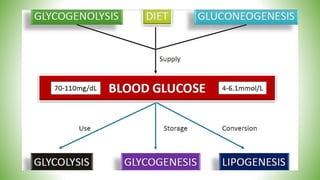
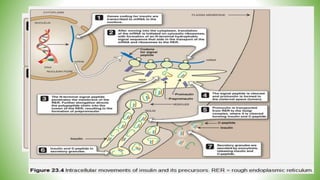
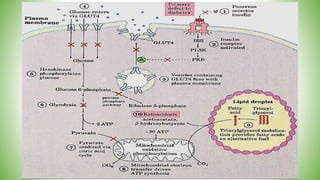
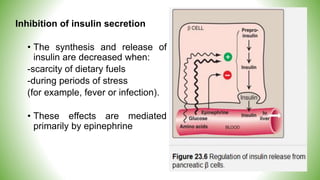
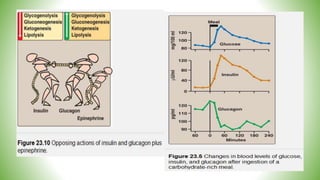
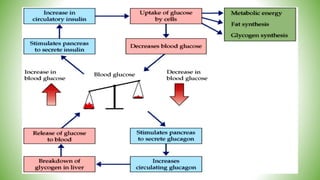
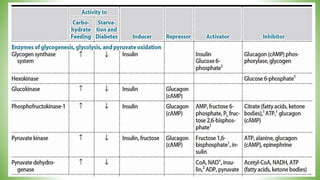
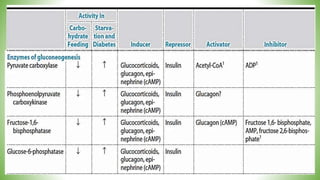
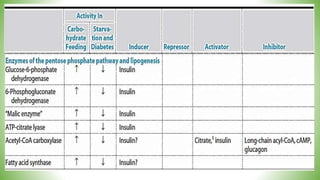
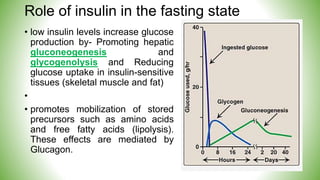
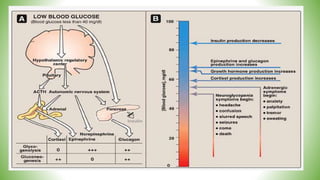
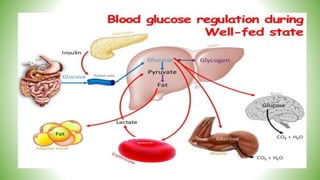
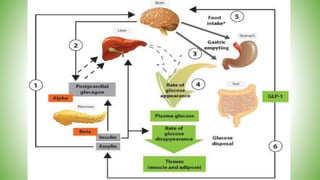
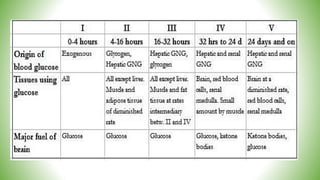
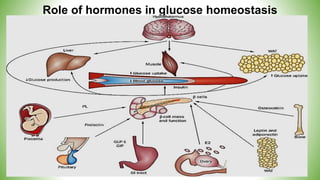
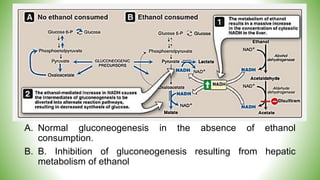
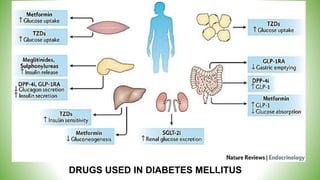
Blood sugar homeostasis is maintained through a balance of hepatic glucose production and peripheral glucose uptake regulated by hormones like insulin and glucagon. In the fasting state, low insulin and high glucagon promote gluconeogenesis and glycogenolysis to increase glucose production. After eating, high insulin and low glucagon stimulate glucose uptake in tissues and inhibit production. Disruptions can cause hyperglycemia or hypoglycemia.