BIOAVAILABILITY IN A NUTSHELL.pdf
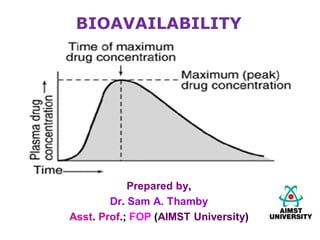
- 3. Peak plasma concentration (Cmax) • Point of max. conc. of drug in plasma is called ‘Peak’. • At this point, absorption rate = elimination rate • The conc. of drug at the peak is called Cmax (mcg/ml) tmax • Time for the conc. of drug to reach the peak level; AUC • Is the integrated area under the plasma conc.–time curve; • It expresses the total amount of the drug that enters into the systemic circulation post-administration. • (mcg/ml) x hours
- 5. INTRODUCTION 1st stated by Osler and colleagues (Osler et al. 1945) – studying absorption of vitamins from p’ceutical products – termed it as ‘physiological availability’; The relative amount of an administered dose that reaches the general circulation, and the rate at which this occurs (APA, 1972). The rate and extent to which the active ingredient(s) or therapeutic moiety(ies) is/are absorbed from a product, and becomes available at the site of drug action (USFDA, 1977). USFDA definition was not well accepted by experts, researchers, and academicians b’coz… • drug concs. are seldom monitored at the site of action in BA studies, and • site of action may not even be known initially.
- 7. Pharmaceutically / Chemically equivalent: 2 or more drug products contain equal amounts of the same therapeutically active ingredient in identical dosage forms. • These dosage forms meet the criteria of purity, content uniformity, etc. Bioequivalence: 2 or more chemically or pharmaceutically equivalent products produce comparable bioavailability characteristics in any individual when administered in equivalent dosage regimen.
- 8. Pharmaceutical alternatives: Drug products that contain the same therapeutic agent but differ in…. salt or ester form; dosage form; strength. Q: Can pharmaceutical alternatives be interchangeable in therapy???? Therapeutically equivalent: 2 or more chemically or pharmaceutically equivalent products produce the same efficacy and / or toxicity in the same individuals when administered in an equivalent dosage regimen.
- 9. Bioavailability (BA ) (BA) (F) 2 types: Absolute and Relative Absolute BA Assessed by comparing (AUC)0 ∞ and / or cumulative mass of drug excreted in the urine (Xu), following administration of a drug in an extravascular dosage form and an equal dose of the same drug in I.V. form; Absolute BA = fraction of drug absorbed (F) Value can range till 1.0 From plasma conc.-time curve data F = (AUC)extravascular/Dose extravascular // (AUC) IV/Dose IV F = [(AUC)extravascular x Dose IV] ______________________ [(AUC) IV x Dose extravascular]
- 10. If we compare BA of oral vs the I.V. F = [(AUC)oral xDose IV] _______________ [(AUC) IV x Dose oral] From urinary data: Xu: • Is the cumulative amt. of drug excreted in the urine; • Analogous to AUC Tu(max): • Is the time required for max. excretion rate; • Analogous to tmax (dXu/dt)max: • Is the max. urinary excretion rate; • Obtained from the peak of plot between urinary excretion rate and midpoint time of urine collection period. (analogous to Cmax)
- 11. From urinary data: Principle: The amount of unchanged drug in the urine is directly proportional to the plasma conc. of the drug. Useful when studying BA of drugs which are excreted unchanged in the urine.
