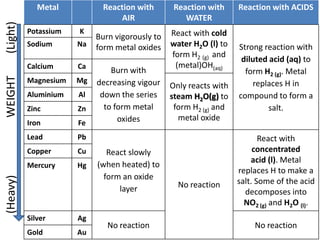
Summary of Metal Reactions
- 1. Metal Reaction with Reaction with Reaction with ACIDS AIR WATER (Light) Potassium K React with cold Burn vigorously to Sodium Na form metal oxides water H2O (l) to Strong reaction with form H2 (g) and diluted acid (aq) to Calcium Ca (metal)OH(aq) Burn with form H2 (g). Metal WEIGHT Magnesium Mg decreasing vigour replaces H in Only reacts with Aluminium Al down the series steam H2O(g) to compound to form a Zinc Zn to form metal form H2 (g) and salt. oxides metal oxide Iron Fe Lead Pb React with Copper Cu React slowly concentrated acid (l). Metal Mercury Hg (when heated) to replaces H to make a form an oxide (Heavy) No reaction salt. Some of the acid layer decomposes into NO2 (g) and H2O (l). Silver Ag No reaction No reaction Gold Au
