MichaelsLia_URS_Poster
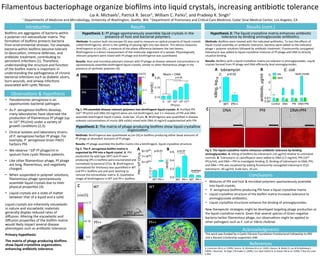
- 1. Mucin + DNA 0 107 108 109 1010 0.10 0.15 0.20 PFU/ml !sin(δ)! p<0.05 tobramycin WT Pf++ 0 1 2 killing,log10 p<0.02 WT Pf++ 0 1 2 3 5×1004 7×1004 9×1004 Pf4/104CFUs p<0.01 WT Pf++ WT Pf++ 0.0 0.2 0.4 0.6 !sin(δ)! washed p<0.01 A WT Pf++ WT Pf++ C 0.75 0.38 0.0 |sin(δ)| References 1. Costerton JW et al. (1999), Science. 2. Whiteley M et al. (2001), Nature. 3. Webb JS, Lau M & Kjelleberg S. (2004), J Bacteriol . 4. Dogic Z & Fraden S. (2006), Curr Opin Colloid In. 5. Glazer AM et al. (1996), P Roy Soc Lond a Mat. Filamentous bacteriophage organize biofilms into liquid crystals, increasing anTbioTc tolerance IntroducTon Lia A. Michaels1, Patrick R. Secor1, William C. Parks2, and Pradeep K. Singh1 1 Departments of Medicine and Microbiology, University of Washington, SeaYle, WA; 2 Department of Pulmonary and CriTcal Care Medicine, Cedar Sinai Medical Center, Los Angeles, CA Biofilms are aggregates of bacteria within a polymer-‐rich extracellular matrix. The formaTon of biofilms protects bacteria from environmental stresses. For example, bacteria within biofilms become tolerant to desiccaTon, immune defenses, and anTbioTc treatment, contribuTng to persistent infecTons (1). Therefore, understanding the structure and funcTon of the biofilm matrix is important in understanding the pathogenesis of chronic bacterial infecTons such as diabeTc ulcers, burn wounds, and airway infecTons associated with cysTc fibrosis . Fig 3. The liquid crystalline matrix enhances an=bio=c tolerance by binding aminoglycosides. A. Killing of biofilms by tobramycin (10 µg/ml) relaTve to untreated controls. B. Tobramycin or ciprofloxacin were added to DNA (2.5 mg/ml), Pf4 (1010 PFUs/ml), and DNA + Pf4 to invesTgate binding. C. Binding of tobramycin to DNA, Pf4, and DNA + Pf4 was visualized by adding fluorescently conjugated tobramycin (Cy5-‐ tobramycin, 40 µg/ml). Scale bars, 20 µm. Conclusions • Mixtures of Pf4 and host & microbial polymers spontaneously assemble into liquid crystals. • P. aeruginosa biofilms producing Pf4 have a liquid crystalline matrix. • Liquid crystalline structure of the biofilm matrix increases tolerance to aminoglycoside anTbioTcs. • Liquid crystalline structures enhance the binding of aminoglycosides. New therapeuTc strategies might be developed targeTng phage producTon or the liquid crystalline matrix. Given that several species of Gram-‐negaTve bacteria harbor filamentous phage, our observaTons might be applied to other pathogens such as E. coli or Vibrio cholerae. A Results (cont.) Acknowledgments This work was funded by a CysTc Fibrosis FoundaTon Postdoctoral Fellowship to PRS and a BasseY scholarship supported LAM. Fig 2. The P. aeruginosa biofilm matrix is organized by Pf4 into a liquid crystal. A. Pf4 producTon by wild type (WT) and Pf over-‐ producing (Pf++) biofilms were enumerated and normalized to bacterial CFUs. B. Birefringence (normalized for thickness) was quanTfied in WT and Pf++ biofilms pre and post washing to remove the extracellular matrix. C. QualitaTve image of birefringence in WT and Pf++ biofilms. Results ObservaTons & Hypothesis • Pseudomonas aeruginosa is an opportunisTc bacterial pathogen. • As P. aeruginosa biofilms develop, many laboratories have observed the producTon of filamentous Pf phage (up to 1011 PFU/ml) under a variety of growth condiTons (2,3). • Clinical isolates and laboratory strains of P. aeruginosa harbor Pf phage. For example, P. aeruginosa strain PAO1 harbors Pf4. • We observe ~108 Pf phage/ml in sputum from cysTc fibrosis paTents. • Like other filamentous phage, Pf phage are long, filamentous, and negaTvely charged. • When suspended in polymer soluTons, filamentous phage spontaneously assemble liquid crystals due to their physical properTes (4). • Liquid crystals are a state of maYer between that of a liquid and a solid. Hypothesis 1: Pf phage spontaneously assemble liquid crystals in the presence of host and bacterial polymers. Methods: A custom built microscope (5) was used to measure an opTcal property of liquid crystals called birefringence, which is the splijng of passing light into two beams. This device measures birefringence as|sin (δ)|, a measure of the phase difference between the two beams. Birefringence is a direct measurement of the molecular alignment of a sample. Physiologically relevant polymers were mixed with Pf phage and birefringence was quanTtated. Hypothesis 2: The matrix of phage producing biofilms show liquid crystalline organizaTon. Methods: Birefringence was quanTtated as|sin (δ)|in biofilms producing either basal amounts of Pf phage or abundant amounts of Pf phage. Results: Pf phage assemble the biofilm matrix into a birefringent, liquid crystalline structure. Hypothesis 3: The liquid crystalline matrix enhances anTbioTc tolerance by binding aminoglycoside anTbioTcs. Methods: Biofilms were treated with the indicated anTbioTcs. To test the effects of liquid crystal assembly on anTbioTc tolerance, bacteria were added to the indicated phage + polymer soluTons followed by anTbioTc treatment. Fluorescently conjugated tobramycin was added to liquid crystalline mixtures of Pf phage and DNA to directly visualize binding. A B Pf4 DNA 0.5 0.25 0.0 |sin(δ)| DNA +PF4 Fig 1. Pf4 assemble disease relevant polymers into birefringent liquid crystals. A. Purified Pf4 (1011 PFU/ml) and DNA (10 mg/ml) alone are not birefringent, but 1:1 mixtures of Pf4 and DNA assemble birefringent liquid crystals. Scale bar, 10 µm. B. Birefringence was quanTfied in disease relevant concentraTons of mucin (8% solids) mixed with DNA (4 mg/ml) supplemented with Pf4. E. coli Control DNA Pf4 DNA+Pf4 0.00 0.01 1 2 3 antibiotic,µg/ml tob cipro liquid crystal B DNA Pf4 DNA+Pf4 C Results: Host and microbial polymers interact with Pf phage at disease relevant concentraTons to spontaneously assemble birefringent liquid crystals, similar to other filamentous phage in the presence of syntheTc polymers (4). Liquid crystals are inherently viscoelasTc in nature and viscoelasTc materials generally display reduced rates of diffusion. Altering the viscoelasTc and diffusion properTes of the biofilm matrix would likely impact several disease phenotypes such as anTbioTc tolerance. Primary hypothesis: The matrix of phage producing biofilms show liquid crystalline organiza=on, enhancing an=bio=c tolerance. B 250 µm 250 µm Results: Biofilms with a liquid crystalline matrix are tolerant to aminoglycosides. Liquid crystals formed from Pf phage and DNA efficiently bind aminoglycosides.