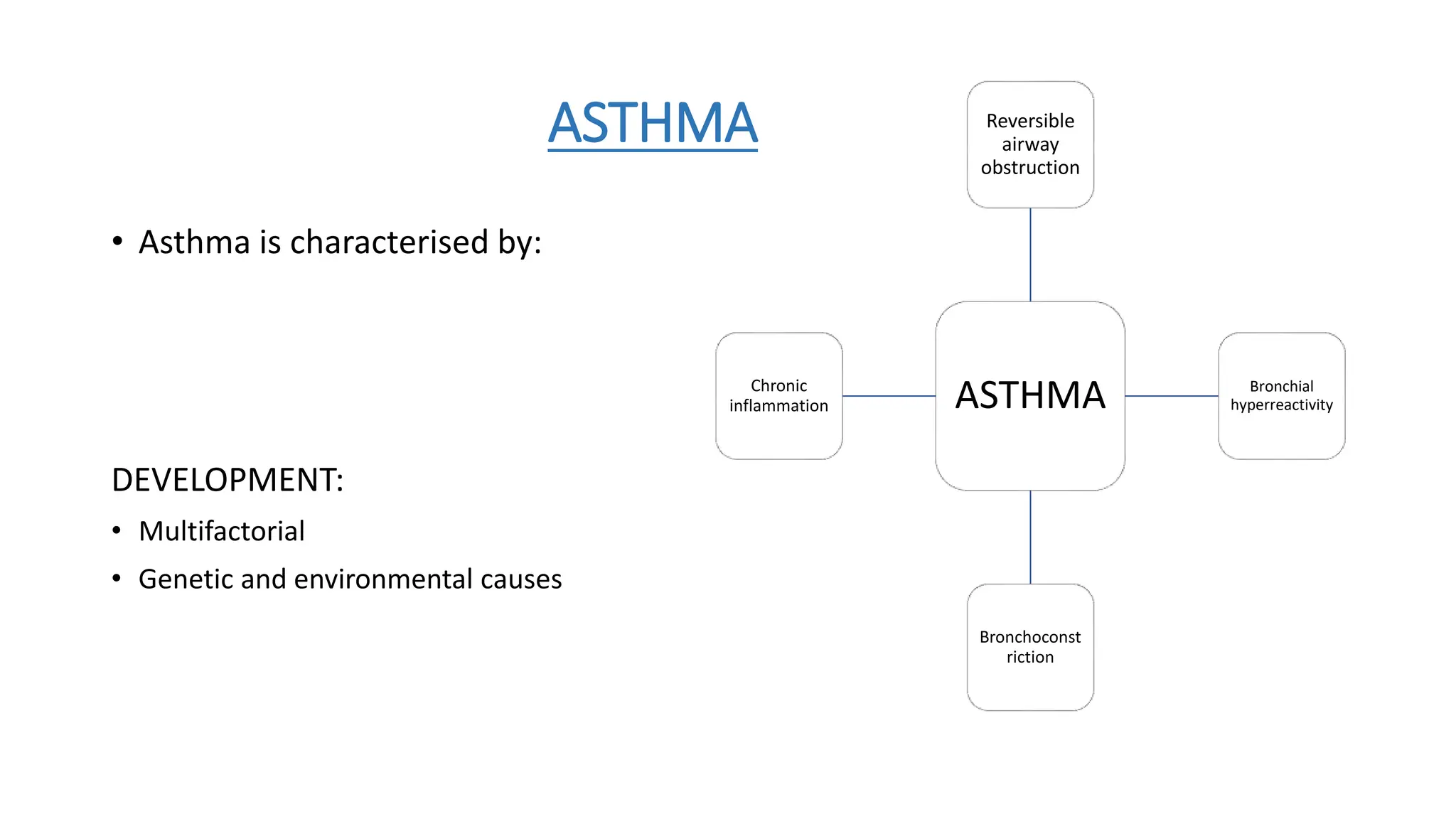
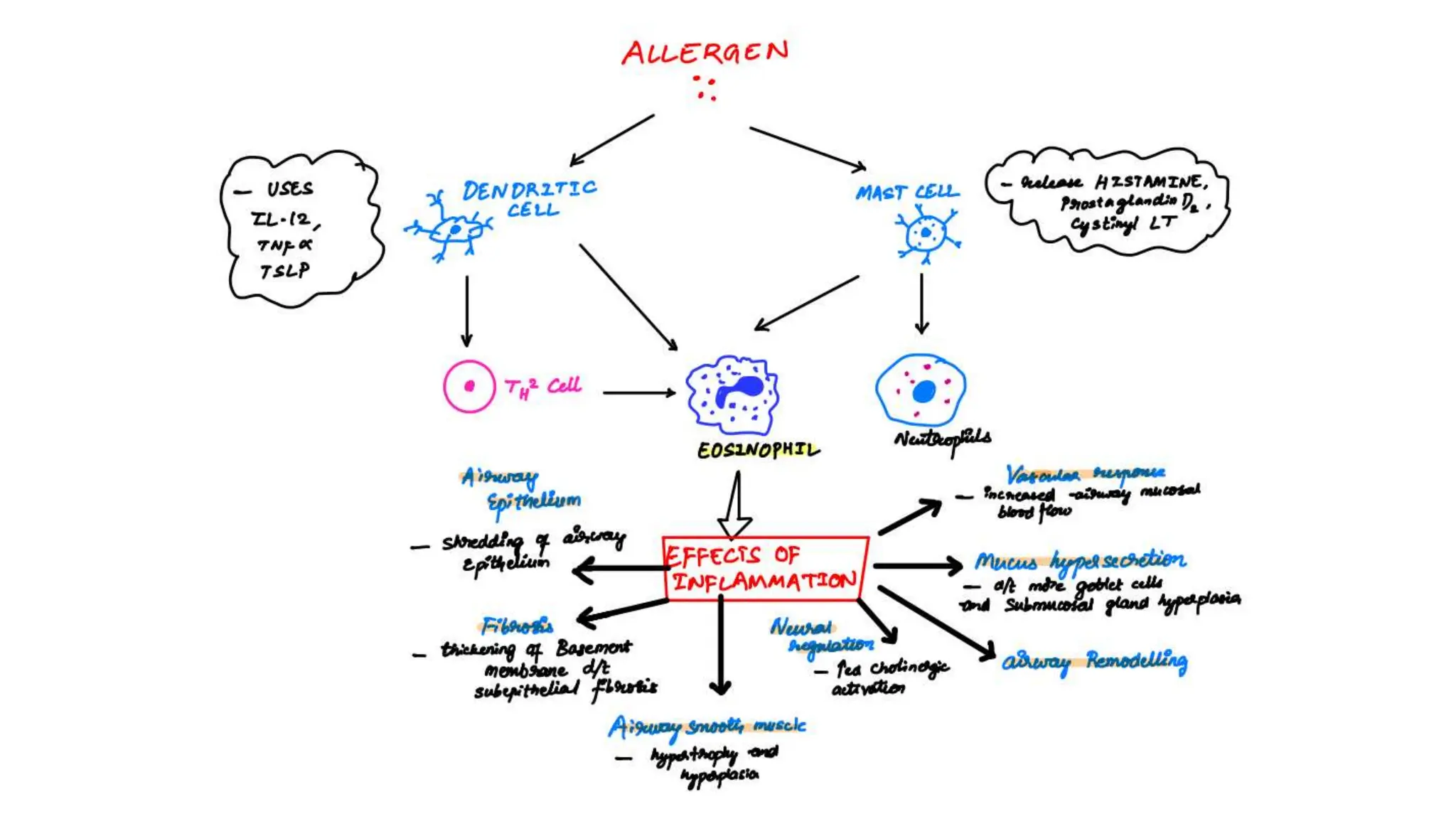
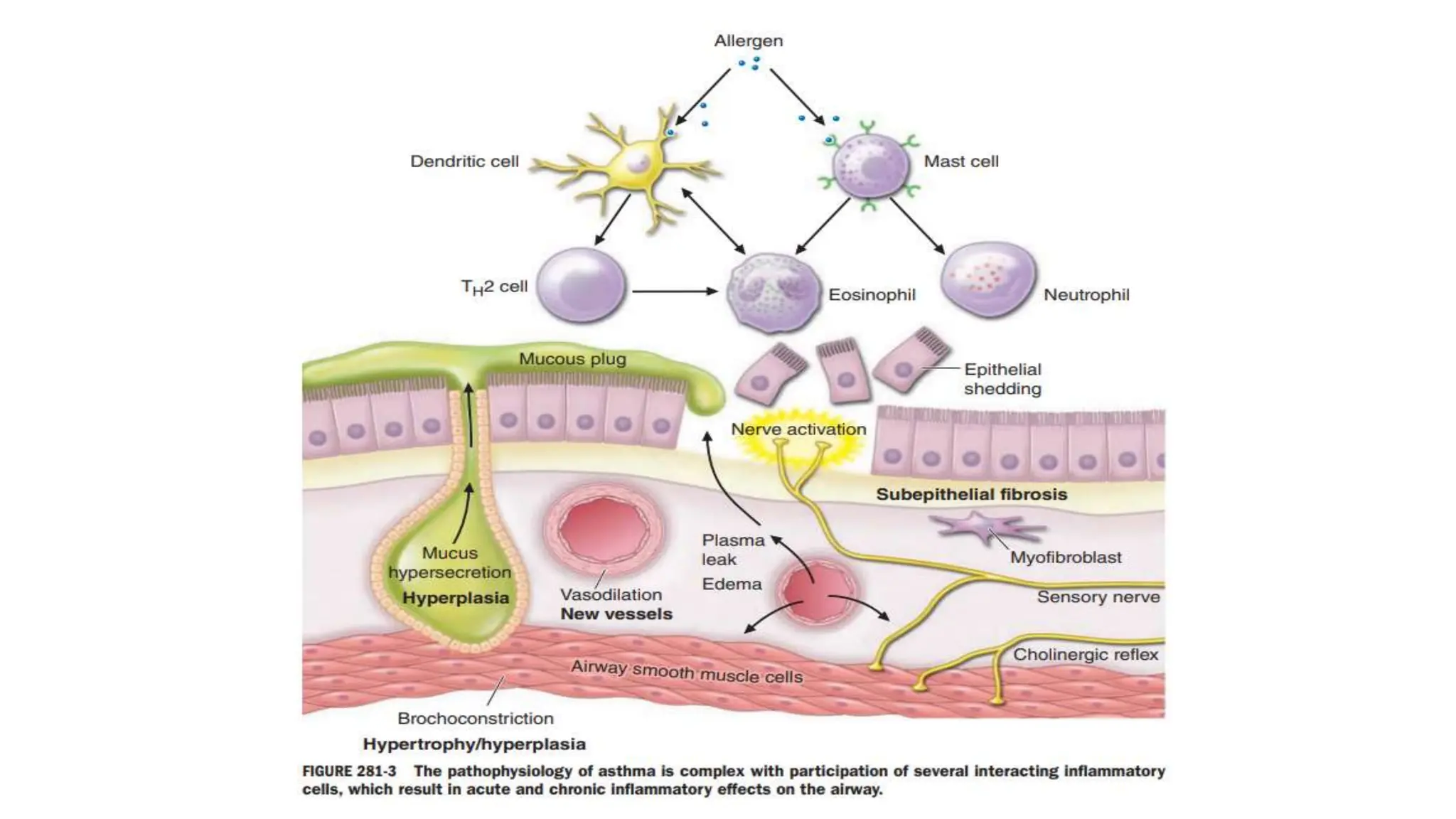
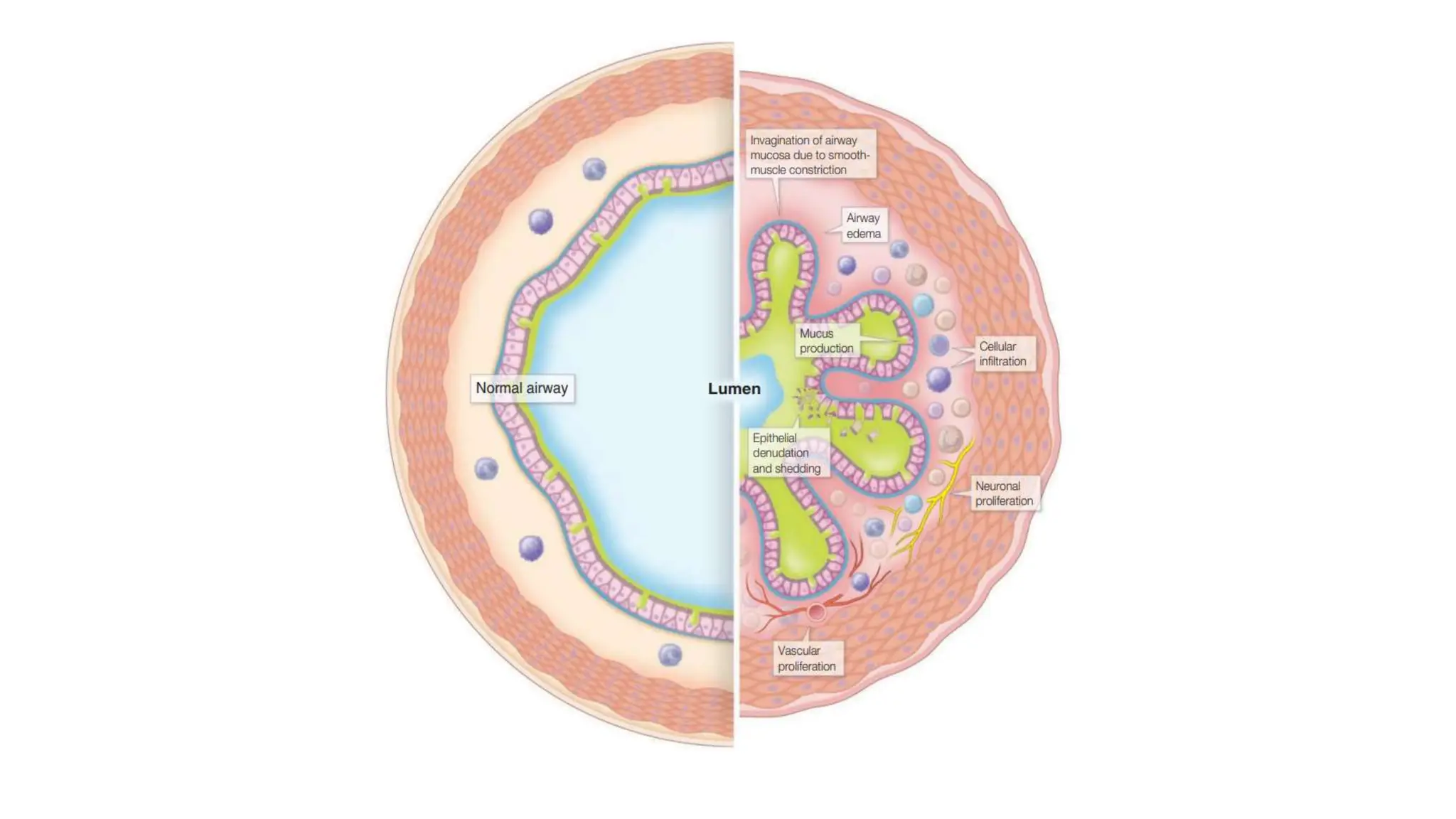
