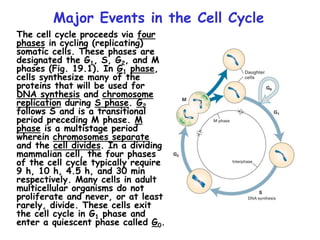
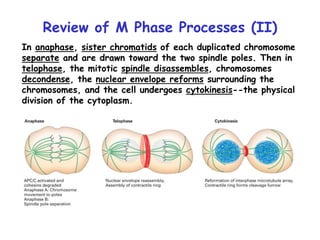
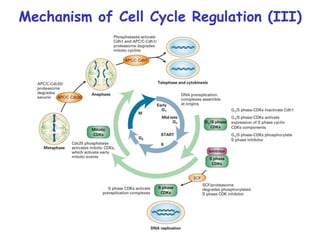
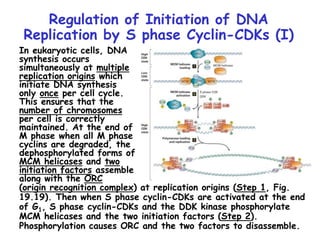
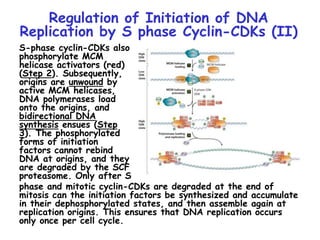
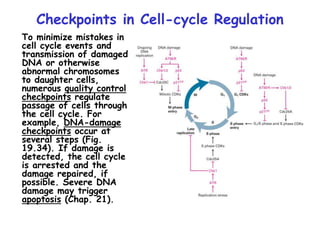
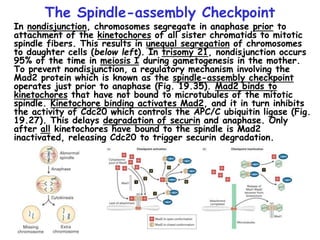
The document discusses regulation of the cell cycle through cyclins, CDKs, and ubiquitin ligases. It covers the four phases of the cell cycle (G1, S, G2, M), regulation of DNA replication and mitosis, checkpoints that ensure quality control, and mechanisms that propel quiescent cells into the cell cycle in response to mitogens. The precise ordering and timing of cell cycle events is controlled by the periodic synthesis and degradation of cyclins and other regulatory proteins.