Progress on the Path to Improving Outcomes in the Treatment of SCLC: Making the Most of Current Standard-of-Care Therapies and Exploring New Promising Research
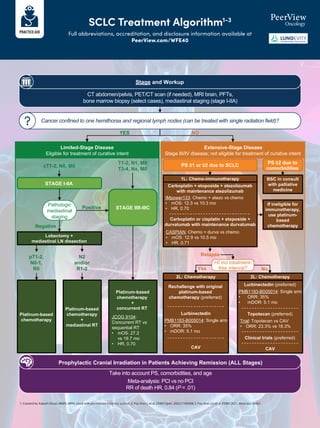
- 1. SCLC Treatment Algorithm1-3 Full abbreviations, accreditation, and disclosure information available at PeerView.com/WFE40 1. Created by Aakash Desai, MBBS, MPH. Used with permission from the author. 2. Paz-Ares L et al. ESMO Open. 2022;7:100408.3. Paz-Ares LG et al. ESMO 2021. Abstract LBA61. CT abdomen/pelvis, PET/CT scan (if needed), MRI brain, PFTs, bone marrow biopsy (select cases), mediastinal staging (stage I-IIA) Stage and Workup Cancer confined to one hemithorax and regional lymph nodes (can be treated with single radiation field)? !"#"$%&'($)*% ?#*@*&#($2,'$+'(%+3(-+$,2$0"'%+*)($*-+(-+ Limited-Stage Disease Eligible for treatment of curative intent %4$%5("6%'($)*% Extensive-Stage Disease Stage III/IV disease; not eligible for treatment of curative intent YES NO STAGE I-IIA STAGE IIB-IIIC Pathologic mediastinal staging Positive Negative Platinum-based chemotherapy + concurrent RT JCOG 9104: Concurrent RT vs sequential RT • mOS: 27.2 vs 19.7 mo • HR, 0.70 Lobectomy + mediastinal LN dissection Platinum-based chemotherapy Platinum-based chemotherapy + mediastinal RT pT1-2, N0-1, R0 N2 and/or R1-2 Take into account PS, comorbidities, and age Meta-analysis: PCI vs no PCI RR of death HR, 0.84 (P = .01) Prophylactic Cranial Irradiation in Patients Achieving Remission (ALL Stages) PS ≤1 or ≥2 due to SCLC PS ≥2 due to comorbidities BSC in consult with palliative medicine 1L: Chemo-immunotherapy If ineligible for immunotherapy, use platinum- based chemotherapy >6 mo treatment- free interval? Relapse Yes No 2L: Chemotherapy 2L: Chemotherapy Carboplatin + etoposide + atezolizumab with maintenance atezolizumab IMpower133: Chemo + atezo vs chemo • mOS: 12.3 vs 10.3 mo • HR, 0.70 Carboplatin or cisplatin + etoposide + durvalumab with maintenance durvalumab CASPIAN: Chemo + durva vs chemo • mOS: 12.9 vs 10.5 mo • HR, 0.71 Rechallenge with original platinum-based chemotherapy (preferred) Lurbinectedin PMB1183-B005014: Single arm • ORR: 35% • mDOR: 5.1 mo CAV Lurbinectedin (preferred) PMB1183-B005014: Single arm • ORR: 35% • mDOR: 5.1 mo Topotecan (preferred) Trial: Topotecan vs CAV • ORR: 23.3% vs 18.3% Clinical trials (preferred) CAV cT1-2, N0, M0 T1-2, N1, M0 T3-4, Nx, M0
- 2. The Immunotherapy Era in SCLC: Approved Treatments and Immune-Related Adverse Event Management Full abbreviations, accreditation, and disclosure information available at PeerView.com/WFE40 Currently Approved PD-L1–Blocking Antibodies in Immuno-Chemotherapies, Supporting Evidence, and Practical Guidance Durvalumab1,2 Granted FDA approval in combination with etoposide and either carboplatin or cisplatin as first-line treatment of patients with ES-SCLC on March 27, 2020 Indicated in combination with etoposide and either carboplatin or cisplatin as first-line treatment of adult patients with ES-SCLC Indication 1,500 mg every 3 weeks prior to chemotherapy in combination with etoposide and either carboplatin or cisplatin, then every 4 weeks as a single agent Dosing2 CASPIAN: A Randomized, Controlled, Open-Label, Phase 3 Trial3-6 Most Common AEs (All Grades)1 • Nausea (34%) • Fatigue/asthenia (32%) • Alopecia (31%) Atezolizumab7,8 Granted FDA approval in combination with carboplatin and etoposide for first-line treatment of adult patients with ES-SCLC on March 18, 2019 Indicated in combination with carboplatin and etoposide for first-line treatment of adult patients with ES-SCLC Indication 1,200 mg every 3 weeks prior to chemotherapy with carboplatin and etoposide; 840 mg every 2 weeks, 1,200 mg every 3 weeks, or 1,680 mg every 4 weeks following completion of 4 cycles of carboplatin and etoposide Dosing7 IMpower33: A Double-Blind, Placebo-Controlled, Phase 3 Trial9 Most Common AEs (All Grades)7 " • Fatigue/asthenia • Nausea • Alopecia • Constipation • Diarrhea • Decreased appetite Platinum-etoposide (n = 269) Durvalumab + Platinum-etoposide (n = 268) 156 (58) 182 (68) 5.1 (4.8-5.3) 