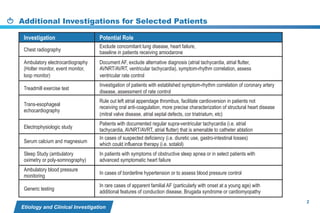
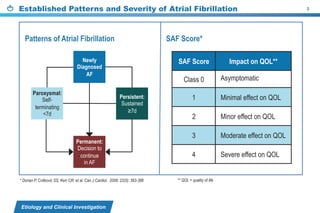
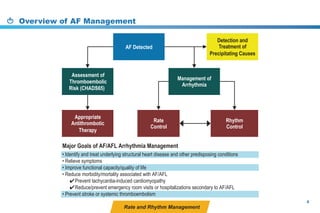
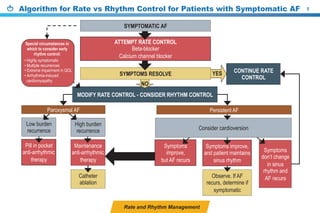
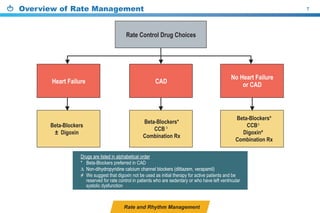
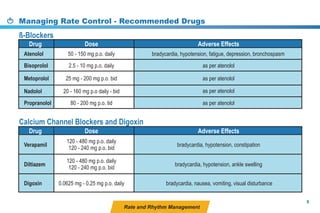
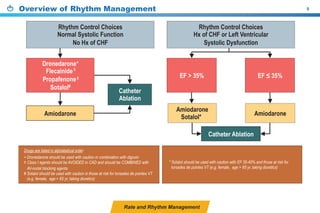
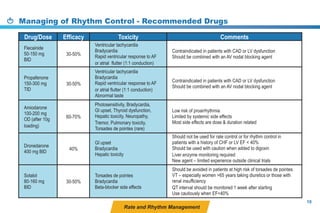
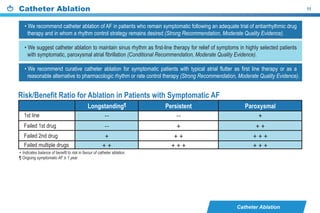
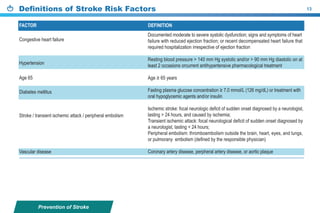
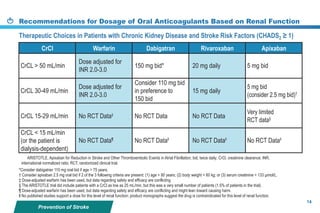
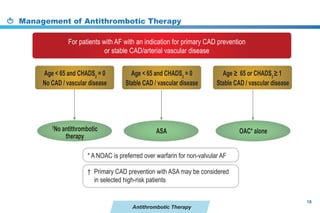
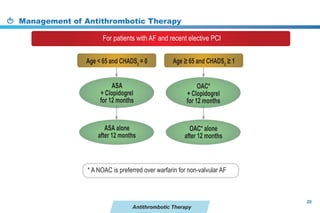
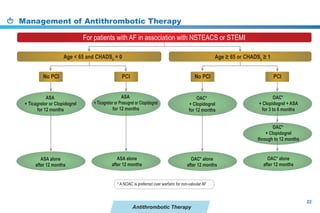
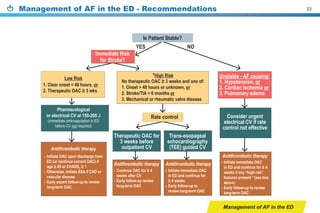
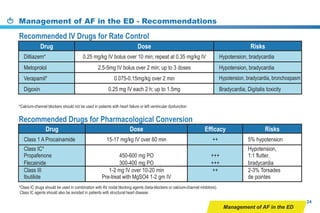
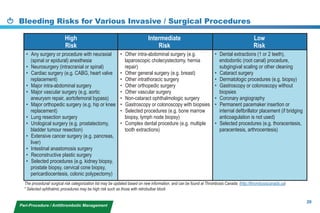
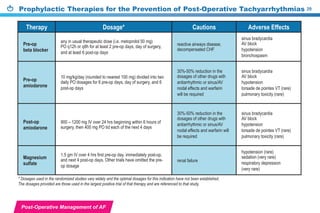
This document provides a summary of the CCS AF guidelines from 2010 to present. It includes diagnostic and treatment recommendations for atrial fibrillation management. The recommendations are intended to provide a practical clinical approach but are not a substitute for clinical judgment. Adherence to the guidelines will not necessarily produce successful outcomes in every case.