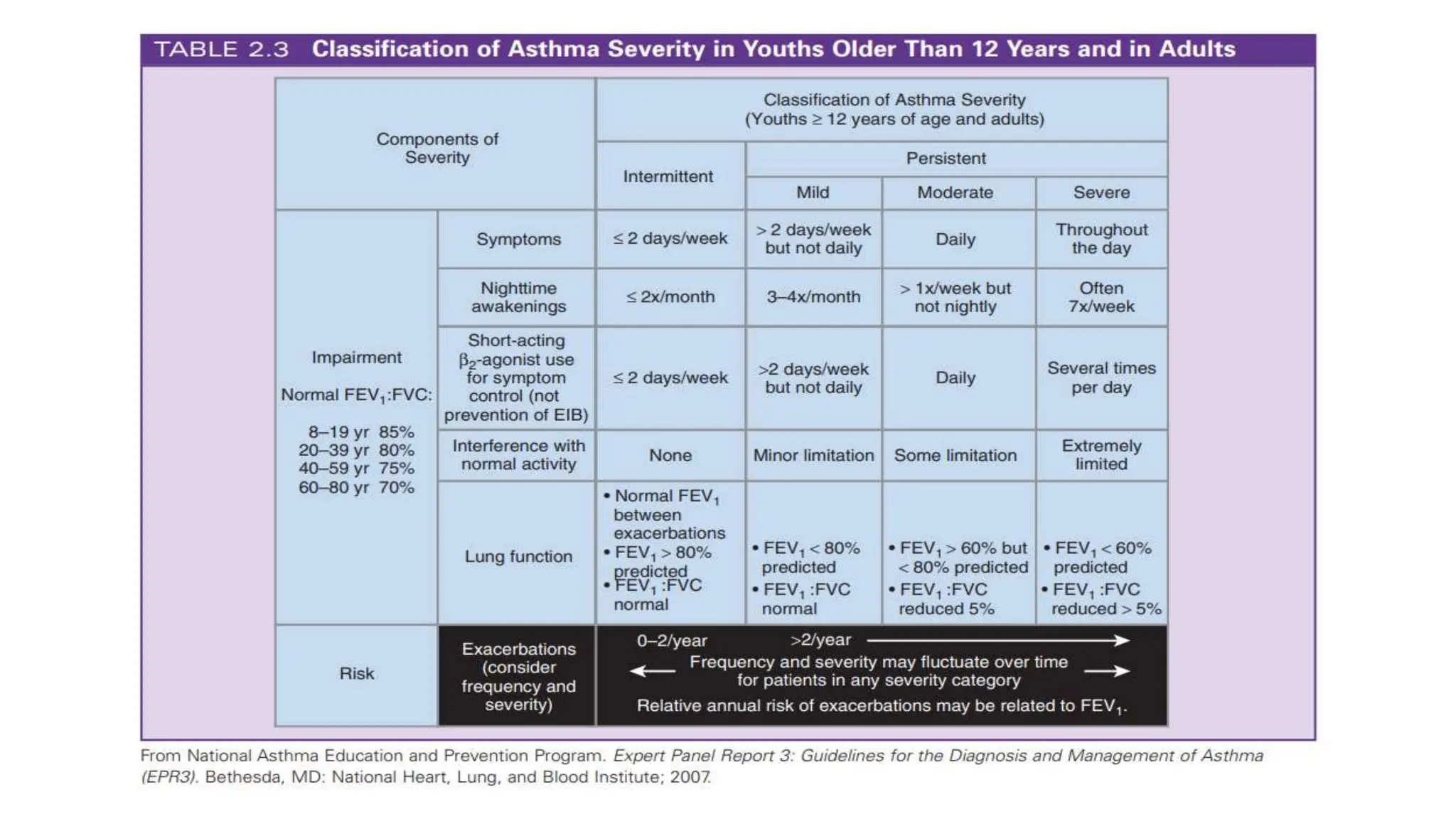
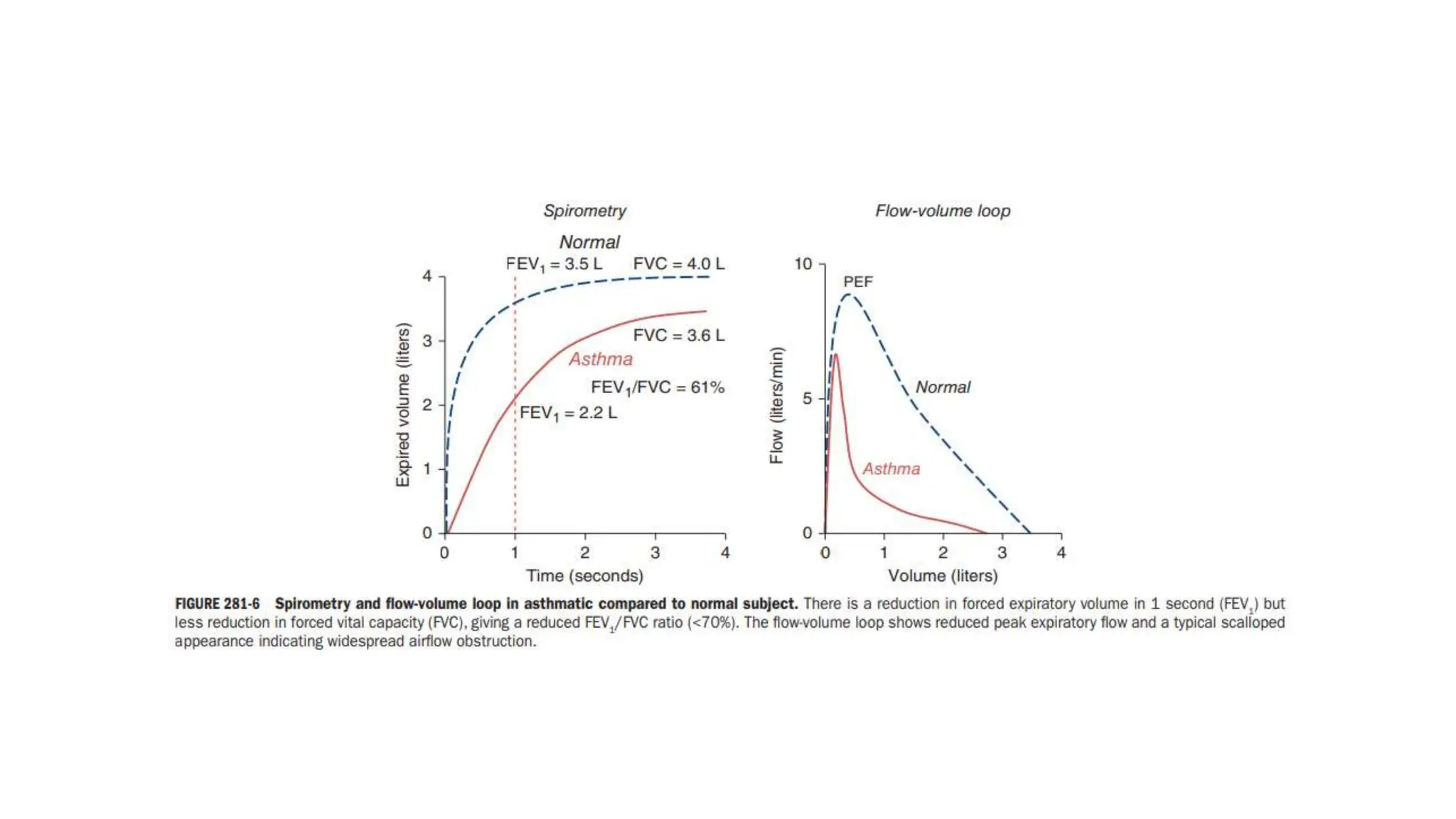
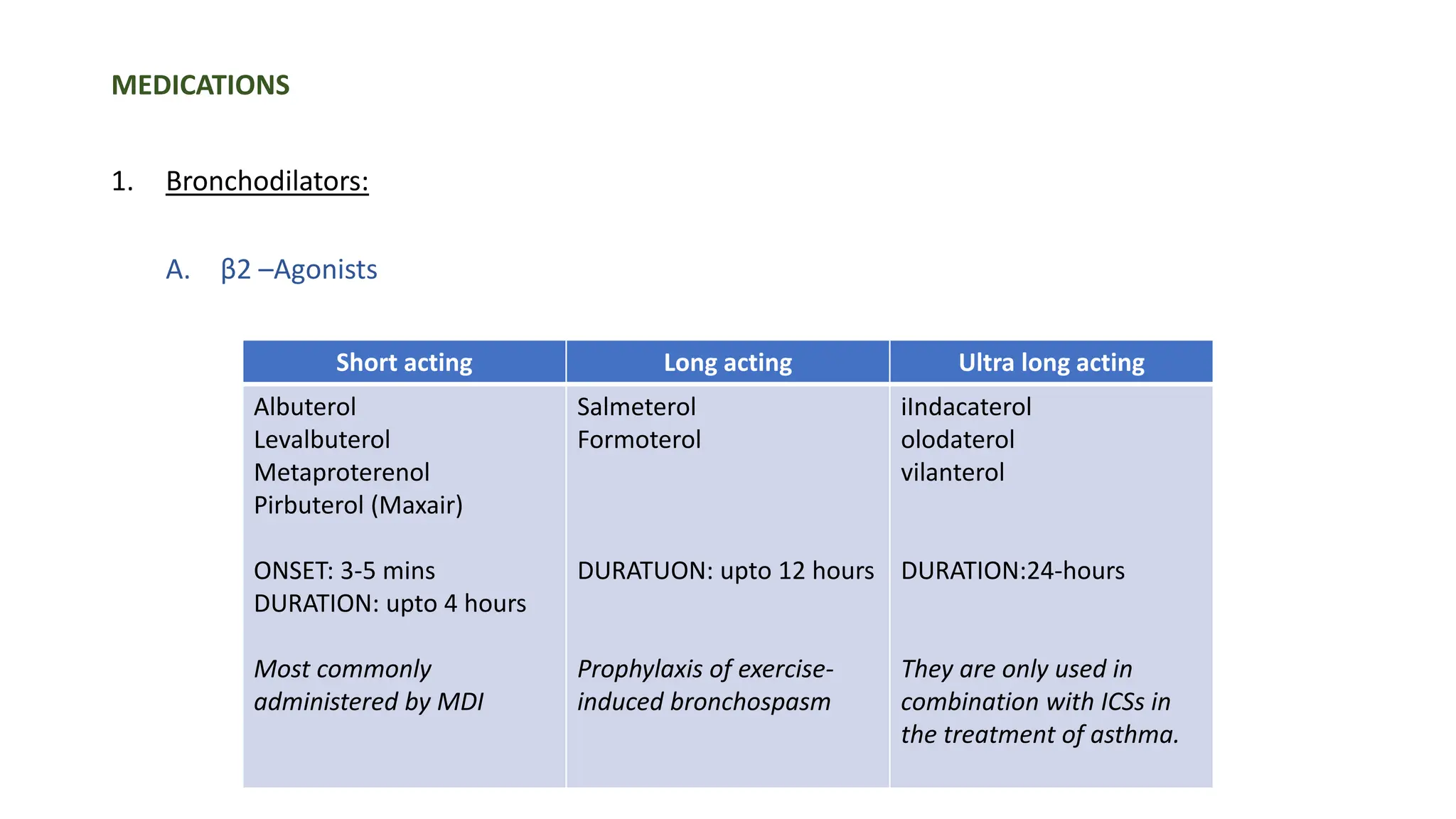
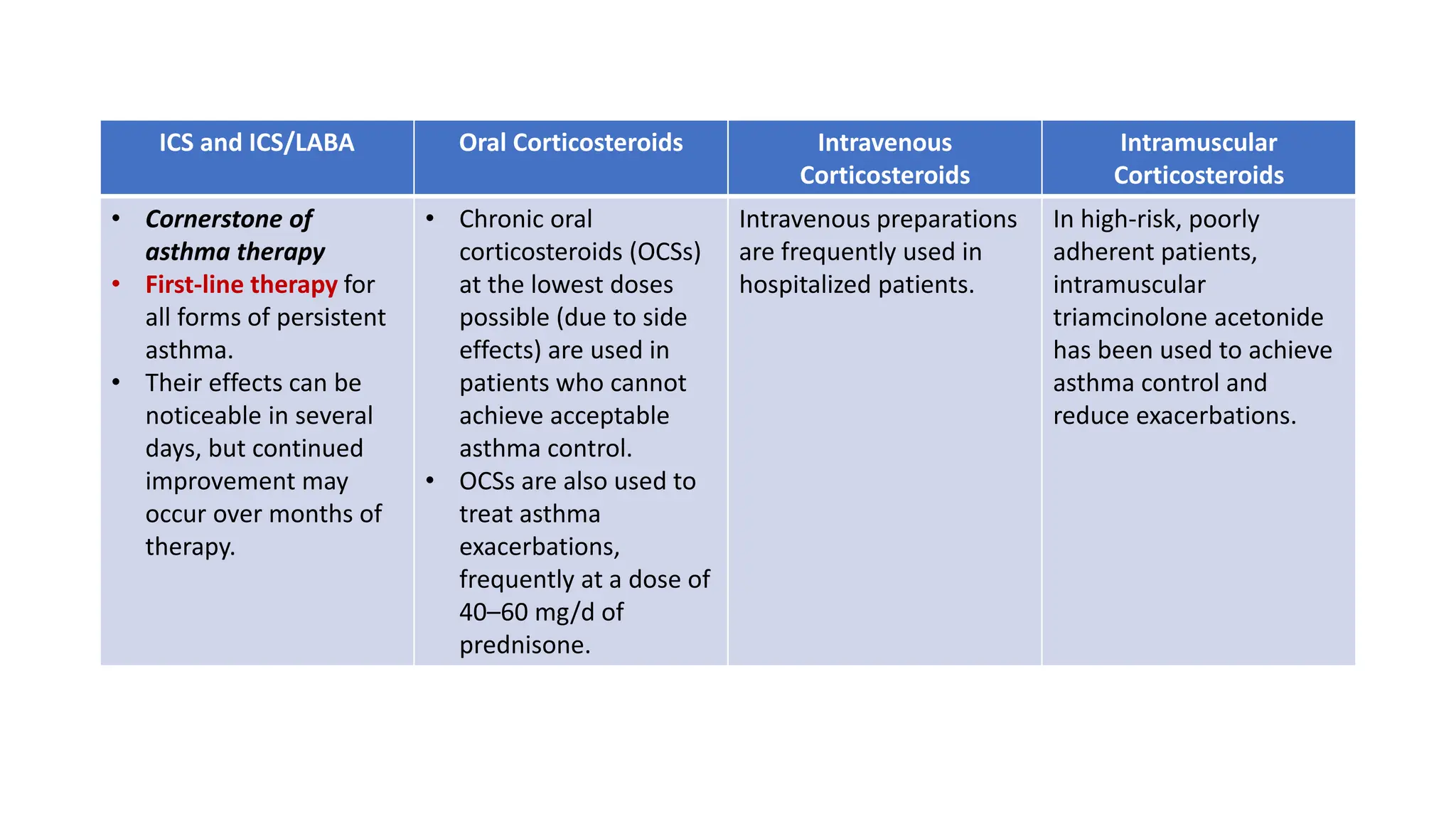
