Report
Share
Download to read offline
Recommended
Recommended
More Related Content
What's hot
What's hot (20)
Viewers also liked
Viewers also liked (14)
The handy science answer book (the handy answer book series)

The handy science answer book (the handy answer book series)
Similar to Potential energy diagram worksheet 2
Similar to Potential energy diagram worksheet 2 (10)
Chemical dynamics, intro,tst, lindemann theory by dr.y. s. thakare

Chemical dynamics, intro,tst, lindemann theory by dr.y. s. thakare
More from sathiakumaran
More from sathiakumaran (19)
Fun way to learn hybridisation from friends mistakes !

Fun way to learn hybridisation from friends mistakes !
Sample quiz paper set 6 (differential rate equation)

Sample quiz paper set 6 (differential rate equation)
Recently uploaded
https://app.box.com/s/x7vf0j7xaxl2hlczxm3ny497y4yto33i80 ĐỀ THI THỬ TUYỂN SINH TIẾNG ANH VÀO 10 SỞ GD – ĐT THÀNH PHỐ HỒ CHÍ MINH NĂ...

80 ĐỀ THI THỬ TUYỂN SINH TIẾNG ANH VÀO 10 SỞ GD – ĐT THÀNH PHỐ HỒ CHÍ MINH NĂ...Nguyen Thanh Tu Collection
https://app.box.com/s/7hlvjxjalkrik7fb082xx3jk7xd7liz3TỔNG ÔN TẬP THI VÀO LỚP 10 MÔN TIẾNG ANH NĂM HỌC 2023 - 2024 CÓ ĐÁP ÁN (NGỮ Â...

TỔNG ÔN TẬP THI VÀO LỚP 10 MÔN TIẾNG ANH NĂM HỌC 2023 - 2024 CÓ ĐÁP ÁN (NGỮ Â...Nguyen Thanh Tu Collection
Recently uploaded (20)
Jual Obat Aborsi Hongkong ( Asli No.1 ) 085657271886 Obat Penggugur Kandungan...

Jual Obat Aborsi Hongkong ( Asli No.1 ) 085657271886 Obat Penggugur Kandungan...
Micro-Scholarship, What it is, How can it help me.pdf

Micro-Scholarship, What it is, How can it help me.pdf
Unit 3 Emotional Intelligence and Spiritual Intelligence.pdf

Unit 3 Emotional Intelligence and Spiritual Intelligence.pdf
NO1 Top Black Magic Specialist In Lahore Black magic In Pakistan Kala Ilam Ex...

NO1 Top Black Magic Specialist In Lahore Black magic In Pakistan Kala Ilam Ex...
80 ĐỀ THI THỬ TUYỂN SINH TIẾNG ANH VÀO 10 SỞ GD – ĐT THÀNH PHỐ HỒ CHÍ MINH NĂ...

80 ĐỀ THI THỬ TUYỂN SINH TIẾNG ANH VÀO 10 SỞ GD – ĐT THÀNH PHỐ HỒ CHÍ MINH NĂ...
TỔNG ÔN TẬP THI VÀO LỚP 10 MÔN TIẾNG ANH NĂM HỌC 2023 - 2024 CÓ ĐÁP ÁN (NGỮ Â...

TỔNG ÔN TẬP THI VÀO LỚP 10 MÔN TIẾNG ANH NĂM HỌC 2023 - 2024 CÓ ĐÁP ÁN (NGỮ Â...
Exploring_the_Narrative_Style_of_Amitav_Ghoshs_Gun_Island.pptx

Exploring_the_Narrative_Style_of_Amitav_Ghoshs_Gun_Island.pptx
Basic Civil Engineering first year Notes- Chapter 4 Building.pptx

Basic Civil Engineering first year Notes- Chapter 4 Building.pptx
Salient Features of India constitution especially power and functions

Salient Features of India constitution especially power and functions
Python Notes for mca i year students osmania university.docx

Python Notes for mca i year students osmania university.docx
Potential energy diagram worksheet 2
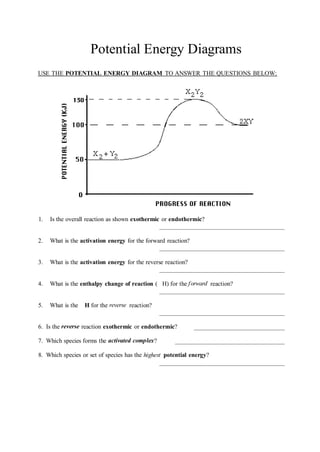
- 1. Potential Energy Diagrams USE THE POTENTIAL ENERGY DIAGRAM TO ANSWER THE QUESTIONS BELOW: 1. Is the overall reaction as shown exothermic or endothermic? 2. What is the activation energy for the forward reaction? 3. What is the activation energy for the reverse reaction? 4. What is the enthalpy change of reaction ( H) for the forward reaction? 5. What is the H for the reverse reaction? 6. Is the reverse reaction exothermic or endothermic? 7. Which species forms the activated complex? 8. Which species or set of species has the highest potential energy?
- 2. 9. Which species or set of species has the highest kinetic energy? 10. Which species or set of species has the weakest bonds? 11. Which species or set of species has the strongest bonds? 12. What is H for the reaction: X2Y2 X2 + Y2 ? 13. Which do you think would be faster, the forward reaction or the reverse reaction? Explain. _ 14. Which species or set of species has the lowest kinetic energy? 15. Show the H, the Activation Energy for the forward reaction and the Activation Energy for the reverse reaction on the graph above. 16. As reactant particles approach each other before a collision, the Potential Energy goes _, while the Kinetic Energy goes _. 17. As particles of newly formed products move away from one another, the Potential Energy goes , while the Kinetic Energy goes 18. As reactant molecules approach each other, they exert forces on each other. Thus, as they move together, their speed and their Potential Energy 19. State the meaning of Activated Complex.
- 3. ABC AB + C A + BC 20. Use the following Potential Energy Diagram to answer the questions below: 100 80 60 40 20 0 Progress of Reaction a) Determine the Activation Energy for the forward reaction... kJ b) Determine the Activation Energy for the reverse reaction.... _kJ c) What is the Enthalpy Change ( H) for the forward reaction?.. _kJ d) What is the Enthalpy Change ( H) for the reverse reaction?.. _kJ e) The forward reaction is thermic. f) The reverse reaction is thermic. g) Which species or set of species forms the Activated Complex? h) Which bond is stronger, A--B or B--C?_ _. Give a reason for your answer. i) Particles from which species or set of species is moving the fastest? State how you arrived at your answer. _
- 4. j) Particles from which species or set of species is moving most slowly? State how you arrived at your answer. k) The compound "AB" is a gas and the element "C" is a solid. What effect would grinding"C" into a fine powder have on the graph shown here? 21. State the meaning of Activation Energy . 22. What two requirements must be met before a collision between two reactant particles is effective? 1. 2. 23. Describe what happens to two reactant particles which collide with less energy than the Activation Energy . 24. Burning coal (Carbon) is a highly exothermic reaction. However coal, in contact with air at room temperature has such a slow reaction that it is not noticeable. Explain these two facts with the help of a Potential Energy Diagram. Potential Energy (KJ) Progress of Reaction