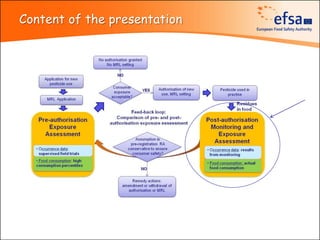
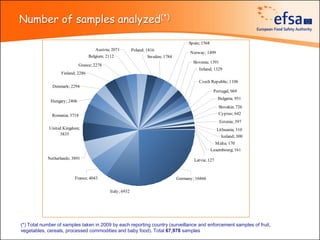
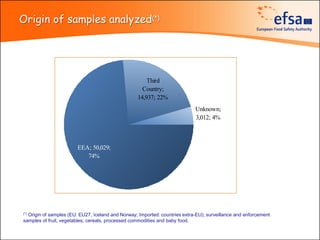
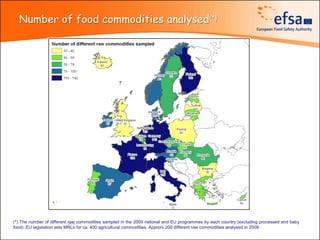
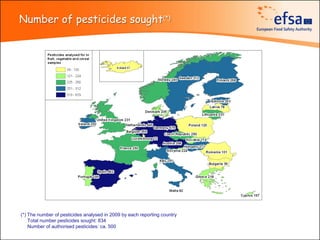
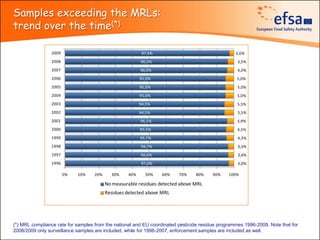
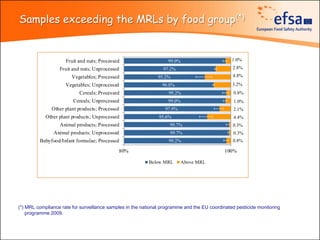
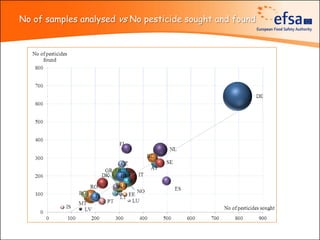
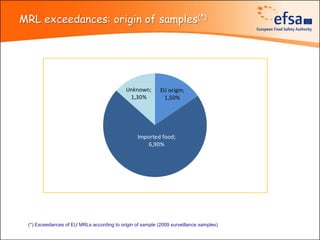
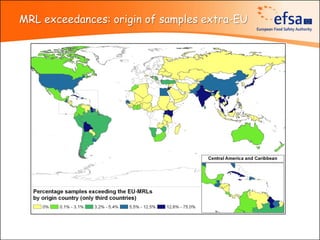
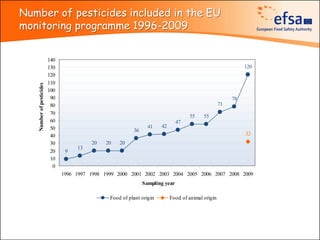
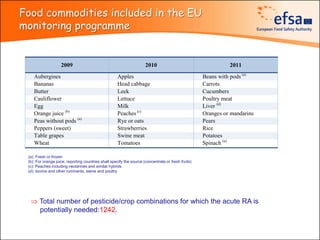
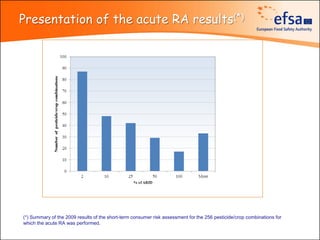
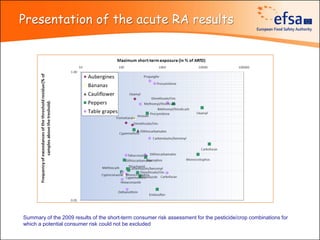
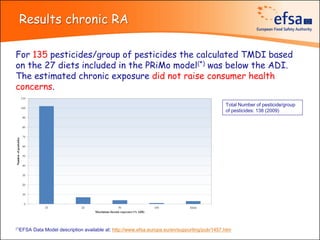
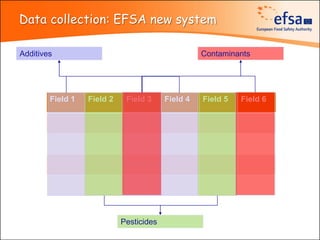
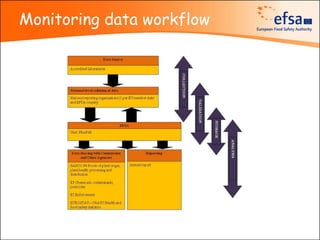
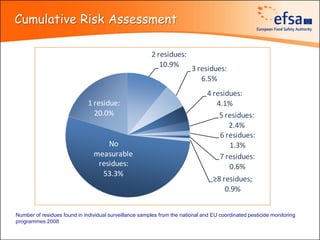
The document summarizes monitoring programs and findings related to pesticide residues in food in Europe. It discusses the legal framework for monitoring in EU countries, EFSA's annual reports on pesticide residues, and a new EU data collection system. The main findings from EFSA's 2009 annual report are that over 97% of samples did not exceed maximum residue levels, with exceedances more common in imported and unprocessed foods. EFSA also conducts cumulative risk assessments of pesticide residues.