Antibody humanization
•
0 likes•21 views
B cell sorting platform is capa- 1 Antibody Discovery Based on B Cell Sorting Platform ble of the high-throughput screening and generation of fully human monoclonal antibodies (mAbs) or natural mAbs of different species in a short period.
Report
Share
Report
Share
Download to read offline
Recommended
HLA sequencing - the barcode on your cells

Slides that walk you through the basics of why the HLA region in the genome is important, and how to analyze it by next generation sequencing.
HLA tissue typing, HLA matching ,Microcytotoxicity test , Mixed lymphocyte R...

Human leukocyte Antigen Tissue Typing
Recommended
HLA sequencing - the barcode on your cells

Slides that walk you through the basics of why the HLA region in the genome is important, and how to analyze it by next generation sequencing.
HLA tissue typing, HLA matching ,Microcytotoxicity test , Mixed lymphocyte R...

Human leukocyte Antigen Tissue Typing
Heterologous expression lecture

Teaching slide, Heterologous Expression
MIC421 Applied Microbiology, KMUTT ~ 9 February 2016
Plant Chromosomes: European Cytogeneticists outline: Trude Schwarzacher and P...

Plant Chromosomes: European Cytogeneticists outline: Trude Schwarzacher and P...Pat (JS) Heslop-Harrison
An overview of plant molecular cytogenetics. The lecture Trude Schwarzacher presented to the ECA conference Strasbourg in July 2015 is http://www.slideshare.net/PatHeslopHarrison/trude-schwarzacher
Immunology and Serology (transplantation reactions)

Host Vs Graft
Graft vs Host
GVHD
Types of graft
types of graft reaction
Transplantation immunology 

Transplant immunology
Presented by Thansinee Saetae, MD.
December14, 2018
Gene Editing for everyone

CRISPR/Cas9 gene editing is based on a microbial restriction system, that has been harnessed for genome targeting using only a short sequence of RNA as a guide.
Lessons learned from high throughput CRISPR targeting in human cell lines

In just a short period of time CRISPR-Cas9 technology has revolutionized the field of genome editing, and taken the scientific community by storm. Already our understanding of how best to apply this technology has advanced significantly and almost every week new publications appear showcasing its application in basic and translational research.
While CRISPR-Cas9 is applicable across many different cell types, we have found it particularly suited for genome editing in near-haploid human cell lines. This has allowed us to establish a robust pipeline for the inactivation of non-essential genes at unprecedented scale and efficiency.
We have now knocked out over 1500 human genes and have generated a resource that is, to the best of our knowledge, the largest collection of human knockout cell lines available, covering comprehensive subsets of genes clustered by biological pathway (e.g. the autophagy pathway, the JAK/STAT pathway) or by phylogenetic relationship (e.g. kinases, bromodomain-containing proteins).
In this talk we will discuss how, through more than 1500 genome editing experiments, we have started to unravel some of the general principles governing the use of CRISPR-Cas9 in mammalian cells. For example, we have analyzed the impact of variation in the guide RNA sequence on Cas9 cleavage efficiency and characterized the mutational signature arising from CRISPR-Cas9 cleavage.
We will also highlight (with examples) how these learnings are now being applied to introduce other genomic modifications in a high throughput manner, including chromosomal deletions, translocations, point mutations and endogenous gene tags.
Transplantation immunology part 1

Transplantation immunology part 1
Presented by Rapisa Nantanee, MD.
February 5, 2021
Human or Humanized Antibody

The first and most straightforward method is to screen our human antibody libraries. The second method is to immunize premade “humanized” mice and then raise hybridoma cell lines [or to create an immune humanized antibody library] that produce human antibodies.https://www.creative-biolabs.com/Human-or-Humanized-Antibody-Services.html
Therapeutic Antibody Platform

BIOTEM Therapeutic Antibody Platform
> Fully Human Antibody by Phage display
> Ultimate Humanization® Platform (NHP Antibody)
> Humanized Antibody (Humanization by modernized CDR-grafting)
> LLama/ Camel Antibody (VHH or nanobodies)
Contact our technical team: info@biotem.fr
Visit our website: https://www.biotem-antibody.com/
CDR-Grafting Antibody Humanization by BIOTEM

Humanization by CDR-grafting consists in transferring parental (commonly rodent) complementarity determining regions (CDR) into human framework (FR) regions. Parental antibody specificity and affinity are conserved thanks to the preservation of residues implicated in antigen binding. BIOTEM uses a validated and modernized version of the classical CDR-grafting technology.
More Related Content
What's hot
Heterologous expression lecture

Teaching slide, Heterologous Expression
MIC421 Applied Microbiology, KMUTT ~ 9 February 2016
Plant Chromosomes: European Cytogeneticists outline: Trude Schwarzacher and P...

