Advanced HCC Therapy Guide
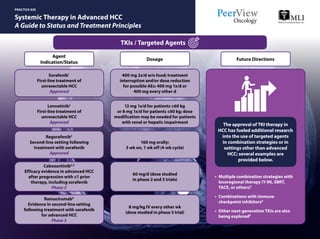
- 1. Systemic Therapy in Advanced HCC A Guide to Status and Treatment Principles PRACTICE AID TKIs / Targeted Agents Sorafenib1 First-line treatment of unresectable HCC Approved 400 mg 2x/d w/o food; treatment interruption and/or dose reduction for possible AEs: 400 mg 1x/d or 400 mg every other d Lenvatinib2 First-line treatment of unresectable HCC Approved Regorafenib3 Second-line setting following treatment with sorafenib Approved Cabozantinib4,5 Efficacy evidence in advanced HCC after progression with ≤1 prior therapy, including sorafenib Phase 3 Ramucirumab6 Evidence in second-line setting following treatment with sorafenib for advanced HCC Phase 3 12 mg 1x/d for patients ≥60 kg or 8 mg 1x/d for patients ≤60 kg; dose modification may be needed for patients with renal or hepatic impairment 160 mg orally; 3 wk on, 1 wk off (4-wk cycle) 60 mg/d (dose studied in phase 2 and 3 trials) 8 mg/kg IV every other wk (dose studied in phase 3 trial) Agent Indication/Status Dosage Future Directions • Multiple combination strategies with locoregional therapy (Y-90, SBRT, TACE, or others)7 • Combinations with immune checkpoint inhibitors8 • Other next-generation TKIs are also being explored9 The approval of TKI therapy in HCC has fueled additional research into the use of targeted agents in combination strategies or in settings other than advanced HCC; several examples are provided below.
- 2. Systemic Therapy in Advanced HCC A Guide to Status and Treatment Principles This Practice Aid has been provided as a quick reference to help learners apply the information to their daily practice and care of patients. CTLA-4: cytotoxic T-lymphocyte–associated antigen-4; HCC: hepatocellular carcinoma; PD-1: programmed cell death protein 1; PD-L1: programmed death-ligand 1; SBRT: stereotactic body radiation therapy; TACE: transarterial chemoembolization; TKI: tyrosine kinase inhibitor; Y-90: yttrium-90. 1. Nexavar (sorafenib) Prescribing Information. https://www.nexavar-us.com/. Accessed October 8, 2018. 2. Lenvima (lenvatinib) Prescribing Information. http://www.lenvima.com/pdfs/prescribing-information.pdf. Accessed October 8, 2018. 3. Stivarga (regorafenib) Prescribing Information. https://www.accessdata.fda.gov/drugsatfda_docs/label/2012/203085lbl.pdf. Accessed October 8, 2018. 4. Abou-Alfa GK et al. ASCO Gastrointestinal Cancers Symposium 2018 (ASCO GI 2018). Abstract 207. 5. Abou-Alfa GK et al. N Engl J Med. 2018;379:54-63. 6. Zhu AX et al. J Clin Oncol. 2018;36(suppl). Abstract 4003. 7. https://clinicaltrials.gov/ct2/show/NCT00846131. Accessed October 19, 2018. 8. https://clinicaltrials.gov/ct2/show/NCT03006926. Accessed October 19, 2018. 9. https://clinicaltrials.gov/ct2/show/NCT02421185. Accessed October 19, 2018. 10. Opdivo (nivolumab) Prescribing Information. https://packageinserts.bms.com/pi/pi_opdivo.pdf. Accessed October 8, 2018. 11. Zhu AX et al. ASCO GI 2018. Abstract 209. 12. Wainberg ZA et al. ASCO 2017. Abstract 4071. 13. Stein S et al. J Clin Oncol. 2018;36(suppl). Abstract 4074. 14. https://clinicaltrials.gov/ct2/show/NCT02519348. Accessed October 19, 2018. 