Acid base balance in body
- 1. Name : Samyak Pradeep nagwanshi
- 3. The body normally maintains a steady Balance between acid produced during Metabolism and bases that neutralize and Promote the excretion of the acids .
- 4. People experiencing vomiting and diarrhoea Can lose acid and base in Addition to loss of fluids and electrolytes . The older adult is at risk – their kidneys and lungs do not compensate so well anymore for acid – base imbalance
- 5. BASE : Any ionic or molecular substance that can act as proton acceptor . ex: NaoH , KOH ACID: Any ionic or molecular substance that can act a proton donor . ex: HCL , H2SO4
- 6. The acidity or alkalinity of a solution depends upon its hydrogen (H+) ion concentration
- 7. An increase in H+ concentration leads to acidity An decrease in H+ concentration leads To alkalinity
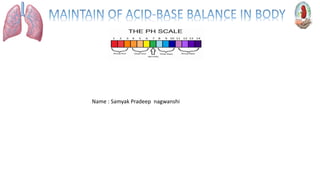
- 8. An acid solution has a pH of less than (<) 7 An alkaline solution has a pH of greater than (>) 7 A neutral solution has a pH of 7
- 9. Blood is slightly alkaline (pH7.35 -7.46) If the pH of Blood drop below 7.35 the person is said to have acidosis If the p H of blood is greater than 7.45 the person has alkalosis
- 10. The body’s metabolic processes are Constantly producing acid . These acid must be neutralize and Excreted to maintain acid base balance.
- 11. Normally the body has three 3 mechanisms by Which it regulates acid base balance to maintain The arterial p H between 7.35 -7.45 . These mechanisms are : 1 . The buffer system 2. The respiratory system 3. The renal system
- 12. • The buffer systems are the fastest acting Systems and are the primary regulator of Acid –base balance • Buffer act chemically to change strong acid into weaker acid 0r to bind acids to neutralize
- 13. • A buffer consists of a weakly ionized acids or base and its salt that function to Minimize the effects of acids on blood p H Until they can be excreted from the body . • Buffers alone cannot maintain blood p H And they need an adequately functioning Respiratory and renal system .
- 14. • Lungs help maintain a normal p H by Excreting Co2 and water which are by Products of cellular metabolism . • The rat of Co2 excretion is controlled by The respiratory center in the medulla in the Brainstem .
- 15. • If increased Co2 or H+ are detected the Respiratory center simulates an increased Rate and depth of breathing • Respiratory rate and depth are inhibited if the center detects low H+ or co2 levels
- 16. i. If a respiratory condition is the cause of an acid base imbalance (respiratory failure for example) The respiratory system loses It’s ability to correct a p H alteration .
- 17. • Under normal circumstances of the kidneys Reabsorb and conserve all of the bicarbonate they filter . • The kidneys can also generate more bicarbonate and eliminate excess H+ to Compensate for acidosis .
- 18. • The kidneys usually excrete acidic urine (average p H 6). • If the renal system is the cause of an acid – Base imbalance (renal failure ,for example ) It loses its ability to correct an acid –base Imbalance.