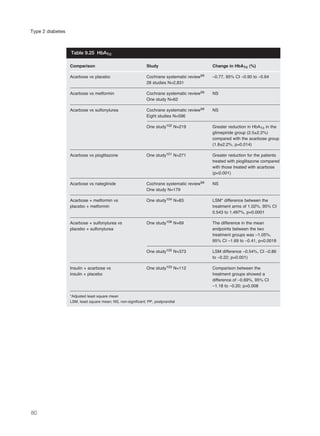
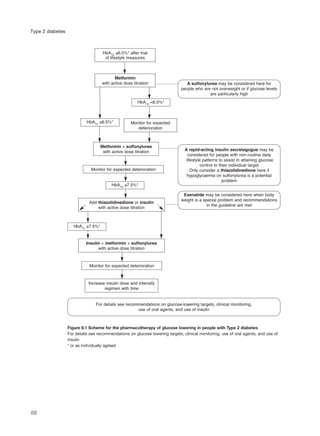
This document provides an introduction to Type 2 diabetes, including:
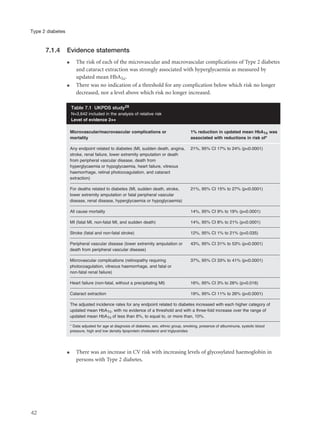
- Type 2 diabetes is caused by insulin insensitivity and a failure of pancreatic insulin secretion to compensate. It is associated with obesity and other cardiovascular risk factors.
- Without treatment, hyperglycemia can cause symptoms like polyuria and weight loss. Insulin deficiency increases risk of ketoacidosis or hyperosmolar coma.
- Type 2 diabetes significantly increases risk of cardiovascular disease like heart attacks and strokes. Management focuses on controlling multiple risk factors to reduce complications.
- Without control, hyperglycemia increases risk of eye, kidney and nerve damage. Treatment aims to lower blood glucose and control associated risks to improve health outcomes. Effective management of Type






























































































































































































![102 Feinbock C, Luger A, Klingler A et al. Effects on blood pressure of the a-glucosidase inhibitor acarbose
compared with the insulin enhancer glibenclamide in patients with hypertension and type 2 diabetes
mellitus. Diabetes, Nutrition & Metabolism - Clinical & Experimental 2003;16(4):214–221.
103 Hwu CM, Ho LT, Fuh MM et al. Effects on blood pressure of the a-glucosidase inhibitor acarbose
compared with the insulin enhancer glibenclamide in patients with hypertension and type 2 diabetes
mellitus. Diabetes Research & Clinical Practice 2003;60(2):111–118.
104 Phillips P, Karrasch J, Scott R et al. Effects on blood pressure of the a-glucosidase inhibitor acarbose
compared with the insulin enhancer glibenclamide in patients with hypertension and type 2 diabetes
mellitus. Diabetes Care 2003;26(2):269–273.
105 Bachmann W, Petzinna D, Raptis SA et al. Effects on blood pressure of the a-glucosidase inhibitor
acarbose compared with the insulin enhancer glibenclamide in patients with hypertension and type 2
diabetes mellitus. Clinical Drug Investigation 2003;23(10):679–686.
106 Lin BJ, Wu HP, Huang HS et al. Effects on blood pressure of the a-glucosidase inhibitor acarbose
compared with the insulin enhancer glibenclamide in patients with hypertension and type 2 diabetes
mellitus. Journal of Diabetes & its Complications 2003;17(4):179–185.
107 Segal P, Eliahou HE, Petzinna D et al. Effects on blood pressure of the a-glucosidase inhibitor acarbose
compared with the insulin enhancer glibenclamide in patients with hypertension and type 2 diabetes
mellitus. Clinical Drug Investigation 2005;25(9):589–595.