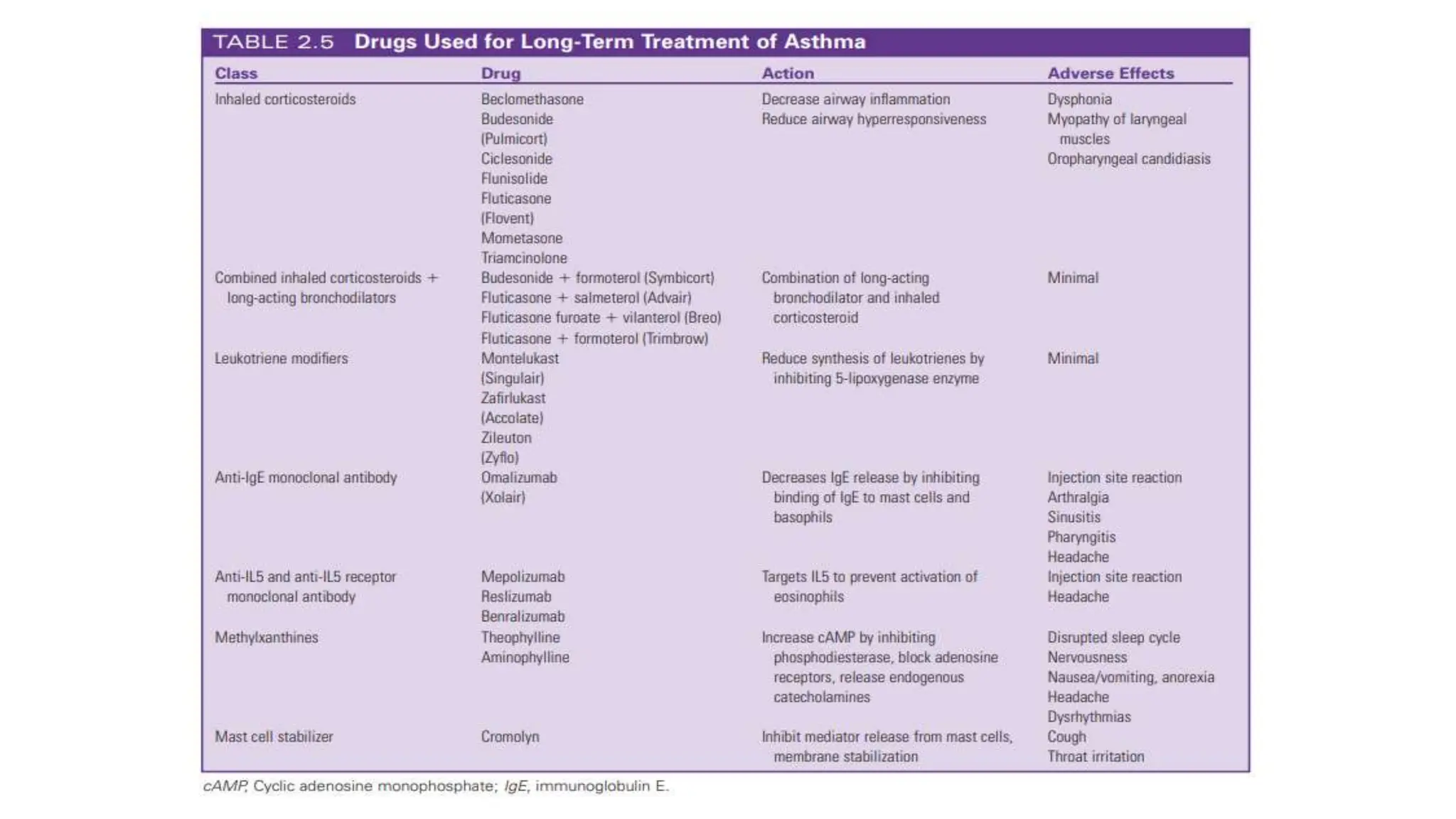
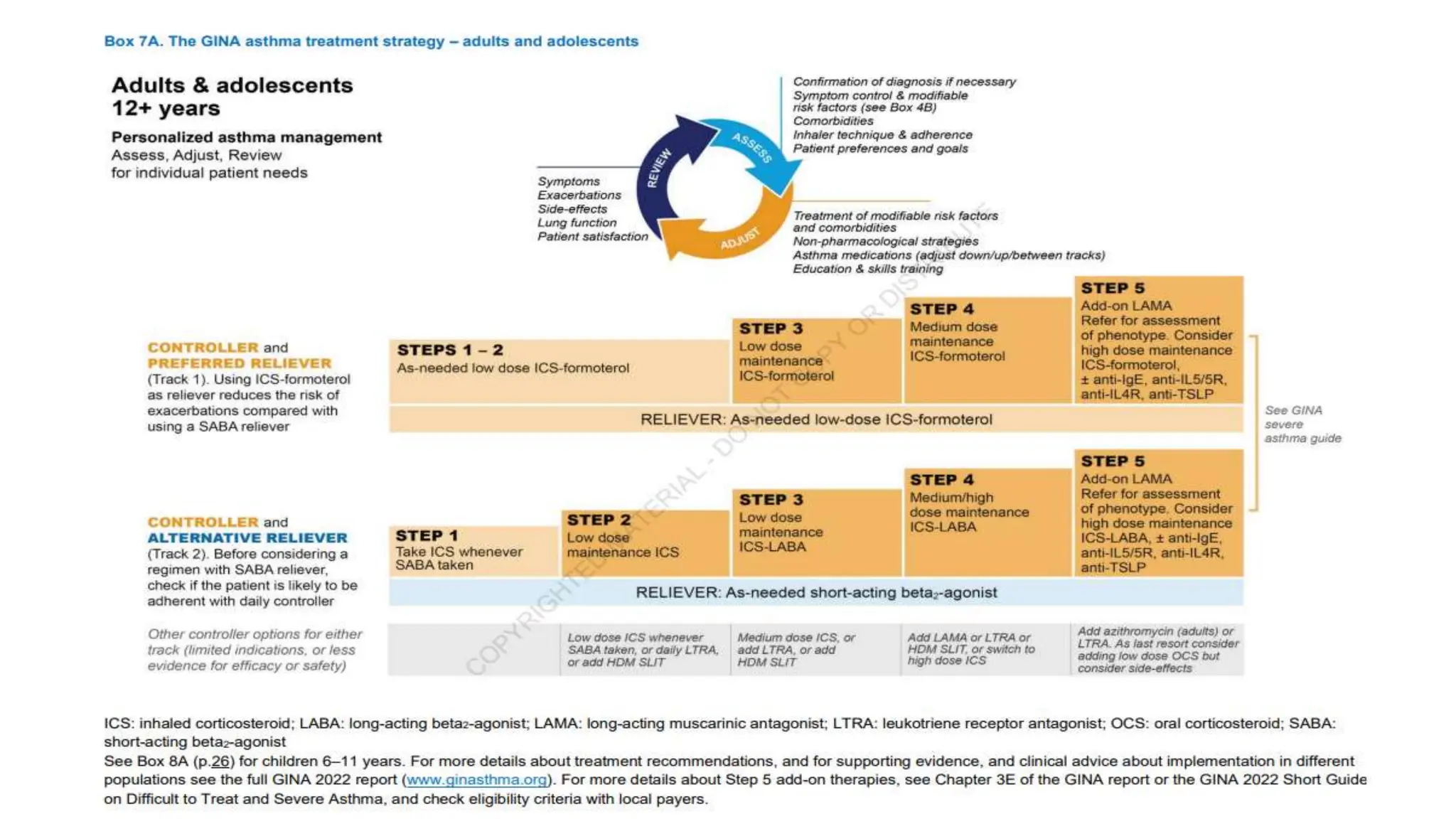
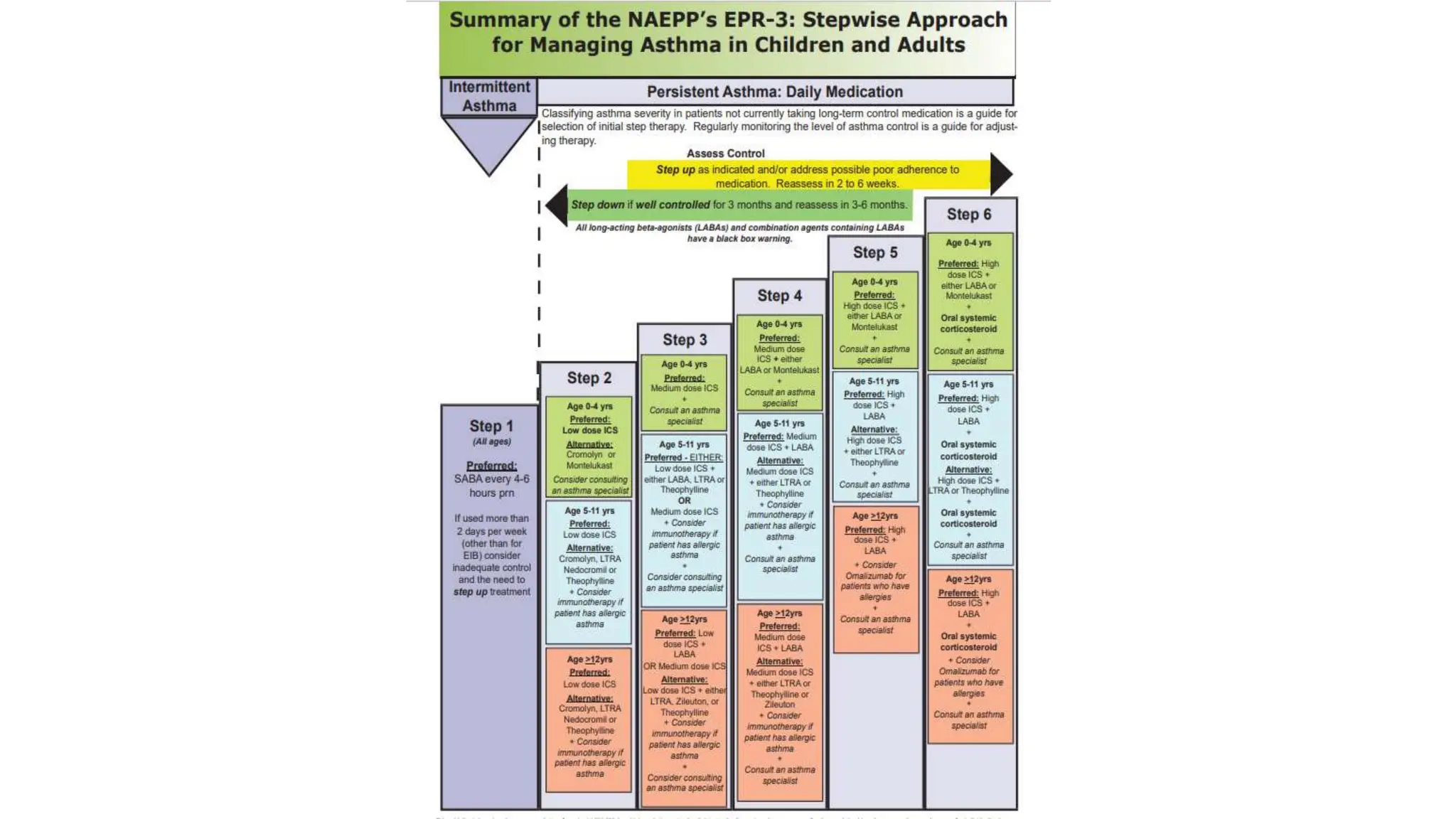
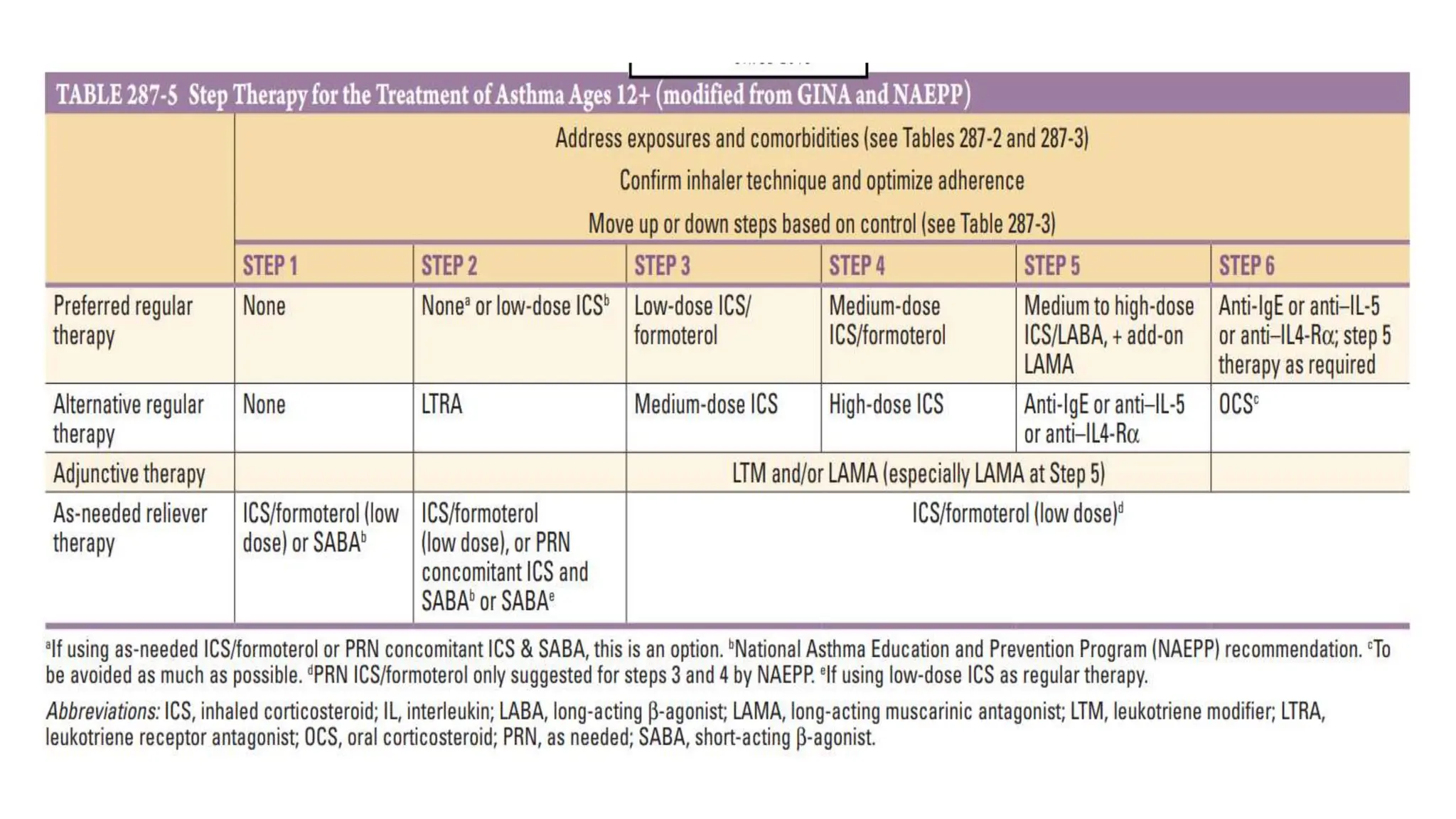
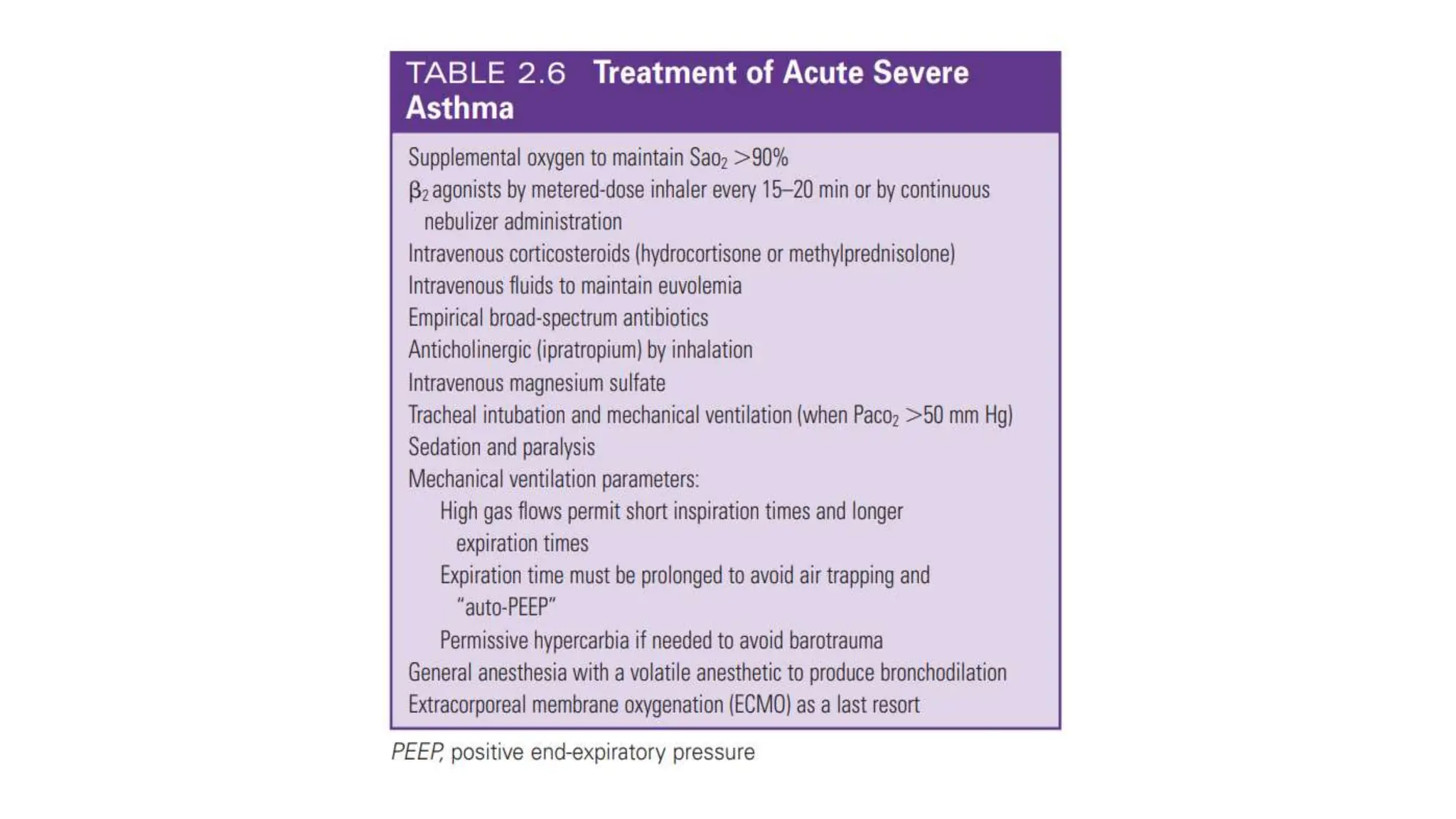