Plant Chromosomes: European Cytogeneticists outline: Trude Schwarzacher and P...Pat (JS) Heslop-Harrison
An overview of plant molecular cytogenetics. The lecture Trude Schwarzacher presented to the ECA conference Strasbourg in July 2015 is http://www.slideshare.net/PatHeslopHarrison/trude-schwarzacher
Immunology and Serology (transplantation reactions)

Host Vs Graft
Graft vs Host
GVHD
Types of graft
types of graft reaction
Transplantation immunology 

Transplant immunology
Presented by Thansinee Saetae, MD.
December14, 2018
Gene Editing for everyone

CRISPR/Cas9 gene editing is based on a microbial restriction system, that has been harnessed for genome targeting using only a short sequence of RNA as a guide.
Lessons learned from high throughput CRISPR targeting in human cell lines

In just a short period of time CRISPR-Cas9 technology has revolutionized the field of genome editing, and taken the scientific community by storm. Already our understanding of how best to apply this technology has advanced significantly and almost every week new publications appear showcasing its application in basic and translational research.
While CRISPR-Cas9 is applicable across many different cell types, we have found it particularly suited for genome editing in near-haploid human cell lines. This has allowed us to establish a robust pipeline for the inactivation of non-essential genes at unprecedented scale and efficiency.
We have now knocked out over 1500 human genes and have generated a resource that is, to the best of our knowledge, the largest collection of human knockout cell lines available, covering comprehensive subsets of genes clustered by biological pathway (e.g. the autophagy pathway, the JAK/STAT pathway) or by phylogenetic relationship (e.g. kinases, bromodomain-containing proteins).
In this talk we will discuss how, through more than 1500 genome editing experiments, we have started to unravel some of the general principles governing the use of CRISPR-Cas9 in mammalian cells. For example, we have analyzed the impact of variation in the guide RNA sequence on Cas9 cleavage efficiency and characterized the mutational signature arising from CRISPR-Cas9 cleavage.
We will also highlight (with examples) how these learnings are now being applied to introduce other genomic modifications in a high throughput manner, including chromosomal deletions, translocations, point mutations and endogenous gene tags.
Transplantation immunology part 1

Transplantation immunology part 1
Presented by Rapisa Nantanee, MD.
February 5, 2021
What's hot (18)
Plant Chromosomes: European Cytogeneticists outline: Trude Schwarzacher and P...

Plant Chromosomes: European Cytogeneticists outline: Trude Schwarzacher and P...
Immunology and Serology (transplantation reactions)

Immunology and Serology (transplantation reactions)
Modulation of MMP and ADAM gene expression in human chondrocytes by IL-1 and OSM

Modulation of MMP and ADAM gene expression in human chondrocytes by IL-1 and OSM
Learning more from direct to-consumer personal genome data

Learning more from direct to-consumer personal genome data
Lessons learned from high throughput CRISPR targeting in human cell lines

Lessons learned from high throughput CRISPR targeting in human cell lines
CRISPR-Cas9 Review: A potential tool for genome editing

CRISPR-Cas9 Review: A potential tool for genome editing
Similar to Antibody humanization
Human or Humanized Antibody

The first and most straightforward method is to screen our human antibody libraries. The second method is to immunize premade “humanized” mice and then raise hybridoma cell lines [or to create an immune humanized antibody library] that produce human antibodies.https://www.creative-biolabs.com/Human-or-Humanized-Antibody-Services.html
Therapeutic Antibody Platform

BIOTEM Therapeutic Antibody Platform
> Fully Human Antibody by Phage display
> Ultimate Humanization® Platform (NHP Antibody)
> Humanized Antibody (Humanization by modernized CDR-grafting)
> LLama/ Camel Antibody (VHH or nanobodies)
Contact our technical team: info@biotem.fr
Visit our website: https://www.biotem-antibody.com/
CDR-Grafting Antibody Humanization by BIOTEM

Humanization by CDR-grafting consists in transferring parental (commonly rodent) complementarity determining regions (CDR) into human framework (FR) regions. Parental antibody specificity and affinity are conserved thanks to the preservation of residues implicated in antigen binding. BIOTEM uses a validated and modernized version of the classical CDR-grafting technology.
RNA-based screening in drug discovery – introducing sgRNA technologies

RNA-based screening in drug discovery
Use of X-MAN™ isogenic cell lines in RNAi screening approaches
Comparison of siRNA and sgRNA screening approaches
The challenges of genome-wide CRISPR-Cas9 knockout (GeCKO) screening
Using CRISPR-Cas9 sgRNA for target identification and patient stratification
Moving from screening hit to target validation
sgRNA screening: not just KOs
Antibody Humanization service creative biolabs

What is antibody humanization? How to humanize the antibody? The definition and humanization process is illustrated by Creative Biolabs in this slide. If you have any questions, welcome to contact us at info@creative-biolabs.com. We will be more than happy to serve you!
Antibody avidity

We use an error-prone PCR integrated DNA-shuffling approach to mutate mainly CDR regions during sub-library construction. If the potential of introducing immunogenic mutations to framework positions is not a concern, we usually use this approach to create mutations at completely random positions across the entire VH and VL fragments. In these cases, the genetic diversity of the sub-library is further increased via passage through our proprietary bacterial mutator strain, CD-affi™.
Introduction to novel antibody sequencing technique

Antibody sequencing is a technique that determines the amino acid sequence of an antibody. Discovering the structures and functions of antibodies in living organism is an important tool in understanding cellular processes, which allow drugs that target specific metabolic pathways to be invented more easily.
Monoclonal Antibodies as drug delivery system

in current scenario apart of traditional route of drug administration monoclonal antibodies can be used which are proved to be more effective in many cases.
Concurrent Determination of ABO RhD Blood Types and the HIV-1 Resistance Mark...

