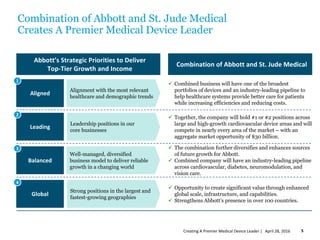
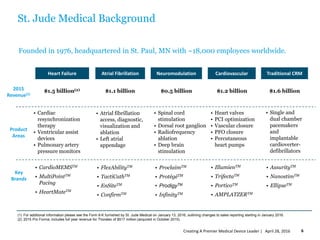
Abbott intends to acquire St. Jude Medical to create a premier medical device leader. The transaction values St. Jude Medical at $25 billion. Abbott will pay St. Jude Medical shareholders $46.75 in cash and 0.8708 Abbott shares per St. Jude Medical share. The combination is expected to generate $500 million in annual synergies by 2020 and be accretive to Abbott's earnings per share in the first full year. The transaction is subject to shareholder and regulatory approvals and is expected to close in the fourth quarter of 2016.