15. https://clinicaltrials.gov/ct2/show/NCT03006926. Accessed October 19, 2018. PRACTICE AID Access the activity,“Surveying the View From the Driver’s Seat in Hepatocellular Carcinoma: Bringing Into Focus Hepatology’s Key Role in Guiding HCC Care Down the Path to Improved Outcomes,”at www.peerview.com/WDR40. Nivolumab10 Second-line setting following treatment with sorafenib Approved Phase 3 testing (CheckMate-459; NCT02576509) as first-line treatment 240 mg every 2 wk or 480 mg every 4 wk Pembrolizumab11 Second-line setting following treatment with sorafenib Priority review Durvalumab12 HCC (Child-Pugh class A) Phase 3 Atezolizumab + bevacizumab13 First-line treatment of advanced or metastatic HCC Breakthrough therapy designation 200 mg every 3 wk (dose studied in phase 2 trial) 10 mg/kg IV every other wk (dose studied in phase 1/2 trial) Atezo 1,200 mg IV every 3 wk or 840 mg every 2 wk and bev 15 mg/kg IV every 3 wk or 10 mg/kg every 2 wk (dose studied in phase 1 trial) Agent Indication/Status Dosage Future Directions • Dual checkpoint blockade (anti–PD-1/L1 + anti–CTLA-4)14 • Combinations with TKIs and with locoregional therapy15 As in other cancer settings, multiple explorations of checkpoint inhibitors in HCC, including immune combinations or as treatments, are underway: Immune Checkpoint Inhibitors
- 3. Managing Adverse Events Associated With Systemic Therapies Used in Patients With Hepatocellular Carcinoma PRACTICE AID • In general, checkpoint inhibitor therapy should be continued with close monitoring, with the exception of some neurologic, hematologic, and cardiac toxicities Minimalornosymptoms;diagnosticchangesonly Immune checkpoint inhibitors are associated with important clinical benefits, but general immunologic enhancement can also lead to a unique spectrum of irAEs Grade 1 What is the spectrum of potential irAEs?Why do irAEs occur? General recommendations and management principles include the following: irAEs are often diagnosed by exclusion; other causes should be ruled out (including AEs of other therapies used), but immunotherapy-related toxicity should always be included in the differential There should be a high level of suspicion that new symptoms are treatment-related; early recognition, evaluation, and treatment of irAEs plus patient education are essential for best outcome Depending on severity of irAEs, management may require corticosteroid or other immunosuppressive treatment and interruption or discontinuation of therapy If appropriate immunosuppressive treatment is used, patients generally recover from irAEs Use of immunosuppressive therapy to manage irAEs does not affect response to immunotherapy How should irAEs be diagnosed and managed? • Hold checkpoint inhibitor therapy for most grade 2 toxicities • Consider resuming immunotherapy when symptoms and/or laboratory values revert to grade 1 or lower • Corticosteroids (initial dose of 0.5-1 mg/kg/d of prednisone or equivalent) may be administered Grade 3 toxicities: • Hold checkpoint inhibitor therapy • Initiate high-dose corticosteroids (prednisone 1-2 mg/kg/d or methylprednisolone IV 1-2 mg/kg/d) • If symptoms do not improve with 48-72 hours of high-dose corticosteroids, infliximab may be offered for some toxicities • Taper corticosteroids over the course of at least 4-6 weeks • When symptoms and/or laboratory values revert to grade 1 or lower, rechallenging with immunotherapy may be offered; however, caution is advised, especially in those patients with early-onset irAEs. Dose adjustments are not recommended Grade 4 toxicities: • In general, permanent discontinuation of checkpoint inhibitor therapy is warranted, with the exception of endocrinopathies that have been controlled by hormone replacement Brahmer JR et al. Management of Immune-Related Adverse Events in PatientsTreated With Immune Checkpoint InhibitorTherapy: American Society of Clinical Oncology Clinical Practice Guideline. J Clin Oncol. 