5.1 (4.9-5.3) Median duration of response, mo (95% CI) 10.5 (9.3-11.2) 12.9 (11.3-14.7) Median OS, mo (95% CI) 5.4 (4.8-6.2) 5.1 (4.7-6.2) Median PFS, mo (95% CI) a Objective response by investigator review per RECIST 1.1 is defined as patients with complete response or partial response on at least one visit (unconfirmed responses); for confirmed responses, a confirmatory scan was required ≥4 weeks after the initial response. Placebo (n = 202) Atezolizumab (n = 201) 130 (64.4) 121 (60.2) 3.9 (2.0-16.1) 4.2 (1.4-19.5) Median duration of response, mo (range)c Confirmed objective response, n (%)a Confirmed objective response, n (%)b 10.3 (9.3-11.3) 12.3 (10.8-15.9) Median OS, mo (95% CI) 4.3 (4.2-4.5) 5.2 (4.4-5.6) Median PFS, mo (95% CI) b Assessed in patients in the ITT population who had measurable disease at baseline. Defined as confirmed complete response or partial response by investigator review per RECIST 1.1. c Assessed in patients who had a confirmed objective response. Defined as the time from first occurrence of a documented objective response to the time of disease progression as determined by the investigator (according to RECIST) or death from any cause, whichever occurred first.
- 3. The Immunotherapy Era in SCLC: Approved Treatments and Immune-Related Adverse Event Management Full abbreviations, accreditation, and disclosure information available at PeerView.com/WFE40 What Are irAEs?1 • Immune checkpoint inhibitors are associated with important clinical benefits, but general immunologic enhancement can also lead to a unique spectrum of immune-related adverse events • Any organ system can be affected, but more commonly occurring are pulmonary (pneumonitis), dermatologic (rash, pruritus, blisters, ulcers, vitiligo), gastrointestinal (diarrhea, enterocolitis, transaminitis, hepatitis, pancreatitis), and endocrine (thyroiditis, hypophysitis, adrenal insufficiency) irAEs Endocrine Hyper- or hypothyroidism Hypophysitis Adrenal insufficiency Diabetes Hepatic Hepatitis Renal Nephritis Dermatologic Rash Pruritus Psoriasis Vitiligo DRESS Stevens-Johnson Hematologic Hemolytic anemia Thrombocytopenia Neutropenia Hemophilia Ocular Uveitis Conjunctivitis Scleritis, episcleritis Blepharitis Retinitis Respiratory Pneumonitis Pleuritis Sarcoid-like granulomatosis Cardiovascular Myocarditis Pericarditis Vasculitis Gastrointestinal Colitis Ileitis Pancreatitis Gastritis Neurologic Neuropathy Guillain Barré Myelopathy Encephalitis Myasthenia Musculoskeletal Arthritis Dermatomyositis Immune-Related Adverse Events (irAEs)10
- 4. General Recommendations for Treating irAEs11-14 Increasing Intensity of Treatment Required Increasing Grade of irAE Managed in Outpatient/Community Setting Generally Requires Hospital Admission Steroids (PO/IV): 1-2 mg/kg/d prednisone or equivalent, slowly taper over 4-6 weeks For some AEs, treatment can be restarted after resolution (eg, rash); generally, ICI can be continued with endocrinopathies once managed Grade 2 Grade 1 Grade 3 Grade 4 Moderate Mild Severe Very Severe Symptomatic & supportive therapy Stop treatment Oral steroids Intravenous steroids. ------------> • Referral to specialist • Strong immune-suppressive treatment intravenous steroids The Immunotherapy Era in SCLC: Approved Treatments and Immune-Related Adverse Event Management Full abbreviations, accreditation, and disclosure information available at PeerView.com/WFE40
- 5. The Immunotherapy Era in SCLC: Approved Treatments and Immune-Related Adverse Event Management Full abbreviations, accreditation, and disclosure information available at PeerView.com/WFE40 Hold immunotherapy and reassess in 1-2 weeks Pulse oximetry rest and ambulation Consider chest imaging with CT (with contrast preferred) Repeat in 3-4 weeks Moderate (grade 2): 25%-50% lung involved Severe (grade 3-4) Grade 3: all lobes of lung or >50% of lung parenchyma; limited ADLs, oxygen requirement Grade 4: life threatening Hold immunotherapy Infectious workup (nasal swab, sputum, blood) Consider bronchoscopy and BAL Chest imaging with CT contrast Repeat in 3-4 weeks Consider empiric