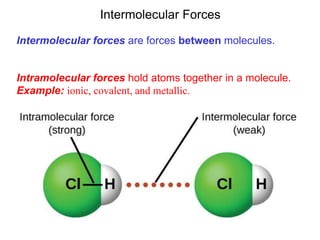
- 1. Intermolecular Forces Intermolecular forces are forces between molecules. Intramolecular forces hold atoms together in a molecule. Example: ionic, covalent, and metallic.
- 2. Electronegativity is the ability of an atom to attract toward itself the electrons in a chemical bond. Electronegativity - relative, F is highest H F electron poor region electron rich region
- 3. H F d+ d-
- 4. e- e- δ - δ + 2 + e- e- 2 + e- e- 2 + e- e- δ - δ + 2 + e- e- δ - δ + 2 + e- e- δ - δ + 2 + repel attract e- e- 2 +
- 5. Types of Intermolecular Forces Dipole-Dipole Forces Attractive forces between polar molecules Orientation of Polar Molecules in a Solid
- 6. Dipole – Dipole Forces d+ d- d+ d- d+ d- d+ d- d+ d-
- 7. Types of Intermolecular Forces Ion-Dipole Forces Attractive forces between an ion and a polar molecule Ion-Dipole Interaction
- 8. Ion – Dipole Forces Exist when we dissolve an ionic compound in a polar solvent, like H2O. The d- end of H2O is attracted to a cation (+). The d+ end of H2O is attracted to an anion (-).
- 9. Types of Intermolecular Forces Dispersion Forces – van der Walls forces/London forces (weakest) Attractive forces that arise as a result of temporary dipoles induced in atoms or molecules ion-induced dipole interaction dipole-induced dipole interaction
- 10. Hydrogen Bonding Hydrogen Bond = an attraction between a slightly positive hydrogen atom and a slightly negative atom, often oxygen or nitrogen
- 11. Hydrogen Bonding
- 12. O H H δ + δ + δ- O H H δ + δ + δ- O H H δ + δ + δ- O H H δ + δ + δ- O H H δ δ + δ- hydrogen bonds
- 13. • A hydrogen bond is an intermolecular bond. F—H O—H N—H • A hydrogen bond is formed between polar molecules that contain hydrogen covalently bonded to a small, highly electronegative atom: F, O, N.
- 14. hydrogen bond covalent bond covalent bond A dipole-dipole bond bond will be formed between the two molecules which is called a hydrogen bond.
- 15. Remember, you need: A hydrogen atom covalently bonded to an electronegative atom … N, O or F. If only one of these conditions is met, you don’t get hydrogen bonding. A lone pair of electrons on the electronegative atom.
- 16. Coordinate Covalent Bond • A coordinate bond (also called a dative covalent bond) is a covalent bond (a shared pair of electrons) in which both electrons come from the same atom. • A coordinate covalent bond is usually shown with an arrow.
- 17. Amino-Borane N has a full octet an a lone pair of electrons. NH3 is an uncharged molecule. B only has 6 electrons, it is deficient. BH3 is an uncharged molecule. N can “donate” its lone pair to B forming a co-ordinate bond.
- 18. Amino-Borane Now we have a new molecule with a covalent bond between N and B. While there are formal charges on N and B, the overall charge on the molecule is neutral. The bond formed between N and B is just as strong as any covalent bond.
- 19. ammonium ion