108 White TJ, Vanderplas A, Chang E et al. The costs of non-adherence to oral antihyperglycemic medication
in individuals with diabetes mellitus and concomitant diabetes mellitus and cardiovascular disease in a
managed care environment. Disease Management and Health Outcomes 2004;12(3):181–188.
109 Quilici S, Chancellor J, Maclaine G et al. Cost-effectiveness of acarbose for the management of impaired
glucose tolerance in Sweden. International Journal of Clinical Practice 2005;59(10):1143–1152.
110 Huang ES, Shook M, Jin L et al. The impact of patient preferences on the cost-effectiveness of intensive
glucose control in older patients with new-onset diabetes. Diabetes Care 2006;29(2):259–264.
111 Johnson JA, Simpson SH, Toth EL et al. Reduced cardiovascular morbidity and mortality associated with
metformin use in subjects with type 2 diabetes. Diabetic Medicine 2005;22(4):497–502.
112 Evans JMM, Ogston SA, EmslieSmith A et al. Risk of mortality and adverse cardiovascular outcomes in
type 2 diabetes: A comparison of patients treated with sulfonylureas and metformin. Diabetologia
2006;49(5):930–936.
113 National Institute for Health and Clinical Excellence. Glitazones in the treatment of type 2 diabetes (Review
of TA9 and TA21). (TA63). London: NICE, 2003.
114 European Medicines Agency. 2007. www.emea.europa.eu
115 Nissen SE, Wolski K. Effect of rosiglitazone on the risk of myocardial infarction and death from
cardiovascular causes. New England Journal of Medicine 2007;356(24):2457–2471.
116 Home PD, Pocock SJ, Beck-Nielsen H et al. Rosiglitazone evaluated for cardiovascular outcomes – an
interim analysis. New England Journal of Medicine 2007;357(1):28–38.
117 Lincoff AM, Wolski K, Nicholls SJ et al. Pioglitazone and risk of cardiovascular events in patients with type
2 diabetes mellitus: a meta-analysis of randomized trials. The Journal of the American Medical Association
2007;298(10):1180–1188.
118 Diamond GA, Bax L, Kaul S. Uncertain effects of rosiglitazone on the risk for myocardial infarction and
cardiovascular death. Annals of Internal Medicine 2007;147(8):578–581.
119 Singh S, Loke YK, Furberg CD. Long-term risk of cardiovascular events with rosiglitazone: a meta-analysis
[see comment]. The Journal of the American Medical Association 2007;298(10):1189–1195.
120 Lago RM, Singh PP, Nesto RW. Congestive heart failure and cardiovascular death in patients with
prediabetes and type 2 diabetes given thiazolidinediones: a meta-analysis of randomised clinical trials.
Lancet 2007;370(9593):1129–1136.
121 GlaxoSmithKline. Coronary heart disease outcomes in patients receiving antidiabetic agents. Uxbridge,
Middlesex: GlaxoSmithKline, 2008.
264
Type 2 diabetes](https://image.slidesharecdn.com/c13nicetype2diabetes2008-170327170946/85/C13-nice-type-2-diabetes-2008-263-320.jpg)



![180 Lechleitner M, Roden M, Haehling E et al. Insulin glargine in combination with oral antidiabetic drugs as
a cost-equivalent alternative to conventional insulin therapy in type 2 diabetes mellitus. Wiener Klinische
Wochenschrift 2005;117(17):593–598.
181 Drummond M, O’Brien B, Stoddart G, Torrance G. Methods for economic evaluation of health care
programmes, 2nd edn. Oxford: Oxford University Press, 2003.
182 Christiansen JS, Vaz JA, Metelko Z. Twice daily biphasic insulin aspart improves postprandial glycaemic
control more effectively than twice daily NPH insulin, with low risk of hypoglycaemia, in patients with
type 2 diabetes. Diabetes, Obesity & Metabolism 2003;5(6):446–454.
183 Kilo C, Mezitis N, Jain R et al. Starting patients with type 2 diabetes on insulin therapy using once-daily
injections of biphasic insulin aspart 70/30, biphasic human insulin 70/30, or NPH insulin in combination
with metformin. Journal of Diabetes & its Complications 2003;17(6):307–313.