antibiotics Refractory: methylprednisolone 1-2 mg/m2 /day; if no response in 3-4 days, treat as grade 3 Permanently discontinue immunotherapy and move to inpatient care Infectious workup (nasal swab, sputum, blood) Pulmonary and infectious disease consultation Bronchoscopy with BAL Empiric antibiotics Methylprednisolone 1-2 mg/m2 /day; when grade 1, taper over 6 weeks Refractory: infliximab, mycophenolate, or IVIG How Should Pulmonary irAEs Be Diagnosed and Managed?11,15 Pneumonitis: focal or diffuse inflammation of the lung parenchyma (typically identified on CT imaging) Diagnostic workup: CXR, CT, pulse oximetry; for grade ≥2, may include infectious workup Mild (grade 1): <25% lung involved Additional considerations • GI and pneumocystis prophylaxis may be offered to patients on prolonged steroid use (>12 weeks) • Consider calcium and vitamin D supplementation with prolonged steroid use • Bronchoscopy and biopsy; if clinical picture is consistent with pneumonitis, no need for biopsy Supportive care: smoking cessation and vaccinations (influenza, pneumococcal)
- 6. The Immunotherapy Era in SCLC: Approved Treatments and Immune-Related Adverse Event Management Full abbreviations, accreditation, and disclosure information available at PeerView.com/WFE40 1. Imfinzi (durvalumab) Prescribing Information. https://www.accessdata.fda.gov/drugsatfda_docs/label/2022/761069s033lbl.pdf. 2. https://www.fda.gov/drugs/resources-information-approved-drugs/fda-approves-durvalumab-extensive-stage-small-cell-lung-cancer. 3. Paz-Ares L et al. Lancet. 2019;394:1929-1939. 4. Goldman JW et al. Lancet Oncol. 2021;22:51-65. 5. Paz-Ares L et al. ESMO Open. 2022;7:100408. 6. Paz-Ares LG et al. ESMO 2021. Abstract LBA61. 7. Tecentriq (atezolizumab) Prescribing Information. https://www.accessdata.fda.gov/ drugsatfda_docs/label/2023/761034s049s051lbl.pdf. 8. https://www.fda.gov/drugs/drug-approvalsand-databases/fda-approves-atezolizumab-extensive-stage-small-cell-lung-cancer. 9. Horn L et al. N Engl J Med. 2018;379:2220-2229. 10. Champiat S et al. Ann Oncol. 2016;27:559- 574. 11. Brahmer JR et al. J Clin Oncol. 2018;36:1714-1786. 12. https://www.esmo.org/content/download/124130/2352601/1/ESMO-Patient-Guide-on-Immunotherapy-Side-Effects.pdf. 13. https://www.nccn.org/professionals/physician_gls/pdf/immunotherapy.pdf. 14. Puzanov I et al. J Immunother Cancer. 2017;5:95. 15. Provided courtesy of Marianne Davies, DNP, ACNP, AOCNP, FAAN, 2021; adapted from AIM with Immunotherapy, NCCN, and CTCAE. 16. https://ascopubs.org/doi/full/10.1200/JCO.21.01440. 17. https://www.esmo.org/content/download/124130/2352601/1/ESMO-Patient-Guide-on-Immunotherapy-Side-Effects.pdf. 18. https://www.nccn.org/professionals/physician_gls/pdf/immunotherapy.pdf. 19. https://www.sitcancer.org/research/cancer-immunotherapy-guidelines/irae/immune-checkpoint-inhibitor-related-adverse-events. Additional Guideline Recommendations for Treating irAEs16-19
- 7. Lurbinectedin: The Newest FDA-Approved Second-Line Treatment Option in SCLC Full abbreviations, accreditation, and disclosure information available at PeerView.com/WFE40 Lurbinectedin (Selective Inhibitor of Oncogenic Transcription)1,2 Granted accelerated FDA approval on June 15, 2020 based on overall response rate and duration of response Indicated for the treatment of adult patients with metastatic SCLC with disease progression on or after platinum-based chemotherapy Indication 3.2 mg/m2 via intravenous infusion every 21 d (consider premedication with corticosteroids and serotonin antagonists) Dosing2 Mechanism of Action1 • Category 2A other recommended regimen following first-line platinum-based chemotherapy • For patients with ECOG PS 0-2 (consider dose reduction or growth factor support for patients with ECOG PS 2) NCCN Guidelines: Second-Line and Beyond Therapy3 ORR by Investigator Assessment1 Most Common Grade 3/4 AEs1 Other common adverse events include lymphopenia, fatigue, increased creatinine, increased alanine aminotransferase, increased glucose, nausea, decreased appetite, musculoskeletal pain, decreased albumin, constipation, dyspnea, decreased