Concurrent Determination of ABO RhD Blood Types and the HIV-1 Resistance Mark...Thermo Fisher Scientific
For optimal outcomes when matching bone marrow donors with their recipients, it is preferable to use bone marrow of identical or compatible blood types. Bone marrow registries thus require high resolution HLA genotyping data to match donor specimens with their recipients. We developed a research assay to aid in these investigations, which utilizes buccal swab DNA from potential donors to determine the ABO and Rh-antigen genotypes. In addition, the assay detects a 32 bp deletion in the CCR5 gene. Homozygous carriers of this deletion are resistant to HIV-1 infection, and thus could be valuable stem cell donors for HIV-infected recipientsReleasing Your AAV Therapy with Confidence: Regulatory Considerations and Key...

Watch the presentation of this webinar here: https://bit.ly/3icKkbZ
Ensuring the safety and quality of your AAV vector is of the utmost importance. Join this webinar for a high-level overview of the regulatory requirements for AAV testing throughout the manufacturing process, as well as a more detailed look at rcAAV and infectious titer assays.
Adeno-associated virus (AAV) vectors possess a number of advantages for use in human therapy including: high titer preparations, low immunogenicity, capacity to infect a wide range of cell types, and replication deficiency. Even with these advantages, there are biosafety concerns to consider when using AAV vectors.
This webinar will discuss key regulatory considerations across the manufacturing process, from the helper/packaging plasmids through to lot release testing. We will highlight critical assays that are required and delve into specifics on replication competent AAV testing and infectious titer determination by TCID50.
In this webinar, you will learn:
• Critical biosafety considerations for AAV vectors based on the latest regulatory guidance
• How replication competent AAV testing fits into your bulk and final release testing package
• The benefits of routine and platform assays over custom assay development
Presented by:
Steven McDade, Senior Technical Specialist, Field Technology Management
Alfonso Lavorgna, Ph.D., Operations Manager, Virology Services
Releasing Your AAV Therapy with Confidence: Regulatory Considerations and Key...

Watch the presentation of this webinar here: https://bit.ly/3icKkbZ
Ensuring the safety and quality of your AAV vector is of the utmost importance. Join this webinar for a high-level overview of the regulatory requirements for AAV testing throughout the manufacturing process, as well as a more detailed look at rcAAV and infectious titer assays.
Adeno-associated virus (AAV) vectors possess a number of advantages for use in human therapy including: high titer preparations, low immunogenicity, capacity to infect a wide range of cell types, and replication deficiency. Even with these advantages, there are biosafety concerns to consider when using AAV vectors.
This webinar will discuss key regulatory considerations across the manufacturing process, from the helper/packaging plasmids through to lot release testing. We will highlight critical assays that are required and delve into specifics on replication competent AAV testing and infectious titer determination by TCID50.
In this webinar, you will learn:
• Critical biosafety considerations for AAV vectors based on the latest regulatory guidance
• How replication competent AAV testing fits into your bulk and final release testing package
• The benefits of routine and platform assays over custom assay development
Presented by:
Steven McDade, Senior Technical Specialist, Field Technology Management
Alfonso Lavorgna, Ph.D., Operations Manager, Virology Services
Sequencing the Human TCRβ Repertoire on the Ion S5TM

understanding of the human immune system, and thereby cancer immunology.
αβT-cells are the primary constituents of human cell-mediated adaptive immunity.
The antigen specificity of each αβT-cell is encoded in the 500-600 bp transcript
encompassing the variable portion of the rearranged TCRα and TCRβ subunits,
which can be read via NGS in a process termed repertoire sequencing. Until now,
the main challenge the field faces is the lack of a technology that can provide a
contiguous read of 600 bp to minimize the complexity of designing bias-prone
primers and informatics challenges of stitching short reads. Here we leverage the
long read capability of Ion 530™ chip to comprehensively sequence all three CDR
domains of the TCRβ chain. The Ion 530™ chip offers greater than 15 M productive
reads, allowing a multiplex of 2-4 samples with sufficient coverage for most repertoire
profiling studies. Initial testing with leukocyte total RNA demonstrates that this
multiplex PCR assay produced repertoires that were much more similar to data
derived from 5’-RACE protocol than the commonly used BIOMED-2 primer set. This
result suggested that the use of long reads minimizes bias by allowing targeting of
less variable regions. To further assess the performance of the assay, we designed a
model system of 30 plasmid controls containing common human T-cell CDR3
sequences. Each plasmid was amplified individually and sequenced to confirm the
detection of a single clonal population. Analytical sensitivity of the assay and
accuracy of the accompanied analysis solution were further evaluated by spiking in
plasmid concentrations from 10 pg to 0.0001 pg (5 million to 50 copies) in a
background of 100 ng cDNA reverse transcribed from leukocyte total RNA. Results
showed the assay offers linearity over 5 orders of magnitude of decreasing input
concentration. In summary, we have demonstrated a NGS workflow for TCRβ
sequencing that offers multiplex flexibility on Ion S5 with sample to answer in less
than 48 hours.
Genomics Technologies