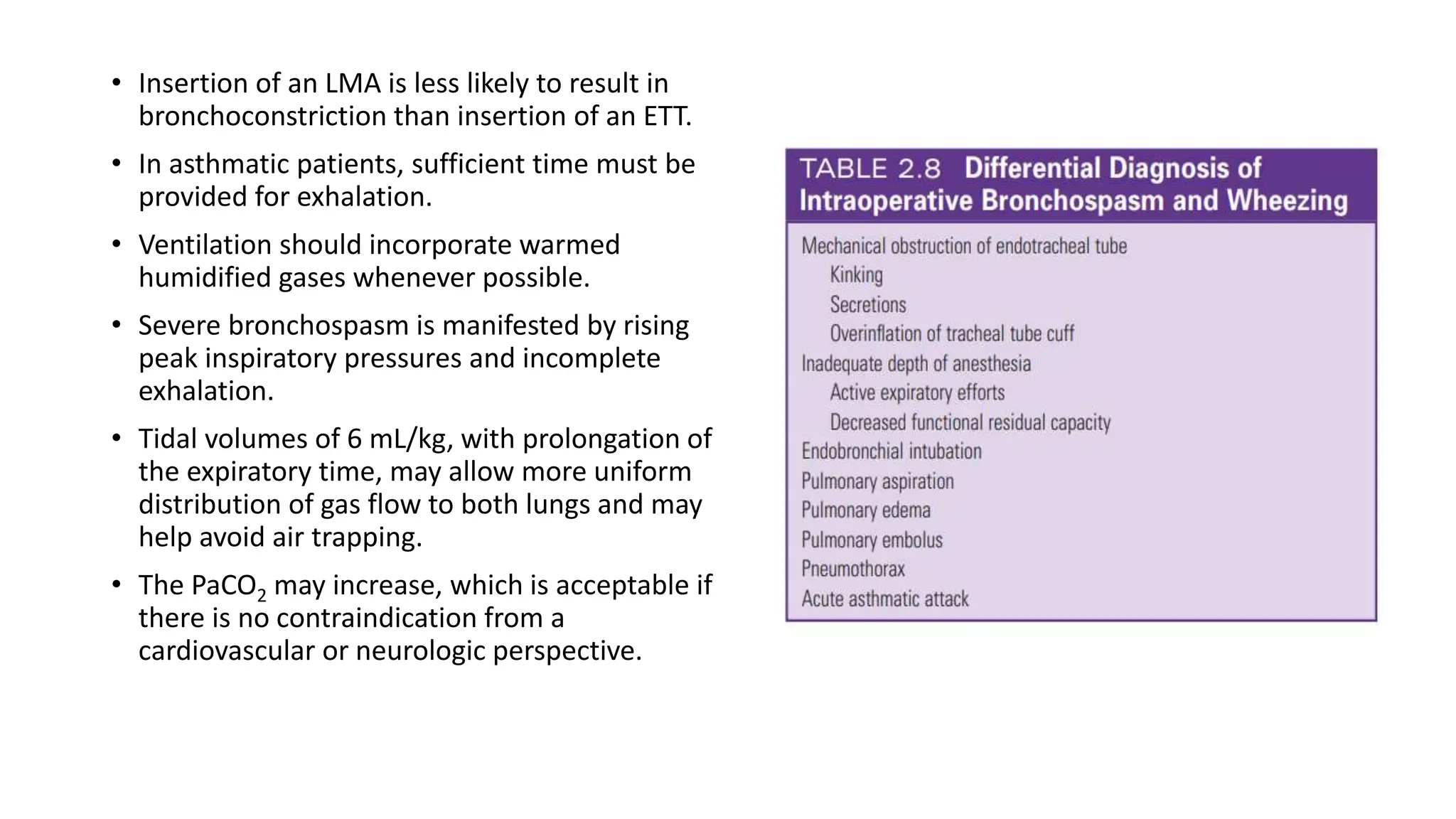
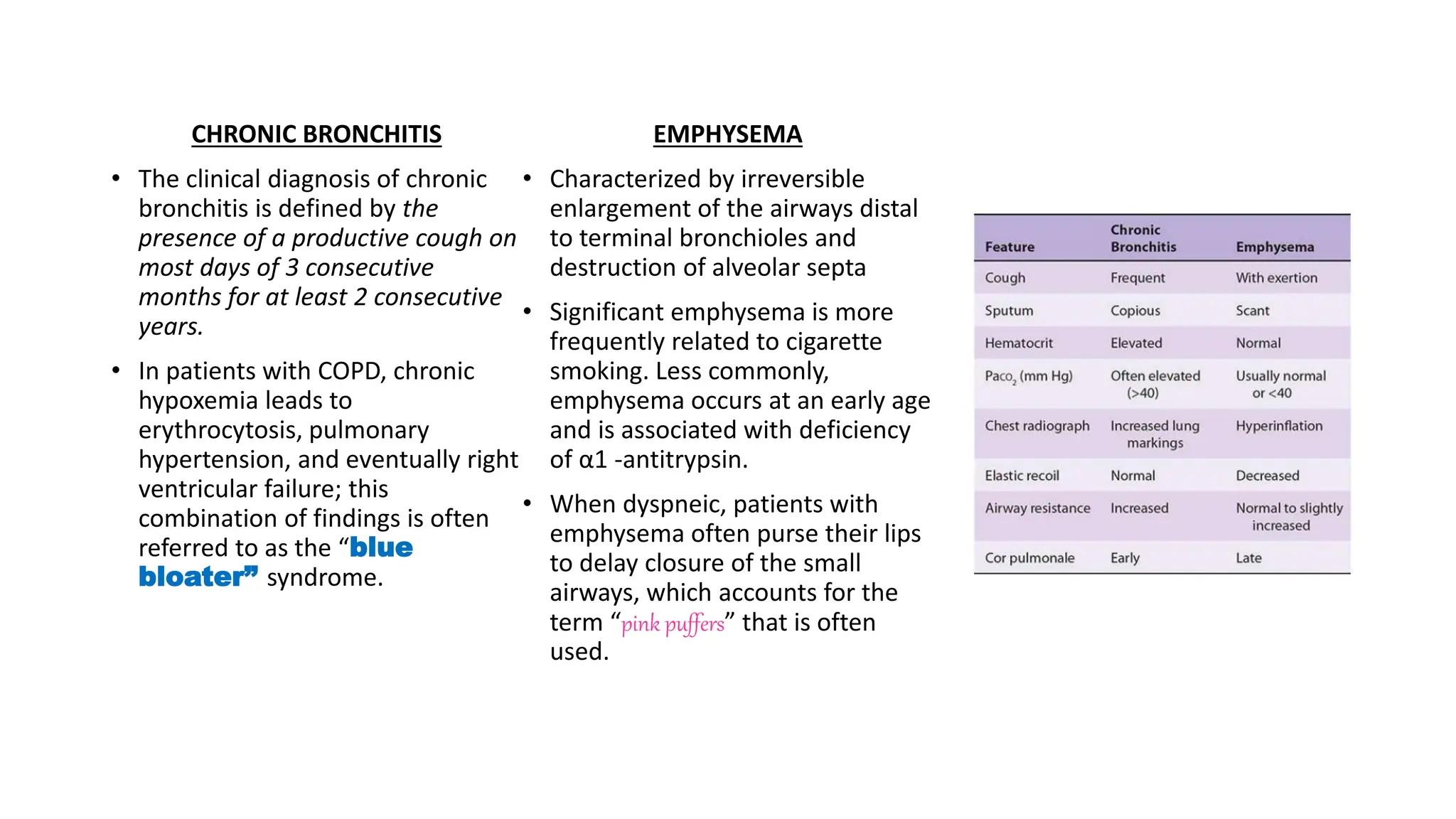
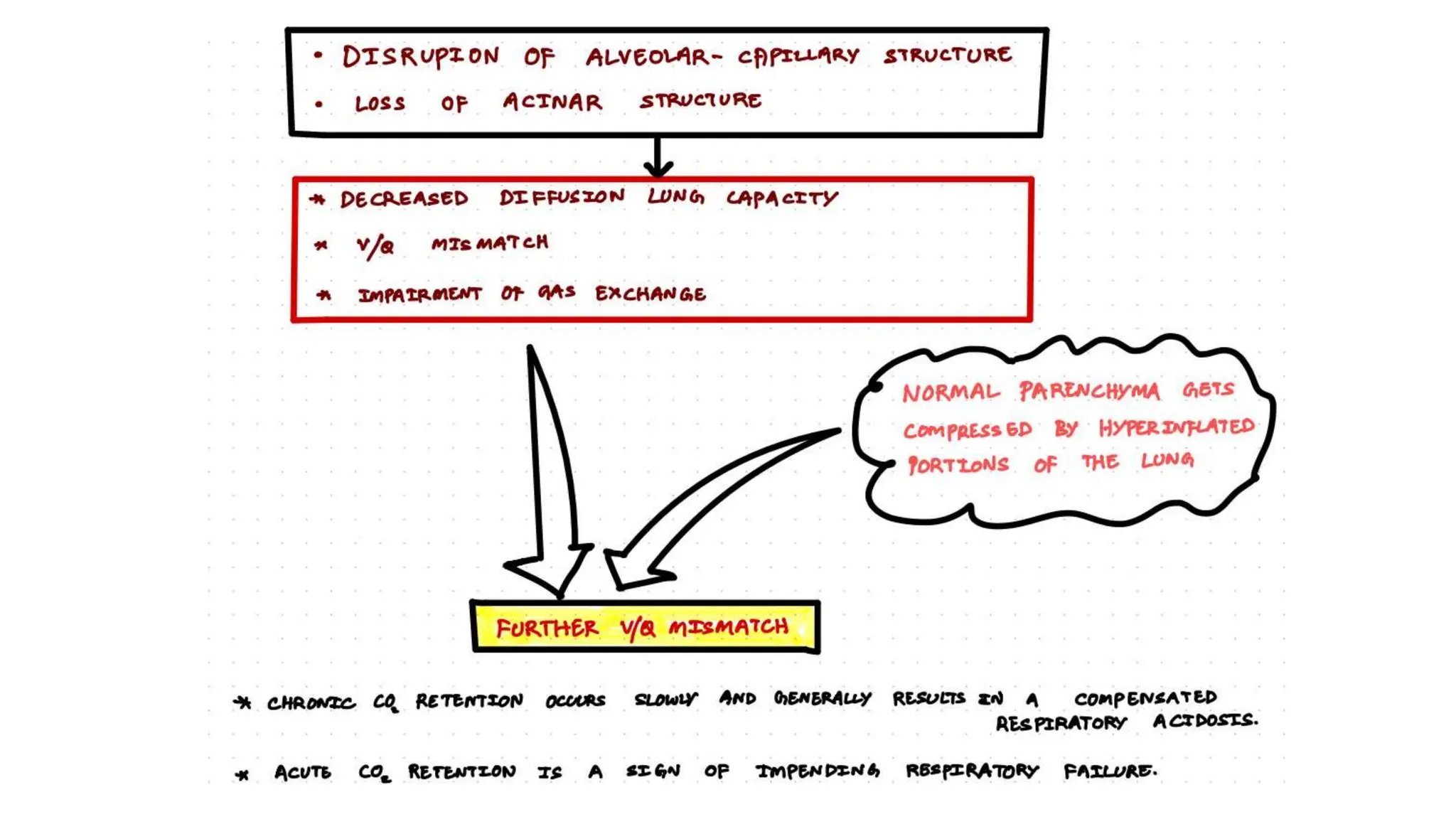
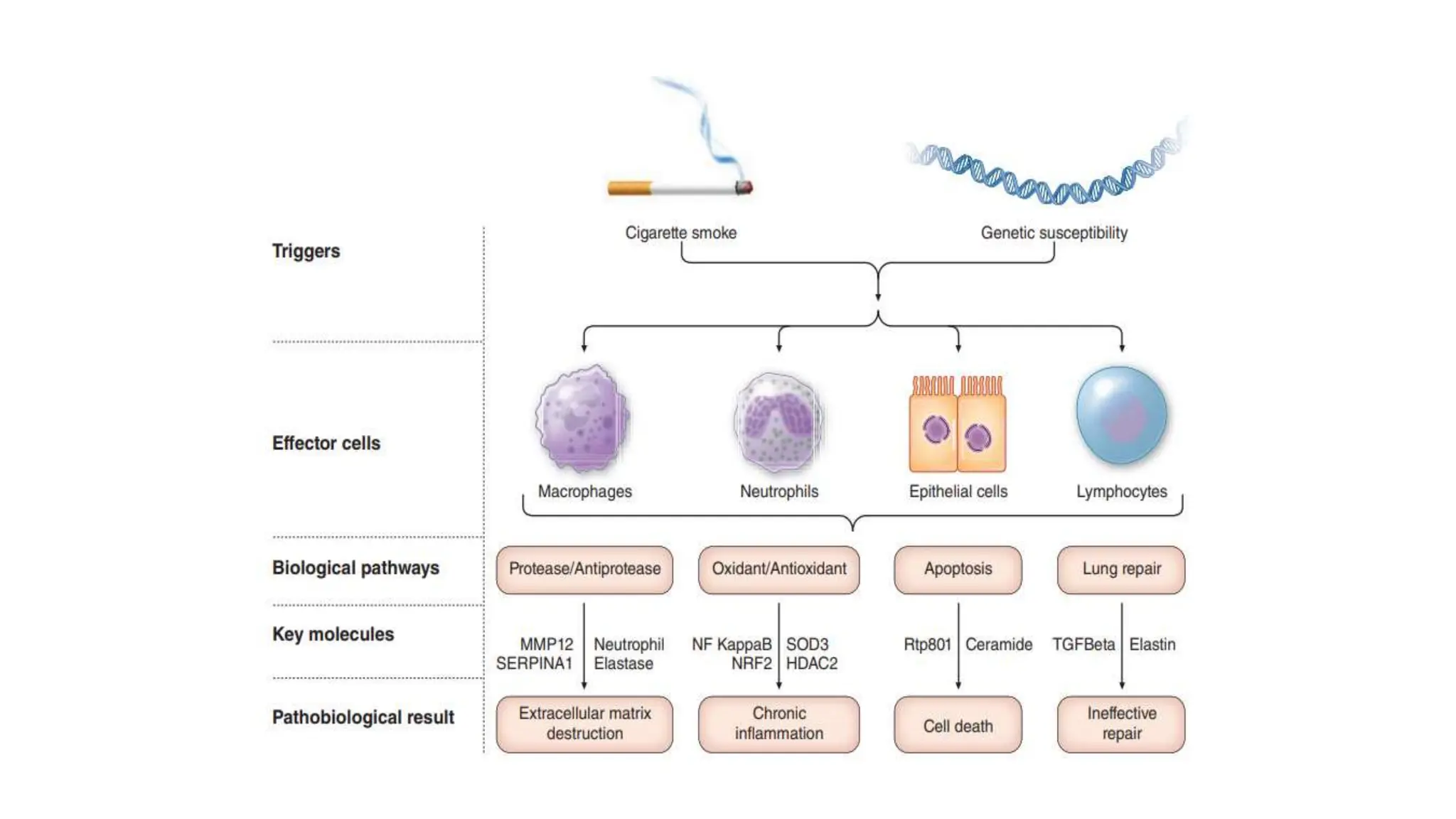
