The blackbody radiation after Planck
•Download as PPTX, PDF•
0 likes•120 views
From the Udemy online course "The weird World of Quantum Physics - A primer on the conceptual foundations of Quantum Physics": https://www.udemy.com/quantum-physics/?couponCode=SLIDESHCOUPON
Report
Share
Report
Share
Recommended
More Related Content
Similar to The blackbody radiation after Planck
Similar to The blackbody radiation after Planck (20)
Proposed experiments to prove the grid dimensions theory 

Proposed experiments to prove the grid dimensions theory
Introduction to quantum mechanics and schrodinger equation

Introduction to quantum mechanics and schrodinger equation
Kepler 3rd law explanation part no.2 (to support my claim against nobel priz...

Kepler 3rd law explanation part no.2 (to support my claim against nobel priz...
More from Satyavan65
More from Satyavan65 (8)
The wave-particle duality and the double slit experiment

The wave-particle duality and the double slit experiment
Recently uploaded
Bentham & Hooker's Classification. along with the merits and demerits of the ...

Bentham & Hooker's Classification. along with the merits and demerits of the ...Nistarini College, Purulia (W.B) India
Recently uploaded (20)
Behavioral Disorder: Schizophrenia & it's Case Study.pdf

Behavioral Disorder: Schizophrenia & it's Case Study.pdf
STERILITY TESTING OF PHARMACEUTICALS ppt by DR.C.P.PRINCE

STERILITY TESTING OF PHARMACEUTICALS ppt by DR.C.P.PRINCE
Recombinant DNA technology( Transgenic plant and animal)

Recombinant DNA technology( Transgenic plant and animal)
PossibleEoarcheanRecordsoftheGeomagneticFieldPreservedintheIsuaSupracrustalBe...

PossibleEoarcheanRecordsoftheGeomagneticFieldPreservedintheIsuaSupracrustalBe...
Unlocking the Potential: Deep dive into ocean of Ceramic Magnets.pptx

Unlocking the Potential: Deep dive into ocean of Ceramic Magnets.pptx
Bentham & Hooker's Classification. along with the merits and demerits of the ...

Bentham & Hooker's Classification. along with the merits and demerits of the ...
Analytical Profile of Coleus Forskohlii | Forskolin .pdf

Analytical Profile of Coleus Forskohlii | Forskolin .pdf
Genomic DNA And Complementary DNA Libraries construction.

Genomic DNA And Complementary DNA Libraries construction.
Grafana in space: Monitoring Japan's SLIM moon lander in real time

Grafana in space: Monitoring Japan's SLIM moon lander in real time
Call Us ≽ 9953322196 ≼ Call Girls In Mukherjee Nagar(Delhi) |

Call Us ≽ 9953322196 ≼ Call Girls In Mukherjee Nagar(Delhi) |
Discovery of an Accretion Streamer and a Slow Wide-angle Outflow around FUOri...

Discovery of an Accretion Streamer and a Slow Wide-angle Outflow around FUOri...
Recombination DNA Technology (Nucleic Acid Hybridization )

Recombination DNA Technology (Nucleic Acid Hybridization )
Scheme-of-Work-Science-Stage-4 cambridge science.docx

Scheme-of-Work-Science-Stage-4 cambridge science.docx
Call Girls in Mayapuri Delhi 💯Call Us 🔝9953322196🔝 💯Escort.

Call Girls in Mayapuri Delhi 💯Call Us 🔝9953322196🔝 💯Escort.
The blackbody radiation after Planck
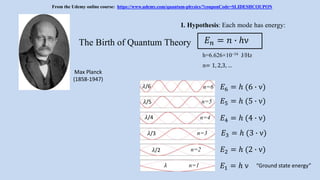
- 1. Max Planck (1858-1947) h=6.626×10−34 J/Hz 𝐸 𝑛 = 𝑛 ∙ ℎν I. Hypothesis: Each mode has energy: The Birth of Quantum Theory n= 1, 2,3, … 𝐸1 = ℎ ν “Ground state energy”n=1 n=2 n=3 n=4 n=5 n=6 𝐸2 = ℎ (2 ∙ ν) 𝐸3 = ℎ (3 ∙ ν) 𝐸4 = ℎ (4 ∙ ν) 𝐸5 = ℎ (5 ∙ ν) 𝐸6 = ℎ (6 ∙ ν)𝜆/6 𝜆 𝜆/5 𝜆/4 𝜆/3 𝜆/2 From the Udemy online course: https://www.udemy.com/quantum-physics/?couponCode=SLIDESHCOUPON
- 2. Max Planck (1858-1947) h=6.626×10−34 J/Hz 𝐸 𝑛 = 𝑛 ∙ ℎν Hydrogen’s and mercury’s spectral emission lines (wavelength units: nm=10−9 m) I. Hypothesis: Each mode has energy: The Birth of Quantum Theory n= 1, 2,3, …
- 3. II. Hypothesis: the modes are not equally probable. Boltzmann’s distribution law (much more modes with lower energy than those with higher ones). Ludwig Boltzmann (1844-1906) 𝑃 𝐸 𝑛+1 < 𝑃 𝐸 𝑛 𝑃 𝐸∞ =0 𝐸1 + 𝐸2+ 𝐸3+...+ 𝐸∞< ∞ ! Max Planck (1858-1947) 𝐸 𝑛 = 𝑛 ∙ ℎν I. Hypothesis: Each mode has energy: The Birth of Quantum Theory h=6.626×10−34 J/Hz n= 1, 2,3, …
- 5. Ultraviolet catastrophe Rayleigh-Jeans formula Planck‘s formula Intensity(a.u.)
- 7. The cosmic microwave background radiation Black body radiation can be explained only if we assume that: Energy is absorbed or emitted only in discrete packets or ”quanta”. 2.7 K