This talk about genomic technologies was given to a Georgetown biostats class in 2010.
Translating Genomes | Personalizing Medicine

Using cell-based assays in the development of efficacious cancer therapies
Similar to Antibody humanization (20)
Biopharma musculoskeletal disorders_draft 6-30-2013 jm edits

Biopharma musculoskeletal disorders_draft 6-30-2013 jm edits
RNA-based screening in drug discovery – introducing sgRNA technologies

RNA-based screening in drug discovery – introducing sgRNA technologies
Introduction to novel antibody sequencing technique

Introduction to novel antibody sequencing technique
Concurrent Determination of ABO RhD Blood Types and the HIV-1 Resistance Mark...

Concurrent Determination of ABO RhD Blood Types and the HIV-1 Resistance Mark...
Commercial production of monoclonal antibodies & mABs produce by rdt

Commercial production of monoclonal antibodies & mABs produce by rdt
Releasing Your AAV Therapy with Confidence: Regulatory Considerations and Key...

Releasing Your AAV Therapy with Confidence: Regulatory Considerations and Key...
Releasing Your AAV Therapy with Confidence: Regulatory Considerations and Key...

Releasing Your AAV Therapy with Confidence: Regulatory Considerations and Key...
Sequencing the Human TCRβ Repertoire on the Ion S5TM

Sequencing the Human TCRβ Repertoire on the Ion S5TM
More from Creative Biolabs
What is oncolytic virus

Recently, the development of molecular biotechnology allows modifications of viral genomes genetically and optimizes the transformation of available viruses with weak pathogenicity. These methods are used to enhance the oncolytic effect and reduce adverse reactions to maximize both efficacy and safety. Indeed, the oncolytic virus can stimulate a pro-inflammatory tumor environment by enhancing antigen recognition and robust immune responses. It overcomes the immune evasiveness and escape of malignant cells to eliminate the tumor cells.
https://www.creative-biolabs.com/oncolytic-virus/definition-of-an-oncolytic-virus.htm
What is oncolytic virus therapy

An oncolytic virus is a form of promising therapeutic tool for the treatment of malignant tumors, which uses viruses to selectively infect and kill tumor cells and further to induce or boost specific antitumor immunity. https://www.creative-biolabs.com/oncolytic-virus/definition-of-an-oncolytic-virus.htm
Reporter-encoding Oncolytic Virus

Oncolytic viruses encoding reporter genes utilized for in vivo molecular imaging are useful to locate the distribution of oncolytic viruses in pre-clinical tests. Optical detection methods mainly include green fluorescent protein (GFP), enhanced GFP (eGFP), discosoma red fluorescent protein (DsRed), and bioluminescence imaging (BLI), which utilizes luciferases. Reporter-encoding oncolytic viruses, including vaccinia virus, adenovirus, herpes simplex virus and vesicular stomatitis virus, allow accurate tracking of gene expression, tumor metastases, viral infection as well as assessment of gene therapy.
https://www.creative-biolabs.com/oncolytic-virus/category-reporter-encoding-oncolytic-virus-293.htm
Pre-made Oncolytic VACV

Vaccinia virus can accommodate more than 30 kb of foreign DNA. Foreign genes can be stably integrated into the viral genome, resulting in efficient and long-term gene expression. The deletion of the viral genes of thymidine kinase (TK) and vaccinia growth factors (VGF) results in enhanced tumor-selectively and antitumor activity, and reduced virus virulence. https://www.creative-biolabs.com/oncolytic-virus/category-pre-made-oncolytic-vaccinia-virus-291.htm
Pre-made Oncolytic Vaccinia Virus

Oncolytic viruses are a class of antitumor agents that selectively kill tumor cells without affecting normal cells. Vaccinia virus (VACV) is a large, enveloped virus that is considered as the most potential live biotherapeutic agent because of its strong oncolytic efficacy and potent antigen presentation capability that can combine well with its natural oncolytic activities for cancer immunotherapy. Many types of modified vaccinia virus have been used for in vitro and in vivo studies, as well as clinical trials.https://www.creative-biolabs.com/oncolytic-virus/category-pre-made-oncolytic-vaccinia-virus-291.htm
Pre-made Oncolytic HSV

Partial deletion of the HSV gene results in superior packaging capacity of >30 kb foreign DNA with low toxicity as an expression vector. Multiple modified purified oncolytic herpes simplex virus (oHSV) products can avoid evading the host immune response and reduce toxicity by gene knock-out, such as ICP0, ICP4, ICP22, ICP27 or ICP47.https://www.creative-biolabs.com/oncolytic-virus/category-pre-made-oncolytic-herpes-simplex-virus-290.htm
Pre-made Oncolytic Herpes Simplex Virus