2018;36:1714-1768. For organ-specific assessment and management of irAEs, please see the ASCO guidelines: Additional resources available on the ASCO website: https://www.asco.org/practice-guidelines/quality-guidelines/guidelines/supportive-care-and-treatment-related-issues#/29866 Grade 2 Mild to moderate symptoms Severe or life-threatening symptoms Grades 3/4 Any organ system can be affected; commonly occurring are pulmonary (pneumonitis), dermatologic (rash, pruritus, blisters, ulcers, vitiligo), gastrointestinal (diarrhea, enterocolitis, transaminitis, hepatitis, pancreatitis), and endocrine (thyroiditis, hypophysitis, adrenal insufficiency) irAEs The precise pathophysiology is unknown, but translational studies have shown that T-cell, antibody, and cytokine responses may be involved Q Q Q A A A Pancreatitis, autoimmune diabetes Colitis Enteritis Encephalitis, aseptic meningitis Thyroiditis, hypothyroidism, hyperthyroidism Dry mouth, mucositis Hypophysitis Uveitis Pneumonitis Thrombocytopenia, anemia Hepatitis Adrenal insufficiency Nephritis Vasculitis Arthralgia Neuropathy Rash, vitiligo Myocarditis Increasing T-cell activity against antigens that are present in tumors and healthy tissue Activated T cell Antithyroid antibodies Increasing levels of inflammatory cytokines Increasing levels of pre-existing autoantibodies Enhancing complement-mediated inflammation due to direct binding of an anti–CTLA-4 antibody with CTLA-4 expressed on normal tissue Activated T cell Anti–CTLA-4 antibody CTLA-4 on pituitary Complement- mediated inflammation Cytokines Tumor with antigen and activated T cells Immune-Related Adverse Events (irAEs) Associated With Immune Checkpoint Inhibitors1, 2
- 4. Managing Adverse Events Associated With Systemic Therapies Used in Patients With Hepatocellular Carcinoma PRACTICE AID Guidelines for Hepatic irAE Management by Grade2 General Management q Monitor AST, ALT, and bilirubin prior to each infusion q Counsel patients q Rule out other causes Grade ICPi Therapy Monitor Corticosteroid Other 1 Asymptomatic (AST or ALT > ULN to 3.0 × ULN and/or total bilirubin > ULN to 1.5 × ULN) Continue Weekly or more No — 2 Asymptomatic (AST or ALT >3 to ≤5 × ULN and/or total bilirubin >1.5 to ≤3 × ULN) Hold Every 3 d 0.5-1 mg/kg/d Consider resuming ICPi when grade 1 or lower 3 Symptomatic liver dysfunction, fibrosis by biopsy, compensated cirrhosis, reactivation of chronic hepatitis (AST or ALT 5-20 × ULN and/or total bilirubin 3-10 × ULN) Discontinue permanently Every 1-2 d 1-2 mg/kg/d Taper at 4-6 wk If no improvement in 3 d, consider mycophenolate mofetil (infliximab not recommended) 4 Decompensated liver function (eg, ascites, coagulopathy, encephalopathy, coma; AST or ALT >20 × ULN and/or total bilirubin >10 × ULN) Discontinue permanently Daily; consider impatient 2 mg/kg/d Taper at 4-6 wk
- 5. Managing Adverse Events Associated With Systemic Therapies Used in Patients With Hepatocellular Carcinoma PRACTICE AID Management of AEs Associated With TKIs Symptoms Prophylaxis3-6 Onset Hand-Foot-Skin Reaction qq Erythema with or without blisters; hyperkeratotic lesions on palms and soles qq Commonly accompanied by dysesthesia (burning, pain, tingling) qq Perform full-body skin examination, focusing on deformities and hyperkeratotic areas on palms and soles, before treatment initiation qq Have patients remove their shoes and examine their feet during each visit qq Recommend podiatrist evaluation (can help with removal of calluses and hyperkeratotic regions) and orthotist evaluation and use of orthotic devices in patients with abnormal weight bearing qq During early therapy (2-4 wk), encourage rest and avoidance of vigorous exercise and traumatic activity qq Typically within 45 d of therapy initiation Grade7,a Characteristic4 Management3-6 1 Tingling, numbness, accompanied by minimal skin changes or dermatitis, such as erythema, edema, or