Service Functions Standards Core Engine HPC
•
1 like•117 views
The document outlines the key functions and components of a clinical research data management system, including standards management, a high performance computing core engine, data storage systems, analysis workspaces, analytics and visualization tools, and collaboration features to support study design, publication, and regulatory activities across a network of sponsors, affiliates, CROs, and research partners. It also references the management of data quality, permissions, resources, and dictionaries within the system.
Report
Share
Report
Share
Download to read offline
Recommended
More Related Content
Similar to Service Functions Standards Core Engine HPC
Similar to Service Functions Standards Core Engine HPC (20)
Advanced Authorization for SAP Global Deployments Part III of III

Advanced Authorization for SAP Global Deployments Part III of III
Advanced Authorization for SAP Global Deployments Part I of III

Advanced Authorization for SAP Global Deployments Part I of III
BSA425 v1.3Project Plan DraftBSA425 v1.3Page 11 of 11P

BSA425 v1.3Project Plan DraftBSA425 v1.3Page 11 of 11P
DataOps - Big Data and AI World London - March 2020 - Harvinder Atwal

DataOps - Big Data and AI World London - March 2020 - Harvinder Atwal
Analyti x mapping manager product overview presentation

Analyti x mapping manager product overview presentation
Empowering Business & IT Teams: Modern Data Catalog Requirements

Empowering Business & IT Teams: Modern Data Catalog Requirements
BSA425 v1.3Supporting Research ReportBSA425 v1.3Page 2 of 

BSA425 v1.3Supporting Research ReportBSA425 v1.3Page 2 of
More from Sandeep Bhat
More from Sandeep Bhat (13)
Observations, Issues, Benefits: Mobility in Life Sciences

Observations, Issues, Benefits: Mobility in Life Sciences
Strategy and Approach for Mobility in Life Sciences

Strategy and Approach for Mobility in Life Sciences
Conceptual Framework for a Clinical Supply Chain solution stack

Conceptual Framework for a Clinical Supply Chain solution stack
A Vision for Personalized Medicine: Collaboration, Enablement, Technology

A Vision for Personalized Medicine: Collaboration, Enablement, Technology
Engage Patients with Innovative Global Digital Patient Platform

Engage Patients with Innovative Global Digital Patient Platform
Nine Dimensions Of Commercial Excellence (Sandeep Bhat)

Nine Dimensions Of Commercial Excellence (Sandeep Bhat)
Social Media Considerations In Pharmacovigilance Visiongain 20110317 (Sande...

Social Media Considerations In Pharmacovigilance Visiongain 20110317 (Sande...
Recently uploaded
Recently uploaded (20)
Presentation on how to chat with PDF using ChatGPT code interpreter

Presentation on how to chat with PDF using ChatGPT code interpreter
Unleash Your Potential - Namagunga Girls Coding Club

Unleash Your Potential - Namagunga Girls Coding Club
Designing IA for AI - Information Architecture Conference 2024

Designing IA for AI - Information Architecture Conference 2024
#StandardsGoals for 2024: What’s new for BISAC - Tech Forum 2024

#StandardsGoals for 2024: What’s new for BISAC - Tech Forum 2024
Automating Business Process via MuleSoft Composer | Bangalore MuleSoft Meetup...

Automating Business Process via MuleSoft Composer | Bangalore MuleSoft Meetup...
The Codex of Business Writing Software for Real-World Solutions 2.pptx

The Codex of Business Writing Software for Real-World Solutions 2.pptx
Artificial intelligence in the post-deep learning era

Artificial intelligence in the post-deep learning era
08448380779 Call Girls In Friends Colony Women Seeking Men

08448380779 Call Girls In Friends Colony Women Seeking Men
08448380779 Call Girls In Diplomatic Enclave Women Seeking Men

08448380779 Call Girls In Diplomatic Enclave Women Seeking Men
Breaking the Kubernetes Kill Chain: Host Path Mount

Breaking the Kubernetes Kill Chain: Host Path Mount
Scanning the Internet for External Cloud Exposures via SSL Certs

Scanning the Internet for External Cloud Exposures via SSL Certs
FULL ENJOY 🔝 8264348440 🔝 Call Girls in Diplomatic Enclave | Delhi

FULL ENJOY 🔝 8264348440 🔝 Call Girls in Diplomatic Enclave | Delhi
Vulnerability_Management_GRC_by Sohang Sengupta.pptx

Vulnerability_Management_GRC_by Sohang Sengupta.pptx
Service Functions Standards Core Engine HPC
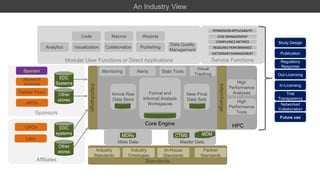
- 1. Service Functions Standards Core Engine HPC Sponsors Affiliates CROs Partner Firms Research Institutes Labs AROs Sponsor EDC Systems Other stores EDC systems Other stores Interchange Arrival Raw Data Store Formal and Informal Analysis Workspaces Near-Final Data Sets Master DataMeta Data Industry Standards Partner Standards In-House Standards Industry Ontologies MDMCTMSMDRs High Performance Analyses High Performance Tools Modular User Functions or Direct Applications Analytics Visualization Publishing Data Quality Management Collaboration Interchange Monitoring Alerts Visual Tracking Code Macros Wizards PERMISSION APPLICABILITY COMPLIANCE METRICS CASE MANAGEMENT RESOURCE PERFORMANCE DICTIONARY MANAGEMENT Study Design Publication Out-Licensing Regulatory Response In-Licensing Trial Transparency Networked Collaboration Future use MDMMDM Stats Tools An Industry View