Law of Conservation of Matter
•
0 likes•660 views
This document contains examples of chemical equations that show reactants reacting to form products through balancing chemical equations. The examples demonstrate balancing elements and polyatomic ions by changing only the coefficients in front of the formulas. Key principles shown are the law of conservation of matter, identifying diatomic elements, and using coefficients to balance chemical equations.
Report
Share
Report
Share
Download to read offline
More Related Content
What's hot
What's hot (19)
Viewers also liked
WITHIN TREE VARIATION IN ANTOMICAL PROPERTIES OF SOME CLONES OF EUCALYPTUS TE...

WITHIN TREE VARIATION IN ANTOMICAL PROPERTIES OF SOME CLONES OF EUCALYPTUS TE...International Journal of Technical Research & Application
Chromatography: Comparison of EPA Methods 300.1, 317, 326 and 302 for Bromate...

Chromatography: Comparison of EPA Methods 300.1, 317, 326 and 302 for Bromate...Chromatography & Mass Spectrometry Solutions
Viewers also liked (17)
WITHIN TREE VARIATION IN ANTOMICAL PROPERTIES OF SOME CLONES OF EUCALYPTUS TE...

WITHIN TREE VARIATION IN ANTOMICAL PROPERTIES OF SOME CLONES OF EUCALYPTUS TE...
Chromatography: Comparison of EPA Methods 300.1, 317, 326 and 302 for Bromate...

Chromatography: Comparison of EPA Methods 300.1, 317, 326 and 302 for Bromate...
Manufacturing of chlorine using diaphragm cell, membrane cell

Manufacturing of chlorine using diaphragm cell, membrane cell
Similar to Law of Conservation of Matter
Similar to Law of Conservation of Matter (20)
Chemistry - Chp 11 - Chemical Reactions - PowerPoint

Chemistry - Chp 11 - Chemical Reactions - PowerPoint
Chemistry - Chp 11 - Chemical Reactions - PowerPoint

Chemistry - Chp 11 - Chemical Reactions - PowerPoint
More from Ginger Bellisario
More from Ginger Bellisario (14)
Recently uploaded
Organic Name Reactions for the students and aspirants of Chemistry12th.pptx

Organic Name Reactions for the students and aspirants of Chemistry12th.pptxVS Mahajan Coaching Centre
call girls in Kamla Market (DELHI) 🔝 >༒9953330565🔝 genuine Escort Service 🔝✔️✔️

call girls in Kamla Market (DELHI) 🔝 >༒9953330565🔝 genuine Escort Service 🔝✔️✔️9953056974 Low Rate Call Girls In Saket, Delhi NCR
Recently uploaded (20)
Call Girls in Dwarka Mor Delhi Contact Us 9654467111

Call Girls in Dwarka Mor Delhi Contact Us 9654467111
Introduction to ArtificiaI Intelligence in Higher Education

Introduction to ArtificiaI Intelligence in Higher Education
Hybridoma Technology ( Production , Purification , and Application ) 

Hybridoma Technology ( Production , Purification , and Application )
Organic Name Reactions for the students and aspirants of Chemistry12th.pptx

Organic Name Reactions for the students and aspirants of Chemistry12th.pptx
Science 7 - LAND and SEA BREEZE and its Characteristics

Science 7 - LAND and SEA BREEZE and its Characteristics
A Critique of the Proposed National Education Policy Reform

A Critique of the Proposed National Education Policy Reform
call girls in Kamla Market (DELHI) 🔝 >༒9953330565🔝 genuine Escort Service 🔝✔️✔️

call girls in Kamla Market (DELHI) 🔝 >༒9953330565🔝 genuine Escort Service 🔝✔️✔️
18-04-UA_REPORT_MEDIALITERAСY_INDEX-DM_23-1-final-eng.pdf

18-04-UA_REPORT_MEDIALITERAСY_INDEX-DM_23-1-final-eng.pdf
Blooming Together_ Growing a Community Garden Worksheet.docx

Blooming Together_ Growing a Community Garden Worksheet.docx
Kisan Call Centre - To harness potential of ICT in Agriculture by answer farm...

Kisan Call Centre - To harness potential of ICT in Agriculture by answer farm...
Law of Conservation of Matter
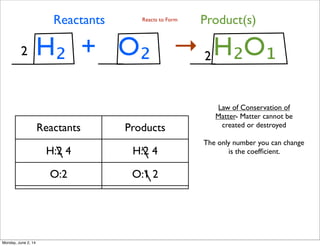
- 1. H₂ + O₂ → H₂O₁ Law of Conservation of Matter- Matter cannot be created or destroyed Reactants Product(s)Reacts to Form Reactants Products H:2 4 H:2 4 O:2 O:1 2 The only number you can change is the coefficient. 2 2 Monday, June 2, 14
- 2. _Al + _H₂(SO₄) → _Al₂ (SO₄)₃ + _H₂ If a polyotomic appears on both sides, we balance it as one. Reactants Products SO₄ :1 3 SO₄:3 Al:1 2 Al:2 H:2 6 H:2 6 3 32 1 Diatomic: -Hydrogen -Nitrogen -Oxygen -Florine -Chlorine -Bromine -Iodine Monday, June 2, 14
- 3. Aluminum and Oxygen react to form Aluminum Oxide. _Al + _O₂ → _Al₂_O₃ Reactants Products Al: 1 4 Al: 2 4 O: 2 6 O: 3 6 3 24 Monday, June 2, 14
- 4. Potassium Chlorate decomposes to form potassium chloride and oxygen gas. _K(CIO₃) → _K(Cl) + _O₂ Reactants Products K: 1 2 K: 12 Cl: 12 Cl: 12 O: 3 6 O: 2 6 32 2 Monday, June 2, 14
- 5. Aluminum Nitrate and Sodium Hydroxide React to from Aluminum Hydroxide and Sodium Nitrate. Al+³ NO-¹₃ Na +¹ OH -¹ Al(NO₃)₃ + _NaOH →Al(OH)₃ + _NaNO₃ Reactants Products Al: 1 Al:1 NO₃: 3 NO₃: 13 Na:1 3 Na:13 OH: 1 3 HO:3 33 Monday, June 2, 14
