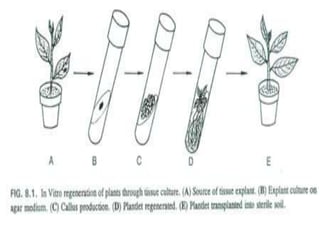
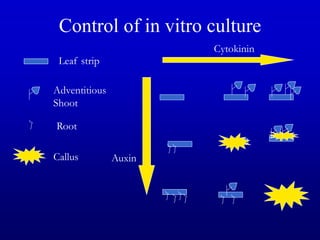
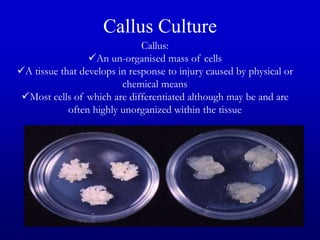
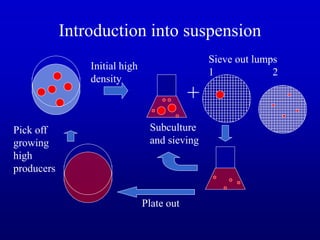
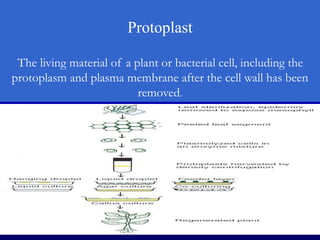
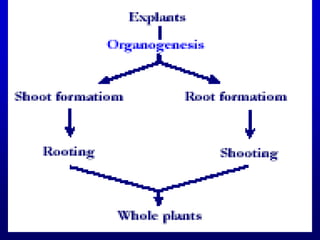
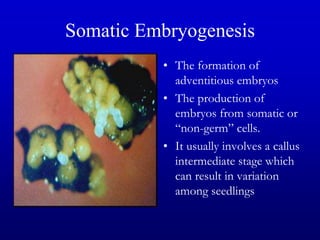
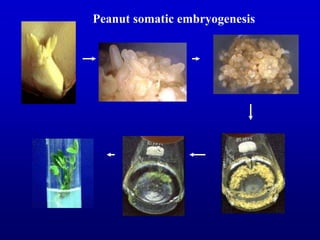
Plant tissue culture techniques allow for the growth and development of plant cells, tissues, organs, or protoplasts in sterile conditions on nutrient media. There are several types of in vitro culture including callus culture, organ culture, and somatic embryogenesis. Plant cells have the properties of totipotency, dedifferentiation, and competency which allow regeneration of whole plants from single cells. Tissue culture is important for applications such as crop improvement, mass propagation, and germplasm conservation.