Oncolytic viruses are using for the treatment of cancer due to the specific antitumor activity in tumor cells. Herpes simplex virus (HSV) is a human neurogenic dsDNA virus that has the characteristic of life-long latent infection of neurons and allows for long-term transgene expression.https://www.creative-biolabs.com/oncolytic-virus/category-pre-made-oncolytic-herpes-simplex-virus-290.htm
Oncolytic Virus Delivery

Oncolytic viruses have the potential to powerfully and selectively kill cancer cells and have shown impressive efficacy in preclinical and clinical settings. However, their potential can be restricted by inefficient delivery into the complex tumor environment. Thus, the efficient delivery of oncolytic viruses remains a significant challenge in the field of oncology, limiting their therapeutic effect. https://www.creative-biolabs.com/oncolytic-virus/approaches-to-delivery-of-oncolytic-viruses.htm
Oncolytic Virus Application

Numerous viruses are being developed pre-clinically and clinically. An investigation of all registered clinical trials in 2017 demonstrates 78 interventional trials regarding OVs. This ability for near-universal therapeutic impact in cancer makes OVs a popular therapeutic tool. Today, both preclinical and early-stage clinical trials are intensively investigating the approach to improve oncolytic virotherapy.
https://www.creative-biolabs.com/oncolytic-virus/applications-of-oncolytic-viruses-in-cancer-treatment.htm
Oncolytic Virus Animal Model

To fully optimize oncolytic virotherapy and provide meaningful mechanistic insight, it is important to have representative animal models of oncolysis in various tumor types. https://www.creative-biolabs.com/oncolytic-virus/animal-models-for-oncolytic-virus-study.htm
Abciximab mechanism of action

Abciximab (also known as abcixifiban or c7E3 Fab), is the Fab fragment of the chimeric human-murine, monoclonal antibody 7E3. It is composed of murine variable regions and human constant regions.https://www.creativebiolabs.net/abciximab-overview.htm
Abagovomab mechanism of action

Abagovomab is a murine monoclonal anti-idiotypic antibody (MW: 165-175 kDa), produced by a mouse hybridoma and generated against OC125, which serves to functionally imitate the human cancer antigen 125 (CA-125). https://www.creativebiolabs.net/abagovomab-overview.htm
Wnt Pathway

Wnt comprises a diverse family of secreted lipid-modified signaling glycoproteins that are 350-400 amino acids in length. Wnt is an acronym in the field of genetics that stands for 'Wingless/Integrated'.https://www.creativebiolabs.net/wnt-signaling-pathway.htm
TNF Pathway

TNF works through two receptors, TNFR1 and TNFR2. TNFR1 is the major signal receptor of TNF-α. TNFR2, which mediates limited biological responses, binds to TNF-α and TNF-β. TNF signaling transduction through TNFR1 and TNFR2 can induce a variety of cellular responses, which depends on many factors, including the metabolic state of the cell and the adaptor proteins present in the cell.https://www.creativebiolabs.net/tnf-signaling-pathway.htm
TLR Pathway

Innate immune receptors, also known as pattern recognition receptors (PRRs), have been identified in the serum, on the cell surface, in endosomes, and in the cytoplasm. Toll-like receptors (TLRs) is one of the particularly important groups of PRRs.https://www.creativebiolabs.net/tlr-signal-pathway.htm
TGF-β Pathway

Transforming growth factor beta (TGF-β) is a cytokine that participates in both physiological and pathological processes.https://www.creativebiolabs.net/tgf-beta-signaling-pathway.htm
TCR Pathway

T-cell receptor (TCR) is a heterodimers composed of α and β peptide chains. TCR is mainly responsible for recognizing the antigens presented by major histocompatibility complex (MHC) molecules on the surface of antigen presenting cells (APC).https://www.creativebiolabs.net/tcr-signal-pathway.htm
RAS Pathway

Ras, which is a low-molecular-weight GDP/GTP-binding guanine triphosphatase, is the prototypical member of the Ras superfamily of proteins. https://www.creativebiolabs.net/ras-signaling-pathway.htm
PPAR Pathway

Peroxisome proliferator-activated receptors (PPARs) are ligand-activated transcription factors, which are responsible for regulating gene expression.https://www.creativebiolabs.net/ppar-signaling-pathway.htm
VPI3K-AKT Pathway

PI3K-Akt signaling pathway is one of the important signal transduction pathways in cells. It is involved in regulating cell metabolism, growth, proliferation, survival, transcription and protein synthesis by affecting the activation of downstream effector molecules. https://www.creativebiolabs.net/pi3k-akt-signaling-pathway.htm
More from Creative Biolabs (20)
Recently uploaded
Hemodialysis: Chapter 3, Dialysis Water Unit - Dr.Gawad