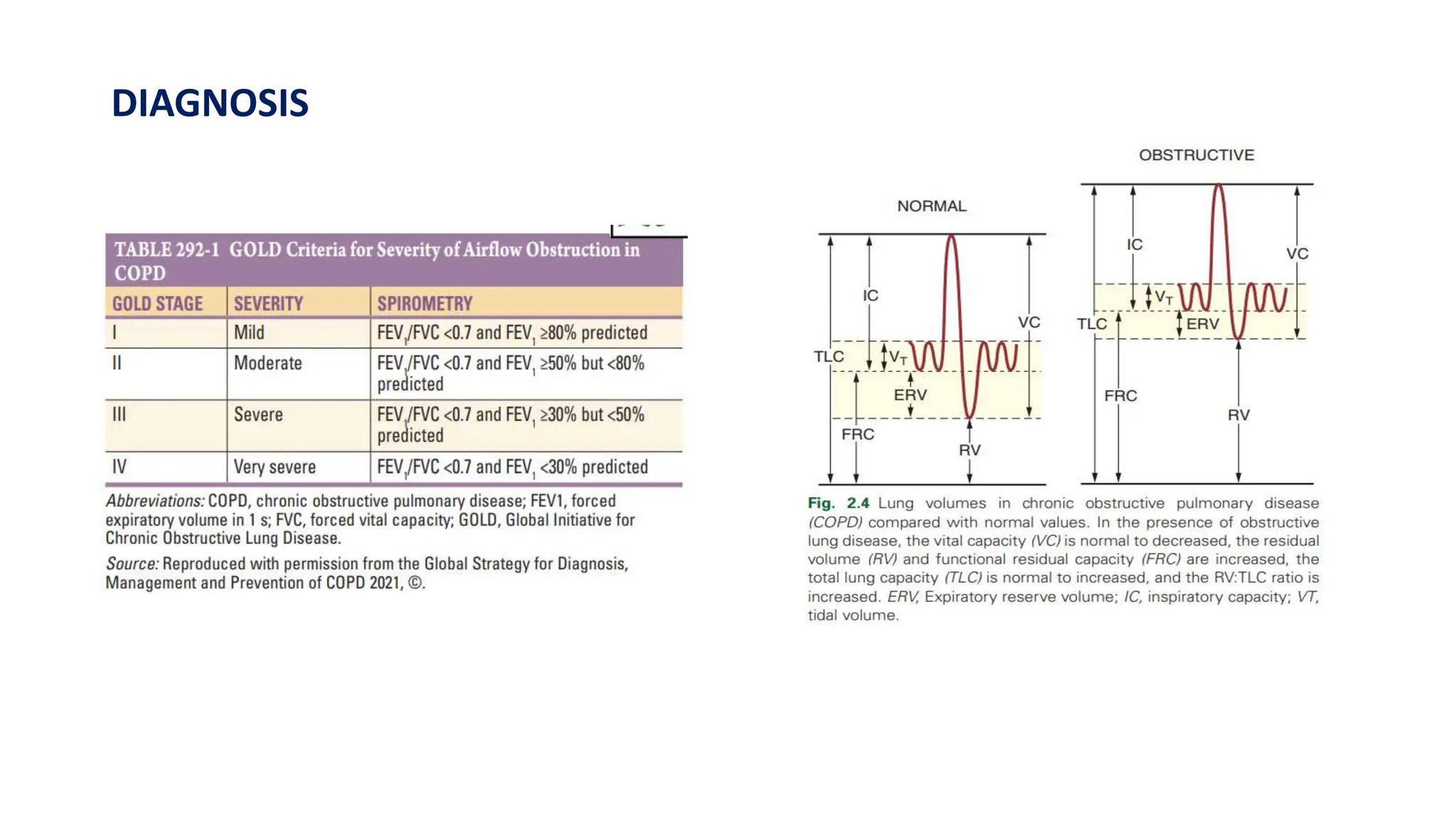
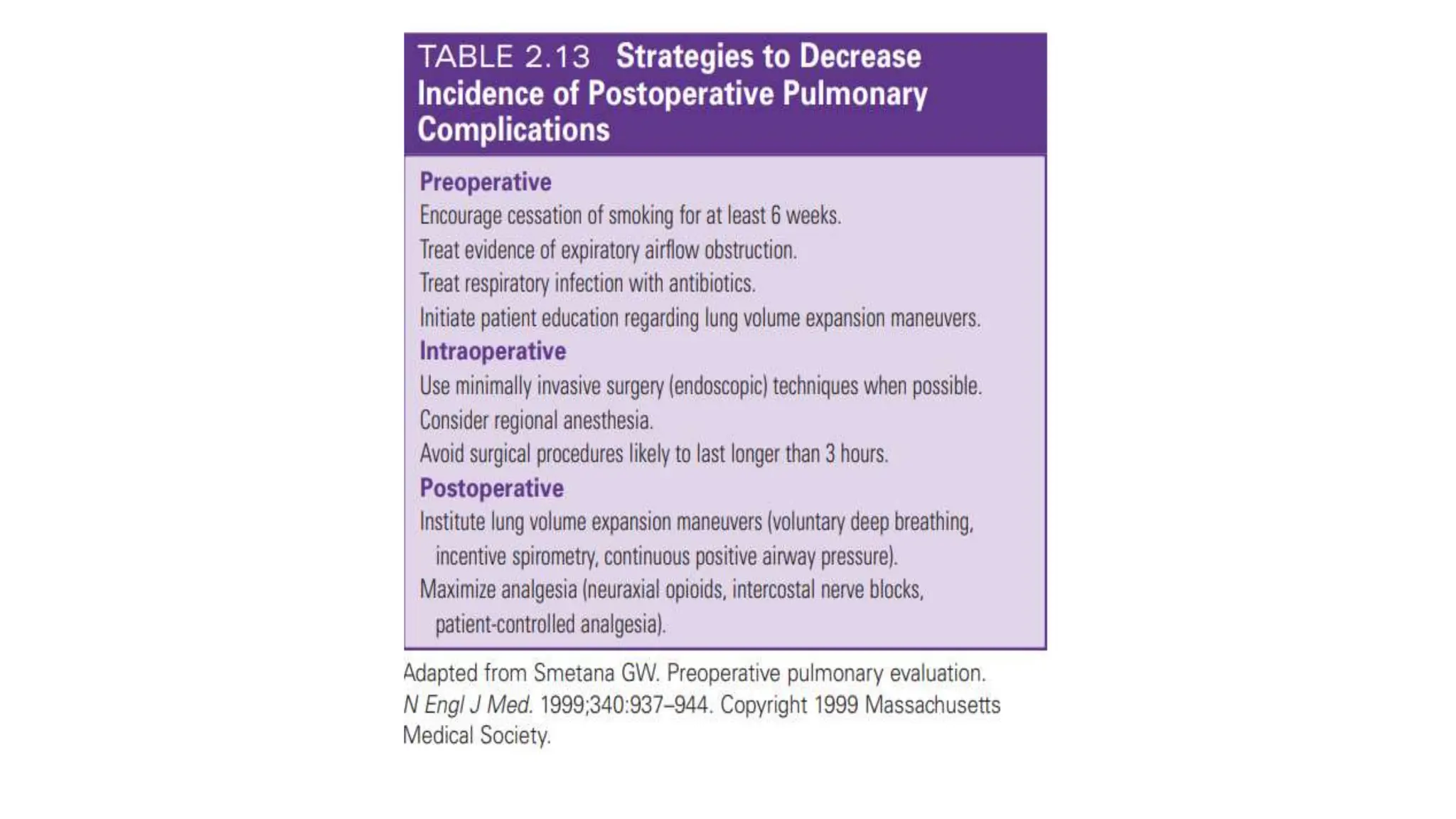
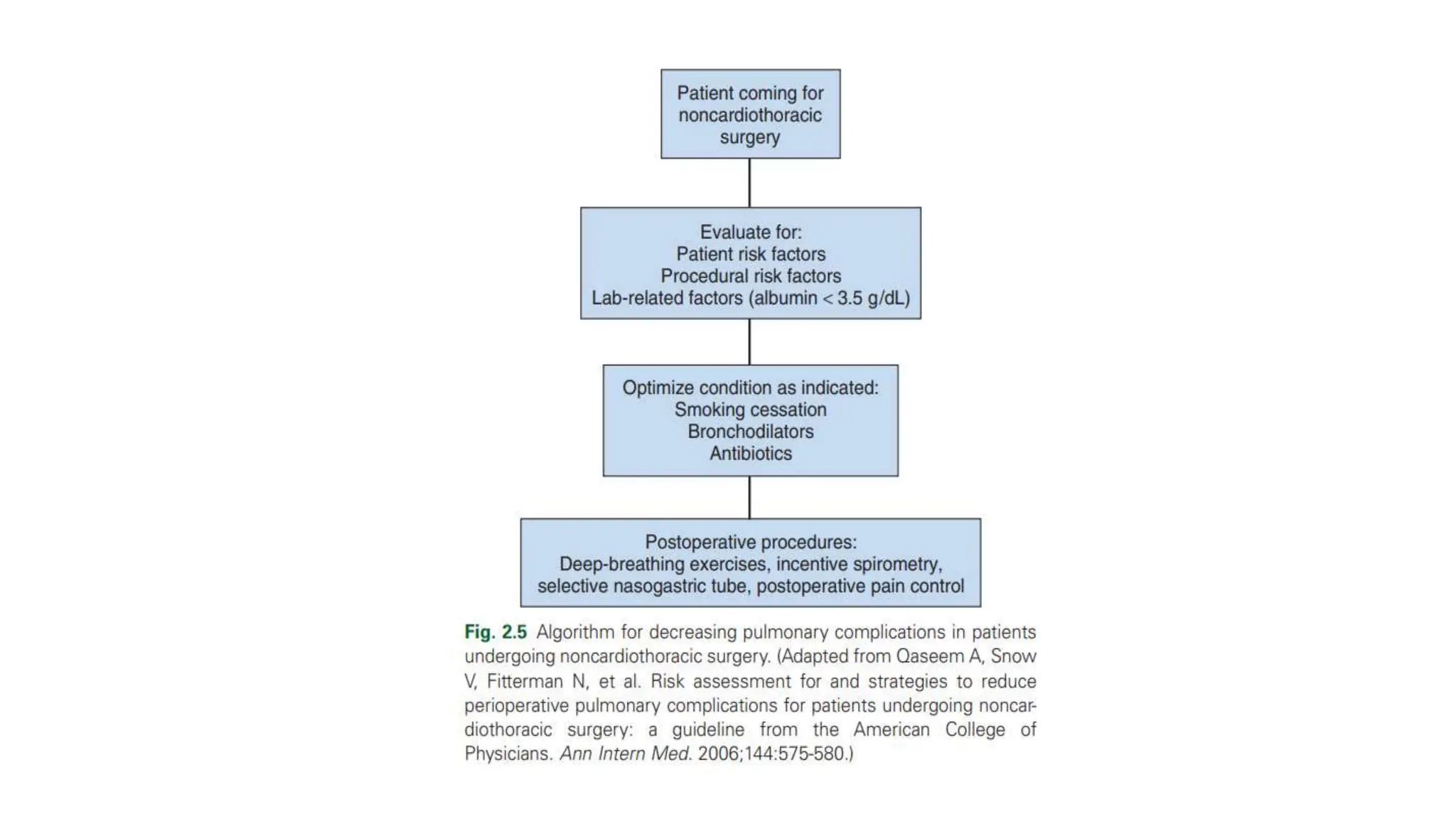
This document provides an overview of asthma and chronic obstructive pulmonary disease (COPD). It discusses the pathophysiology, diagnosis, and treatment of asthma including medications, management of acute exacerbations, and considerations for anesthesia. For COPD, it defines the condition, describes emphysema and chronic bronchitis, guidelines for diagnosis, and treatment including smoking cessation and medications. It also outlines preoperative, intraoperative, and postoperative management strategies for patients with COPD undergoing anesthesia and surgery.