hyperkeratosis of the hands and/or feet without pain; does not disrupt ADLs Avoid hot water, wear thick socks, wear cotton gloves/socks at night, use moisturizing creams and keratolytics (urea 20% to 40%; salicylic acid 5% to 10%); no dose reduction needed; follow up within 2 wk 2 Skin changes of the hands and/or feet, may include peeling, blisters, bleeding, edema, or hyperkeratosis with pain; discomfort affecting ADLs Employ grade 1 strategies, consider clobetasol 0.05% ointment 2x/d for erythematous areas, use topical and systemic analgesics (if no contraindications [eg, bleeding, kidney dysfunction]); consider 50% dose reduction for 7-28 d until HFSR is grade 1/0 → full dose 3 Severe skin changes of the hands and/or feet; may include peeling, blisters, bleeding, edema, or hyperkeratosis with pain and/or severe discomfort causing inability to work or perform ADLs Employ grade 1/2 strategies; treatment interruption for ≥7 d until HFSR is grade 1/0 → 50% of full dose → escalation, if possible; resume treatment at lower dose as recommended in package insert; dose may be escalated if reaction does not reoccur Moisturizer (daily), cold packs (indirectly) for 20 min/d; wear thick cotton gloves and socks, gently pat hands/feet dry after washing Hot water, direct sunlight, constrictive footwear, excessive friction, vigorous activity, and contact with cleaning products with strong chemicals Signs and symptoms immediately Calluses and hyperkeratotic regions REMOVE AVOID REPORTAPPLY RAAR6
- 6. Managing Adverse Events Associated With Systemic Therapies Used in Patients With Hepatocellular Carcinoma PRACTICE AID This Practice Aid has been provided as a quick reference to help learners apply the information to their daily practice and care of patients. a Images courtesy of Elizabeth Manchen, RN, MS, OCN. ADL: activities of daily living; ASCO: American Society of Clinical Oncology; CTLA-4: cytotoxic T-lymphocyte–associated protein 4; HFSR: hand-foot-skin reaction; ICPi: immune checkpoint inhibitor; irAE: immune-related adverse event; RAAR: remove, avoid, apply, and report; TKI: tyrosine kinase inhibitor; ULN: upper limit of normal. 1. Postow MA et al. N Engl J Med. 2018;378:158-168. 2. Brahmer JR et al. J Clin Oncol. 2018;36:1714-1768. 3. Lacouture ME et al. Oncologist. 2008;13:1001-1011. 4. McLellan B et al. Ann Oncol. 2015;26:2017-2026. 5. Brose MS et al. Semin Oncol. 2014;41(suppl 2):s1-s16. 6. Walko CM et al. Semin Oncol. 2014;41(suppl 2):s17-s28. 7. CTCAE version 5.0. https://ctep.cancer.gov/protocolDevelopment/electronic_applications/ ctc.htm#ctc_50. Accessed October 11, 2018. Access the activity,“Surveying the View From the Driver’s Seat in Hepatocellular Carcinoma: Bringing Into Focus Hepatology’s Key Role in Guiding HCC Care Down the Path to Improved Outcomes,”at www.peerview.com/WDR40. General Management Patient Education Medical Intervention Diarrhea5,6 Fatigue5,7 Patient should notify medical team of diarrhea or abdominal distress! Frequent, watery, bloody, or nocturnal stools Educating your patients on managing fatigue is essential! qq Monitor bowel habits, and report any increase in activity above normal qq Avoid spicy or fatty foods; plain, simple foods are best qq Avoid fruit and caffeine qq Maintain adequate fluid intake to avoid dehydration qq Monitor/manage electrolytes qq Staying as active as possible helps regulate sleep qq Maintain a normal work and social schedule qq Take breaks as needed qq Tell your medical team if activity is intolerable or fatigue worsens qq Loperamide is usually effective qq If loperamide is ineffective, consider diphenoxylate/atropine Management of AEs Associated With TKIs