- Video recording of this lecture in English language: https://youtu.be/lK81BzxMqdo
- Video recording of this lecture in Arabic language: https://youtu.be/Ve4P0COk9OI
- Link to download the book free: https://nephrotube.blogspot.com/p/nephrotube-nephrology-books.html
- Link to NephroTube website: www.NephroTube.com
- Link to NephroTube social media accounts: https://nephrotube.blogspot.com/p/join-nephrotube-on-social-media.html
Pharma Pcd Franchise in Jharkhand - Yodley Lifesciences

Join Monopoly Pharma PCD Franchise in Jharkhand :Ranchi, Jamshedpur, Dhanbad, Bokaro Steel City, Hazaribagh, Deoghar, Giridih, etc.
Are There Any Natural Remedies To Treat Syphilis.pdf

Explore natural remedies for syphilis treatment in Singapore. Discover alternative therapies, herbal remedies, and lifestyle changes that may complement conventional treatments. Learn about holistic approaches to managing syphilis symptoms and supporting overall health.
Novas diretrizes da OMS para os cuidados perinatais de mais qualidade

Novas diretrizes da OMS para os cuidados perinatais de mais qualidadeProf. Marcus Renato de Carvalho
Recomendações da OMS sobre cuidados maternos e neonatais para uma experiência pós-natal positiva.
Em consonância com os ODS – Objetivos do Desenvolvimento Sustentável e a Estratégia Global para a Saúde das Mulheres, Crianças e Adolescentes, e aplicando uma abordagem baseada nos direitos humanos, os esforços de cuidados pós-natais devem expandir-se para além da cobertura e da simples sobrevivência, de modo a incluir cuidados de qualidade.
Estas diretrizes visam melhorar a qualidade dos cuidados pós-natais essenciais e de rotina prestados às mulheres e aos recém-nascidos, com o objetivo final de melhorar a saúde e o bem-estar materno e neonatal.
Uma “experiência pós-natal positiva” é um resultado importante para todas as mulheres que dão à luz e para os seus recém-nascidos, estabelecendo as bases para a melhoria da saúde e do bem-estar a curto e longo prazo. Uma experiência pós-natal positiva é definida como aquela em que as mulheres, pessoas que gestam, os recém-nascidos, os casais, os pais, os cuidadores e as famílias recebem informação consistente, garantia e apoio de profissionais de saúde motivados; e onde um sistema de saúde flexível e com recursos reconheça as necessidades das mulheres e dos bebês e respeite o seu contexto cultural.
Estas diretrizes consolidadas apresentam algumas recomendações novas e já bem fundamentadas sobre cuidados pós-natais de rotina para mulheres e neonatos que recebem cuidados no pós-parto em unidades de saúde ou na comunidade, independentemente dos recursos disponíveis.
É fornecido um conjunto abrangente de recomendações para cuidados durante o período puerperal, com ênfase nos cuidados essenciais que todas as mulheres e recém-nascidos devem receber, e com a devida atenção à qualidade dos cuidados; isto é, a entrega e a experiência do cuidado recebido. Estas diretrizes atualizam e ampliam as recomendações da OMS de 2014 sobre cuidados pós-natais da mãe e do recém-nascido e complementam as atuais diretrizes da OMS sobre a gestão de complicações pós-natais.
O estabelecimento da amamentação e o manejo das principais intercorrências é contemplada.
Recomendamos muito.
Vamos discutir essas recomendações no nosso curso de pós-graduação em Aleitamento no Instituto Ciclos.
Esta publicação só está disponível em inglês até o momento.
Prof. Marcus Renato de Carvalho
www.agostodourado.com
ARTHROLOGY PPT NCISM SYLLABUS AYURVEDA STUDENTS

PPT RELATED TO ARTHROLOGY ACCORDING TO NCISM AYURVEDA
micro teaching on communication m.sc nursing.pdf

Microteaching is a unique model of practice teaching. It is a viable instrument for the. desired change in the teaching behavior or the behavior potential which, in specified types of real. classroom situations, tends to facilitate the achievement of specified types of objectives.
Light House Retreats: Plant Medicine Retreat Europe

Our aim is to organise conscious gatherings and retreats for open and inquisitive minds and souls, with and without the assistance of sacred plants.
NVBDCP.pptx Nation vector borne disease control program

NVBDCP was launched in 2003-2004 . Vector-Borne Disease: Disease that results from an infection transmitted to humans and other animals by blood-feeding arthropods, such as mosquitoes, ticks, and fleas. Examples of vector-borne diseases include Dengue fever, West Nile Virus, Lyme disease, and malaria.
TEST BANK for Operations Management, 14th Edition by William J. Stevenson, Ve...