- 12. Method: • Collection of urine at regular intervals for a time span of 7 t1/2; • Analyze unchanged drug in the samples; • Determine amt. of drug excreted in each interval, and cumulative amt. excreted; • At each sample collection, total emptying of the bladder is mandatory (to prevent addition of residual amt. of drug into the next sample collection which will lead to errors) F = [(Xu)oral 7t1/2 xDose IV] _______________ [(Xu) IV 7t1/2 x Dose oral]
- 13. Relative BA (Frel) Is assessed by comparing the BA parameters (either from plasma con.- time curve or urinary data) of the same drug moiety….. • in two or more different dosage forms (tablet, soln., syrup, capsule, etc..); • Via two or more different extravascular routes of admn. (oral, IM, IP) F = [(Xu)tablet 7t1/2 x Dose solution] _______________ [(Xu) solution 7t1/2 x Dose tablet] Frel = [(AUC)oral x Dose IM] _______________ [(AUC) IM x Dose oral] The reference standard when determining comparative or Frel must be chosen by considering which dosage form is being compared with the other dosage form. Can range from <1, 1, or >1 Plasma conc.-time curve Urinary excretion data
- 14. Factors affecting BA Formulation factors: • Excipients used • Particle size of active ingredient • Crystalline or amorphous nature of the drug • Hydrous or anhydrous form • Polymorphic nature of the drug • Drug solubility and dissolution rate; Disintegration time (tablets) • Hydrophilicity; Lipophilicity; • Drug stability • Drug pKa and pH
- 15. Physiological factors: • Age of the patient; • Gastric emptying rate; • Intestinal motility; Intestinal transit time; • Changes in GI pH; • Changes in nature of intestinal wall; • Blood flow through the GIT; • Disease states; • GI contents (food, fluids, other drugs); • Pre-systemic clearance (First-pass effect);
- 16. FIRST-PASS EFFECT After oral administration, drug passes through the gut wall and gut lumen, then is taken to the liver via the hepatic portal vein. 2 events occur: • Drug is absorbed from the gut into portal circulation; • In the liver, metabolism by enzymes occurs (First-pass effect). Extraction of the orally-administered drug by the liver is called ‘First- pass Effect’. F* = 1 – E • F* is the fraction of drug that escapes the first-pass effect • E is the hepatic extraction ratio. Compared to the I.V. form, the oral dose first goes into the portal circulation (not the systemic circulation), leading to high potential for experiencing ‘first-pass effect’.
- 18. Hepatic Extraction Ratio (E) or (ER): ER = CL liver / Q Where, Q = hepatic blood flow (usually about 90 L per hour) The overall BA is: F = fa x F* • F = fraction of drug that reaches the systemic circulation • fa = fraction of the dose absorbed from gut into portal circulation • F* = fraction of absorbed dose that survives the first-pass effect
- 19. Bioequivalence studies: A type of comparative or relative BA study, where the peak plasma concentration and peak time are determined for two (or more) chemically or pharmaceutically-equivalent products, where one of them is an ‘Innovator’. Innovator: Is the reference or standard drug. F = [(AUC)generic xDose standard] _______________ [(AUC) standard x Dose generic]
- 20. THE END
- 21. To Refresh your memory…
- 22. • Drug Absorption: the rate at which a drug enters the systemic circulation. Instantaneous for bolus intravenous administration. • Bioavailability: F, the fraction of the dose that reaches the systemic circulation. F=1 for IV administration. • Absolute Bioavailability: Estimation of ‘F’ for any other route in comparison to intravenous administration. • Relative Bioavailability: Estimation of ‘F’ for a dosage form to another given by an extravascular (non-intravenous) route of administration.
- 23. • Distribution: Movement of drug from the central compartment (tissues) to peripheral compartments (tissues) where the drug will accumulate (mostly). • Elimination: The processes that encompass the effective removal of the drug from the body through excretion or metabolism. • Excretion: The removal of drug from the body by a physical process such as excretion into urine, bile, or sweat. • Metabolism: The removal of drug from the body by metabolic transformation of the drug into other compounds. These processes include phase 1 (oxidative) or phase 2 (conjugative) metabolism.
- 24. • Volume of Distribution: the theoretical size (volume) of the space necessary to contain the amount of drug in the body given its concentration in specific fluids. • Clearance: the characterization of the volume which the body through elimination can completely remove all drug in a given period of time. • Half-Life: the length of time necessary to eliminate 50% of the remaining amount of drug present in the body. • Steady-State: the equilibrium condition reached when the amount of drug put into the kinetic system over time exactly equals the amount of drug eliminated by the system over that same period of time. (rate in = rate out)
- 25. • Concentration: the measurement of the amount of drug contained in a specific volume of a biological fluid, typically plasma or urine. Cp • Maximum Concentration (Cmax): the highest OBSERVED concentration from those included as the measurements of the time course of drug. • Time of Maximum Concentration (Tmax): the time at which the highest concentration is measured from those included as the measurements of the time course of drug. • Area Under the Curve: the integration of drug concentration measurements over time using calculus. AUC0-∞ AUC0-24 AUC0-t