184 Ceriello A, Del PS, Bue VJ et al. Premeal insulin lispro plus bedtime NPH or twice-daily NPH in patients
with type 2 diabetes: acute postprandial and chronic effects on glycemic control and cardiovascular risk
factors. Journal of Diabetes & its Complications 2007;21(1):20–27.
185 Siebenhofer A, Plank J, Berghold A et al. Short acting insulin analogues versus regular human insulin in
patients with diabetes mellitus. Cochrane Database of Systematic Reviews 2006;(2):CD003287.
186 Boehm BO, Home PD, Behrend C et al. Premixed insulin aspart 30 vs. premixed human insulin 30/70
twice daily: a randomized trial in type 1 and type 2 diabetic patients [erratum appears in Diabetic Medicine.
2002 Sep;19(9):797]. Diabetic Medicine 2002;19(5):393–399.
187 Boehm BO, Vaz JA, Brondsted L et al. Long-term efficacy and safety of biphasic insulin aspart in patients
with type 2 diabetes. European Journal of Internal Medicine 2004;15(8):496–502.
188 Abrahamian H, Ludvik B, Schernthaner G et al. Improvement of glucose tolerance in type 2 diabetic
patients: traditional vs. modern insulin regimens (results from the Austrian Biaspart Study). Hormone &
Metabolic Research 2005;37(11):684–689.
189 Schernthaner G, Kopp HP, Ristic S et al. Metabolic control in patients with type 2 diabetes using Humalog
Mix50 injected three times daily: crossover comparison with human insulin 30/70. Hormone & Metabolic
Research 2004;36(3):188–193.
190 Ligthelm RJ, Mouritzen U, Lynggaard H et al. Biphasic insulin aspart given thrice daily is as efficacious as
a basal-bolus insulin regimen with four daily injections: a randomised open-label parallel group four
months comparison in patients with type 2 diabetes. Experimental & Clinical Endocrinology & Diabetes
2006;114(9):511–519.
191 Joshi SR, Kalra S, Badgandi M et al. Designer insulins regimens in clinical practice – pilot multicenter
Indian study. Journal of the Association of Physicians of India 2005;53(Sept):775–779.
192 Davies M, Storms F, Shutler S. Initiation of Insulin Glargine in type 2 patients with suboptimal glycaemic
control on twice-daily premix insulin: results from the AT.LANTUS trial. Diabetologia 2004;47(Suppl 1):
319.
193 National Institute for Clinical Excellence. The clinical effectiveness and cost effectiveness of long acting insulin
analogues for diabetes (TA53). London: NICE, 2002.
194 Rosenstock J, Dailey G, Massi-Benedetti M et al. Reduced hypoglycemia risk with insulin glargine: a meta-
analysis comparing insulin glargine with human NPH insulin in type 2 diabetes. Diabetes Care 2005;
28(4):950–955.
195 Horvath K, Jeitler K, Berghold A et al. Long-acting insulin analogues versus NPH insulin (human isophane
insulin) for type 2 diabetes mellitus. Cochrane Database of Systematic Reviews 2007;(2):CD005613.
196 Yki JH, Kauppinen MR, Tiikkainen M et al. Insulin glargine or NPH combined with metformin in type 2
diabetes: the LANMET study. Diabetologia 2006;49(3):442–451.
197 Herman WH, Ilag LL, Johnson SL et al. A clinical trial of continuous subcutaneous insulin infusion versus
multiple daily injections in older adults with type 2 diabetes. Diabetes Care 2005;28(7):1568–1573.
198 Raskin P, Allen E, Hollander P et al. Initiating insulin therapy in type 2 diabetes: a comparison of biphasic
and basal insulin analogs. Diabetes Care 2005;28(2):260–265.
268
Type 2 diabetes](https://image.slidesharecdn.com/c13nicetype2diabetes2008-170327170946/85/C13-nice-type-2-diabetes-2008-267-320.jpg)







![332 National Institute for Health and Clinical Excellence. Clopidogrel in the treatment of non-ST-segment-
elevation acute coronary syndrome. (TA80). London: NICE, 2004.
333 Weintraub WS, Mahoney EM, Lamy A et al. Long-term cost-effectiveness of clopidogrel given for up to
one year in patients with acute coronary syndromes without ST-segment elevation. Journal of the American
College of Cardiology 2005;45(6):838–845.