TEST BANK for Operations Management, 14th Edition by William J. Stevenson, Verified Chapters 1 - 19, Complete Newest Version.pdf
TEST BANK for Operations Management, 14th Edition by William J. Stevenson, Verified Chapters 1 - 19, Complete Newest Version.pdf
263778731218 Abortion Clinic /Pills In Harare ,

263778731218 Abortion Clinic /Pills In Harare ,ABORTION WOMEN’S CLINIC +27730423979 IN women clinic we believe that every woman should be able to make choices in her pregnancy. Our job is to provide compassionate care, safety,affordable and confidential services. That’s why we have won the trust from all generations of women all over the world. we use non surgical method(Abortion pills) to terminate…Dr.LISA +27730423979women Clinic is committed to providing the highest quality of obstetrical and gynecological care to women of all ages. Our dedicated staff aim to treat each patient and her health concerns with compassion and respect.Our dedicated group ABORTION WOMEN’S CLINIC +27730423979 IN women clinic we believe that every woman should be able to make choices in her pregnancy. Our job is to provide compassionate care, safety,affordable and confidential services. That’s why we have won the trust from all generations of women all over the world. we use non surgical method(Abortion pills) to terminate…Dr.LISA +27730423979women Clinic is committed to providing the highest quality of obstetrical and gynecological care to women of all ages. Our dedicated staff aim to treat each patient and her health concerns with compassion and respect.Our dedicated group of receptionists, nurses, and physicians have worked together as a teamof receptionists, nurses, and physicians have worked together as a team wwww.lisywomensclinic.co.za/
Alcohol_Dr. Jeenal Mistry MD Pharmacology.pdf

Ethanol (CH3CH2OH), or beverage alcohol, is a two-carbon alcohol
that is rapidly distributed in the body and brain. Ethanol alters many
neurochemical systems and has rewarding and addictive properties. It
is the oldest recreational drug and likely contributes to more morbidity,
mortality, and public health costs than all illicit drugs combined. The
5th edition of the Diagnostic and Statistical Manual of Mental Disorders
(DSM-5) integrates alcohol abuse and alcohol dependence into a single
disorder called alcohol use disorder (AUD), with mild, moderate,
and severe subclassifications (American Psychiatric Association, 2013).
In the DSM-5, all types of substance abuse and dependence have been
combined into a single substance use disorder (SUD) on a continuum
from mild to severe. A diagnosis of AUD requires that at least two of
the 11 DSM-5 behaviors be present within a 12-month period (mild
AUD: 2–3 criteria; moderate AUD: 4–5 criteria; severe AUD: 6–11 criteria).
The four main behavioral effects of AUD are impaired control over
drinking, negative social consequences, risky use, and altered physiological
effects (tolerance, withdrawal). This chapter presents an overview
of the prevalence and harmful consequences of AUD in the U.S.,
the systemic nature of the disease, neurocircuitry and stages of AUD,
comorbidities, fetal alcohol spectrum disorders, genetic risk factors, and
pharmacotherapies for AUD.
Non-respiratory Functions of the Lungs.pdf

These simplified slides by Dr. Sidra Arshad present an overview of the non-respiratory functions of the respiratory tract.
Learning objectives:
1. Enlist the non-respiratory functions of the respiratory tract
2. Briefly explain how these functions are carried out
3. Discuss the significance of dead space
4. Differentiate between minute ventilation and alveolar ventilation
5. Describe the cough and sneeze reflexes
Study Resources:
1. Chapter 39, Guyton and Hall Textbook of Medical Physiology, 14th edition
2. Chapter 34, Ganong’s Review of Medical Physiology, 26th edition
3. Chapter 17, Human Physiology by Lauralee Sherwood, 9th edition
4. Non-respiratory functions of the lungs https://academic.oup.com/bjaed/article/13/3/98/278874
HOT NEW PRODUCT! BIG SALES FAST SHIPPING NOW FROM CHINA!! EU KU DB BK substit...

Contact us if you are interested:
Email / Skype : kefaya1771@gmail.com
Threema: PXHY5PDH
New BATCH Ku !!! MUCH IN DEMAND FAST SALE EVERY BATCH HAPPY GOOD EFFECT BIG BATCH !
Contact me on Threema or skype to start big business!!
Hot-sale products:
NEW HOT EUTYLONE WHITE CRYSTAL!!
5cl-adba precursor (semi finished )
5cl-adba raw materials
ADBB precursor (semi finished )
ADBB raw materials
APVP powder
5fadb/4f-adb
Jwh018 / Jwh210
Eutylone crystal
Protonitazene (hydrochloride) CAS: 119276-01-6
Flubrotizolam CAS: 57801-95-3
Metonitazene CAS: 14680-51-4
Payment terms: Western Union,MoneyGram,Bitcoin or USDT.
Deliver Time: Usually 7-15days
Shipping method: FedEx, TNT, DHL,UPS etc.Our deliveries are 100% safe, fast, reliable and discreet.
Samples will be sent for your evaluation!If you are interested in, please contact me, let's talk details.
We specializes in exporting high quality Research chemical, medical intermediate, Pharmaceutical chemicals and so on. Products are exported to USA, Canada, France, Korea, Japan,Russia, Southeast Asia and other countries.
Knee anatomy and clinical tests 2024.pdf

