Thesis Poster Presentation
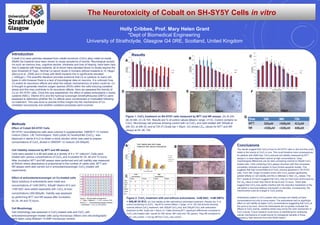
- 1. The Neurotoxicity of Cobalt on SH-SY5Y Cells in vitro Holly Cribbes, Prof. Mary Helen Grant *Dept of Biomedical Engineering University of Strathclyde, Glasgow G4 0RE, Scotland, United Kingdom Introduction Cobalt (Co) wear particles released from cobalt-chromium (CoCr) alloy metal-on-metal (MoM) hip implants have been shown to cause symptoms of toxicity. Neurological sympto ms such as memory loss, cognitive decline, blindness and loss of hearing, have been repo rted in patients with these implants, all of whom have elevated blood Co levels beyond the toxic threshold of 7µg/L. Normal Co serum levels in humans without implants is <0.19µg/L (Bocca et al., 2006) and in those with MoM implants this is significantly elevated ( >400µg/L). The scientific literature provides evidence that Co is cytotoxic to many cell types in vitro however there is a lack of toxicological data on neurons. It is unknown how Co exerts its neurotoxic effects and what the cellular mechanism(s) of action could be. Co is thought to generate reactive oxygen species (ROS) within the cells inducing oxidative stress and this may contribute to its neurotoxic effects. Here we assessed the toxicity of Co on SH-SY5Y cells. Once this was established, the effect of added antioxidants n-acetyl cysteine (NAC), Vitamin B12 and the hydroxyl scavenger dimethylthiourea (DMTU) were assessed to determine whether the Co effects were counteracted or modulated following co-treatment. This was done to provide further insight into the mechanisms of Co- mediated neurotoxicity and whether oxidative processes were involved. Conclusions The results suggest that CoCl2 is toxic to SH-SY5Y cells in vitro and this could relate to the toxicity of CoCl2 in vivo. This could therefore have consequences for patients with MoM hips. CoCl2 reduced cell viability measured by MTT assays in a dose-dependant manner at high concentrations. Clear morphological differences can be seen comparing controls to 500µM CoCl2 treated cells. Cells containing CoCl2 appear shrunken with their processes completely retracted and appear to have formed apoptotic bodies. This suggests that high concentrations of CoCl2 induces apoptosis in SH-SY5Y cells. From 48h, longer incubation times with CoCl2 caused significantly greater effects on cell viability and this is reflected in their LD50 values. The MTT results at 24 hours suggest that CoCl2 may be more toxic at this point as the LD50 value is lower than that at 48 hours and 72 hours. These data suggest that CoCl2 may rapidly interfere with the reductive metabolism of the cell before a neuronal defence mechanism is activated. Consequently, the mitochondria could be a target in CoCl2 toxicity. Antioxidants added to CoCl2 treated cells increase cell viability at lower concentrations but only to some extent. The antioxidants had no significant effect on cell viability at higher CoCl2 concentrations suggesting that CoCl2 at this point is too toxic. Since the antioxidants did not greatly modulate the effects of CoCl2 toxicity, oxidative stress may not be the main mechanism of CoCl2 toxicity. Future research could focus on identifying the underlying cellular mechanisms of cobalt toxicity for therapeutic benefits in those exposed to high amounts from their MoM implant. Figure 1: CoCl2 treatment on SH-SY5Y cells measured by MTT and NR assays. (A) At 24h. (B) At 48h. (C) At 72h. Results are % of control values (Mean+ range, n=12). Control contains no CoCl2. Microscopy cell pictures showing control with no CoCl2 and cells treated with 500µM at 24h (D), at 48h (E) and at 72h (F).Scale bar = 50µm. (G) shows LD50 values for MTT and NR assays at 24, 48, 72h Figure 2: CoCl2 treatment with and without antioxidants, 1mM NAC, 1mM DMTU + 500µM Vit B12. (A) Cell viability at 48h with/without antioxidant treatment. Results are % of control containing no CoCl2. *sig dif to control (Mean + range, n=12 (B) Cell pictures showing controls without CoCl2 treatment, with 300µM CoCl2 only, and 300µM CoCl2 with antioxidant treatment at 48h. Scale bar = 50µm (C) Table showing MTT significant differences compared to CoCl2 only treated cells. results for 24h (blue), 48h (red) and 72h (green). +sig diff compared to CoCl2 only control. – not sig diff from CoCl2 only control. Methods Effect of Cobalt SH-SY5Y Cells SH-SY5Y neuroblastoma cells were cultured in supplemented DMEM/ F-12 nutrient mixture (Gibco, Life Technologies). Solid cobalt (II) hexahydrate (CoCl2) was dissolved in sterile d-H2O to obtain a stock solution which was used to prepare concentrations of CoCl2 diluted in DMEM/F-12 medium (25-500µM). Cell Viability measured by MTT and NR assays Cells were seeded in a 96-well plate at a density of 5 x 104 cells/cm2. .Cells were treated with various concentrations of CoCl2 and incubated for 24, 48 and 72 hours. After incubation MTT and NR assays were performed and cell viability was measured at 540nm where absorbance is proportional to the number of viable cells. MTT and NR assays were also carried out in antioxidant/scavenger CoCl2-treated cell experiments. Effect of antioxidants/scavenger on Co-treated cells Stock solutions of antioxidants were made and concentrations of 1mM DMTU, 500µM Vitamin B12 and 1mM NAC were added separately with CoCl2 at toxic concentrations (300-600µM). Viability was assessed by performing MTT and NR assays after incubation for 24, 48 and 72 hours. Cell Morphology Cell morphology was assessed in CoCl2 treated cells and CoCl2 with antioxidant/scavenger treated cells using microscopy (Nikon) and cell photographs were taken using Moticam 10.0MP microscopy camera. Figure 1. 96 well plate arrangement X – Cells + Medium; X - CoCl2 controls (300- 600µM); Y – Antioxidant/scavenger controls; Y – Antioxidant + CoCl2 at 300-600µM A B CResults D E F G Cell Viability after 48h Cobalt treatment with/ without AntioxidantsA B C