334 Ringborg A, Lindgren P, Jonsson B. The cost-effectiveness of dual oral antiplatelet therapy following
percutaneous coronary intervention: a Swedish analysis of the CREDO trial. European Journal of Health
Economics 2005;6(4):354–362.
335 Cowper PA, Udayakumar K, Sketch MH Jr et al. Economic effects of prolonged clopidogrel therapy after
percutaneous coronary intervention. Journal of the American College of Cardiology 2005;45(3):369–376.
336 Harvey JN. Trends in the prevalence of diabetic nephropathy in type 1 and type 2 diabetes. Current
Opinion in Nephrology & Hypertension 2003;12(3):317–322.
337 Banerjee S, Ghosh US, Saha SJ. Role of GFR estimation in assessment of the status of nephropathy in type
2 diabetes mellitus. Journal of the Association of Physicians of India 2005;53:181–4.
338 Baskar V, Venugopal H, Holland MR et al. Clinical utility of estimated glomerular filtration rates in
predicting renal risk in a district diabetes population. Diabetic Medicine 2006;23(10):1057–1060.
339 Cortes SL, Martinez RH, Hernandez JL et al. Utility of the Dipstick Micraltest II in the screening of
microalbuminuria of diabetes mellitus type 2 and essential hypertension. Revista de Investigacion Clinica
2006;58(3):190–197.
340 Incerti J, Zelmanovitz T, Camargo JL et al. Evaluation of tests for microalbuminuria screening in patients
with diabetes. Nephrology Dialysis Transplantation 2005;20(11):2402–2407.
341 MacIsaac RJ, Tsalamandris C, Panagiotopoulos S et al. Nonalbuminuric renal insufficiency in type 2
diabetes. Diabetes Care 2004;27(1):195–200.
342 Middleton RJ, Foley RN, Hegarty J et al. The unrecognized prevalence of chronic kidney disease in
diabetes. Nephrology Dialysis Transplantation 2006;21(1):88–92.
343 Parikh CR, Fischer MJ, Estacio R et al. Rapid microalbuminuria screening in type 2 diabetes mellitus:
simplified approach with Micral test strips and specific gravity [erratum appears in Nephrol Dial
Transplant 2004;19(9):2425]. Nephrology Dialysis Transplantation 2004;19(7):1881–1885.
344 Poggio ED, Wang X, Greene T et al. Performance of the modification of diet in renal disease and
Cockcroft-Gault equations in the estimation of GFR in health and in chronic kidney disease. Journal of the
American Society of Nephrology 2005;16(2):459–466.
345 Rigalleau V, Lasseur C, Perlemoine C et al. A simplified Cockcroft-Gault formula to improve the
prediction of the glomerular filtration rate in diabetic patients. Diabetes & Metabolism 2006;32(1):56–62.
346 Younis N, Broadbent DM, Vora JP et al. Incidence of sight-threatening retinopathy in patients with type
2 diabetes in the Liverpool Diabetic Eye Study: a cohort study. Lancet 2003;361(9353):195–200.
347 UK National Screening Committee. Essential elements in developing a diabetic retinopathy screening
programme. Workbook 4:(1–79). Available from: UK National Screening Committee.
348 Max MB, Lynch SA, Muir J et al. Effects of desipramine, amitriptyline, and fluoxetine on pain in diabetic
neuropathy. New England Journal of Medicine 1992;326(19):1250–1256.
349 Sindrup SH, Gram LF, Skjold T et al. Clomipramine vs desipramine vs placebo in the treatment of diabetic
neuropathy symptoms. A double-blind cross-over study. British Journal of Clinical Pharmacology
1990;30(5):683–691.
350 Sindrup SH, Tuxen C. Lack of effect of mianserin on the symptoms of diabetic neuropathy. European
Journal of Clinical Pharmacology 1992;43(3):251–255.
351 Morello CM, Leckband SG, Stoner CP et al. Randomized double-blind study comparing the efficacy of
gabapentin with amitriptyline on diabetic peripheral neuropathy pain. Archives of Internal Medicine
1999;159(16):1931–1937.
352 Jose VM, Bhansali A, Hota D et al. Randomized double-blind study comparing the efficacy and safety of
lamotrigine and amitriptyline in painful diabetic neuropathy. Diabetic Medicine 2007;24(4):377–383.
276
Type 2 diabetes](https://image.slidesharecdn.com/c13nicetype2diabetes2008-170327170946/85/C13-nice-type-2-diabetes-2008-275-320.jpg)