This includes all relevant anatomy and clinical tests compiled from standard textbooks, Campbell,netter etc..It is comprehensive and best suited for orthopaedicians and orthopaedic residents.
Recently uploaded (20)
Hemodialysis: Chapter 3, Dialysis Water Unit - Dr.Gawad

Hemodialysis: Chapter 3, Dialysis Water Unit - Dr.Gawad
Pharma Pcd Franchise in Jharkhand - Yodley Lifesciences

Pharma Pcd Franchise in Jharkhand - Yodley Lifesciences
Are There Any Natural Remedies To Treat Syphilis.pdf

Are There Any Natural Remedies To Treat Syphilis.pdf
Novas diretrizes da OMS para os cuidados perinatais de mais qualidade

Novas diretrizes da OMS para os cuidados perinatais de mais qualidade
Light House Retreats: Plant Medicine Retreat Europe

Light House Retreats: Plant Medicine Retreat Europe
NVBDCP.pptx Nation vector borne disease control program

NVBDCP.pptx Nation vector borne disease control program
TEST BANK for Operations Management, 14th Edition by William J. Stevenson, Ve...

TEST BANK for Operations Management, 14th Edition by William J. Stevenson, Ve...
Maxilla, Mandible & Hyoid Bone & Clinical Correlations by Dr. RIG.pptx

Maxilla, Mandible & Hyoid Bone & Clinical Correlations by Dr. RIG.pptx
Triangles of Neck and Clinical Correlation by Dr. RIG.pptx

Triangles of Neck and Clinical Correlation by Dr. RIG.pptx
HOT NEW PRODUCT! BIG SALES FAST SHIPPING NOW FROM CHINA!! EU KU DB BK substit...

HOT NEW PRODUCT! BIG SALES FAST SHIPPING NOW FROM CHINA!! EU KU DB BK substit...
Antibody humanization
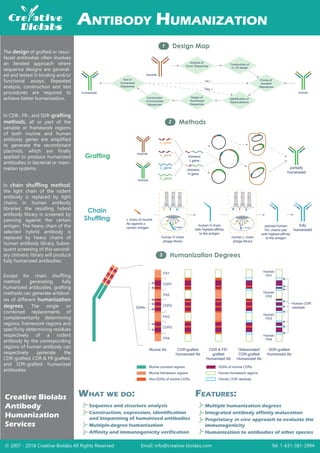
- 1. Design Map1 Methods2 © 2007 - 2018 Creative-Biolabs All Rights Reserved Email: info@creative-biolabs.com Tel: 1-631-381-2994 Grafting Chain Shuffling ANTIBODY HUMANIZATION Creative Biolabs Antibody Humanization Services Multiple humanization degrees Integrated antibody affinity maturation Proprietary approach to evaluate the immunogenicity Humanization to antibodies of other species Sequence and structure analysis Construction, expression, identification and biopanning of humanized antibodies Multiple-degree humanization Affinity and immunogenicity verification FEATURES:WHAT WE DO: VL gene The design of grafted or resur- faced antibodies often involves an iterated approach where sequence designs are generat- ed and tested in binding and/or functional assays. Repeated analysis, construction and test procedures are required to achieve better humanization. In CDR-, FR-, and SDR-grafting methods, all or part of the variable or framework regions of both murine and human antibody genes are amplified to generate the recombinant plasmids, which are finally applied to produce humanized antibodies in bacterial or mam- malian systems. Except for chain shuffling method generating fully humanized antibodies, grafting methods can generate antibod- ies of different humanization degrees. The single or combined replacements of complementarity determining regions, framework regions and specificity determining residues respectively of a rodent antibody by the corresponding regions of human antibody can respectively generate the CDR-grafted, CDR & FR grafted, and SDR-grafted humanized antibodies. murine human VH gene CL gene CH gene chimeric L gene chimeric H gene L chain of murine Ab against a certain antigen human H chain phage library G3P H chain human L chain phage library L chain G3P human H chain with highest-affinity to the antigen panned human H/L chains pair with highest-affinity to the antigen fully humanized partially humanized Humanization Degrees3 murine Analysis of Donor Sequences Construction of Fv 3D Model Choice of Acceptor Sequences Identification of Backmutations Design of Humanized Sequences Construction of Humanized Sequences Test of Humanized Sequences humanized human No ? Yes ? SDRs CDR1 CDR2 CDR3 FR1 FR2 FR3 FR4 Human CDR residues Human FR1 Human FR2 Human FR3 Human FR4 Murine Ab CDR-grafted Humanized Ab CDR & FR- grafted Humanized Ab “Abbreviated” CDR-grafted Humanized Ab SDR-grafted Humanized Ab Murine constant regions Murine framework regions Non-SDRs of murine CDRs SDRs of murine CDRs Human framework regions Human CDR residues In chain shuffling method, the light chain of the rodent antibody is replaced by light chains in human antibody libraries; the resulting hybrid antibody library is screened by panning against the certain antigen. The heavy chain of the selected hybrid antibody is replaced by heavy chains of human antibody library. Subse- quent screening of this second- ary chimeric library will produce fully humanized antibodies. in vivo