sodium, increased aspartate aminotransferase, vomiting, cough, decreased magnesium, and diarrhea Neutropenia 46% Anemia 9% Leukopenia 29% Thrombocytopenia 7% All patients 35.2% Chemotherapy-free interval ≥90 d 45.0% Chemotherapy-free interval <90 d 22.2% Tumor CCL2 Induction of apoptosis Monocyte CCL2 CXCL8 VEGF Reduced cancer-related inflammation TAM Inhibition of cell migration LURBINECTEDIN • Synthetically produced agent, originally derived from Ecteinascidia turbinata • A selective inhibitor of oncogenic transcription 1. Zepzelca (lurbinectedin) Prescribing Information. https://www.accessdata.fda.gov/drugsatfda_docs/label/2022/213702s004lbl.pdf. 2. https://www.fda.gov/drugs/drug-approvals-and-databases/fda-grants-accelerated-approval-lurbinectedin-metastatic-small-cell-lung-cancer. 3. https://www.nccn.org/professionals/physician_gls/pdf/sclc.pdf.
- 8. LUNGevity Foundation: Small Cell Lung Cancer Resource Compendium Full abbreviations, accreditation, and disclosure information available at PeerView.com/WFE40 The following pages include information for patients with lung cancer for use as a printable resource lungevity.org Through research, advocacy, and community, LUNGevity is transforming what it means to be diagnosed and live with lung cancer www.lungevity.org/order-materials LUNGevity educational materials order form for providers: .
- 9. LUNGevity Foundation: Small Cell Lung Cancer Resource Compendium 1 What Is Small Cell Lung Cancer (SCLC)? The Stages of SCLC • Lung cancer staging describes where the cancer is located, if and where it has spread, and whether it is affecting other parts of the body • Treatments are available for every stage Extensive-stage: cancer has spread more widely throughout the lung, to the other lung, to lymph nodes on the other side of the chest, or to distant organs Limited-stage: cancer is only in one side of the chest: one lung, the tissues between the lungs, and nearby lymph nodes only Most commonly used system: 2 stages 3 2 4 Diagnosis of SCLC Treatment Options Many different tests are used to diagnose SCLC, including • Biopsies: tests in which small amounts of tissue are removed for examination to find out if a person has lung cancer and, if so, which type • Imaging: tests that create pictures of the inside of the body by x-rays, magnetic fields, sound waves, or radioactive particles Currently approved treatment options for SCLC include: • Surgery • Radiation therapy • Chemotherapy • Immunotherapy In addition, several new types of treatment are being studied for SCLC. These are available through clinical trials. https://sclc.lungevity.org/sclc/about-small-cell-lung-cancer • SCLC is one of the two major types of lung cancer • It accounts for about 15% of all lung cancers • It is most often found in people with a smoking history • SCLC derives its name from the way the cancer cells look under a microscope Trachea Bronchoscope or endoscope Throat Lymph nodes Lung Bronchi Small cell lung cancer (cells under microscope)
- 10. Booklets: www.LUNGevity.org/booklets In booklet 1, you will learn • What SCLC is • What lung cancer staging is • The stages of SCLC What is small cell lung cancer (SCLC)? One-Page Fact Sheets In booklet 2, you will learn • Types of tests doctors do to find SCLC • What happens during imaging tests • Why doctors do biopsies and what happens during them • Other ways doctors learn about your health In booklet 3, you will learn • How you and your doctor will decide on a treatment plan • What the treatment options are for SCLC • Common side effects of treatment Comprehensive booklet Questions to ask your doctor to better understand SCLC, stage, and treatment options Resources and support for SCLC 2 Educational Materials and Supporting Resources The Small Cell Lung Cancer Patient Gateway is a tailored portal for people with SCLC to learn more about treatment for SCLC, connect to experts and SCLC patient communities, and access educational resources Find a Specialist Find a Clinical Trial Blog and Forum Find Your Community 3 1 2 LUNGevity Foundation: Small Cell Lung Cancer Resource Compendium
