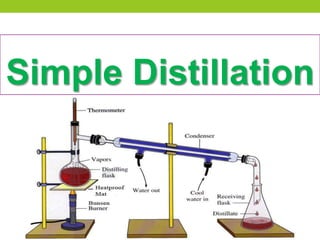
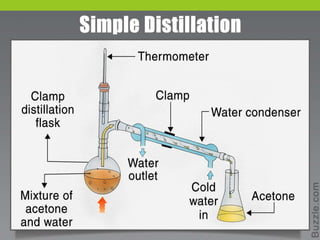
Distillation is a process that separates liquids with different boiling points by heating a liquid mixture and collecting the vapor of the more volatile components. It works based on the principle that the vapor pressure of a component in a mixture depends on its mole fraction. Distillation is used to purify liquids and separate components by taking advantage of differences in boiling points, with common applications including separating crude oil, purifying water, and producing distilled alcoholic beverages. There are different types of distillation that vary based on factors like the temperature range, differences in boiling points, and use of vacuum or steam.