Synthesis report template 3 asperin
•Download as DOCX, PDF•
1 like•119 views
extended diploma synthesis report of asperin organic chemistry
Report
Share
Report
Share
Recommended
More Related Content
What's hot
What's hot (18)
The Analytical Methods And Technologies Of Cyanide Chemistry Training

The Analytical Methods And Technologies Of Cyanide Chemistry Training
Preparation of p-nitroacetanilide from Acetanilide

Preparation of p-nitroacetanilide from Acetanilide
Similar to Synthesis report template 3 asperin
Similar to Synthesis report template 3 asperin (20)
Synthesis, Characterization and Study of Antioxidant Activities of Some New P...

Synthesis, Characterization and Study of Antioxidant Activities of Some New P...
Synthesis and Biological Evaluation of Novel3, 5-Disubstituted 4h-1, 2, 4-Tri...

Synthesis and Biological Evaluation of Novel3, 5-Disubstituted 4h-1, 2, 4-Tri...
A green synthesis of isatoic anhydrides from isatins with urea–hydrogen perox...

A green synthesis of isatoic anhydrides from isatins with urea–hydrogen perox...
Synthesis, Identification and Surface Active Properties of Some Nonionic Surf...

Synthesis, Identification and Surface Active Properties of Some Nonionic Surf...
Synthesis, Characterization and invitro Anti- inflammatory activity of 1, 3, ...

Synthesis, Characterization and invitro Anti- inflammatory activity of 1, 3, ...
Oxidation of 7-Methyl Sulfanyl-5-Oxo-5H-Benzothiazolo-[3, 2-A]-Pyrimidine-6-...![Oxidation of 7-Methyl Sulfanyl-5-Oxo-5H-Benzothiazolo-[3, 2-A]-Pyrimidine-6-...](data:image/gif;base64,R0lGODlhAQABAIAAAAAAAP///yH5BAEAAAAALAAAAAABAAEAAAIBRAA7)
![Oxidation of 7-Methyl Sulfanyl-5-Oxo-5H-Benzothiazolo-[3, 2-A]-Pyrimidine-6-...](data:image/gif;base64,R0lGODlhAQABAIAAAAAAAP///yH5BAEAAAAALAAAAAABAAEAAAIBRAA7)
Oxidation of 7-Methyl Sulfanyl-5-Oxo-5H-Benzothiazolo-[3, 2-A]-Pyrimidine-6-...
Statistical Optimization of Synthetic Soda Ash for Water Softening

Statistical Optimization of Synthetic Soda Ash for Water Softening
Simple Synthesis of Some Novel Polyfunctionally Derivatives of 2H-Coumarin-2-...

Simple Synthesis of Some Novel Polyfunctionally Derivatives of 2H-Coumarin-2-...
Synthesis and Studies of Eco-friendly Acid Dye Metal Complexes and its Applic...

Synthesis and Studies of Eco-friendly Acid Dye Metal Complexes and its Applic...
Heterogeneous solid liquid catalysis of n-glycosylation by natural phosphate ...

Heterogeneous solid liquid catalysis of n-glycosylation by natural phosphate ...
A new approach for preparing methyl 6 azido-2,3,4-tri-o

A new approach for preparing methyl 6 azido-2,3,4-tri-o
A new approach for preparing methyl 6 azido-2,3,4-tri-o

A new approach for preparing methyl 6 azido-2,3,4-tri-o
Determination_of_Nitrogen_in_the_Wastewater_By_Kjeldahl_Method-1.pdf

Determination_of_Nitrogen_in_the_Wastewater_By_Kjeldahl_Method-1.pdf
Synthesis And Characterization of Novel Processable Poly (EtherAzomethine)S C...

Synthesis And Characterization of Novel Processable Poly (EtherAzomethine)S C...
Recently uploaded
9953056974 Young Call Girls In Mahavir enclave Indian Quality Escort service

9953056974 Young Call Girls In Mahavir enclave Indian Quality Escort service9953056974 Low Rate Call Girls In Saket, Delhi NCR
Recently uploaded (20)
STERILITY TESTING OF PHARMACEUTICALS ppt by DR.C.P.PRINCE

STERILITY TESTING OF PHARMACEUTICALS ppt by DR.C.P.PRINCE
Traditional Agroforestry System in India- Shifting Cultivation, Taungya, Home...

Traditional Agroforestry System in India- Shifting Cultivation, Taungya, Home...
Labelling Requirements and Label Claims for Dietary Supplements and Recommend...

Labelling Requirements and Label Claims for Dietary Supplements and Recommend...
Discovery of an Accretion Streamer and a Slow Wide-angle Outflow around FUOri...

Discovery of an Accretion Streamer and a Slow Wide-angle Outflow around FUOri...
Botany krishna series 2nd semester Only Mcq type questions

Botany krishna series 2nd semester Only Mcq type questions
TEST BANK For Radiologic Science for Technologists, 12th Edition by Stewart C...

TEST BANK For Radiologic Science for Technologists, 12th Edition by Stewart C...
Spermiogenesis or Spermateleosis or metamorphosis of spermatid

Spermiogenesis or Spermateleosis or metamorphosis of spermatid
9953056974 Young Call Girls In Mahavir enclave Indian Quality Escort service

9953056974 Young Call Girls In Mahavir enclave Indian Quality Escort service
Lucknow 💋 Russian Call Girls Lucknow Finest Escorts Service 8923113531 Availa...

Lucknow 💋 Russian Call Girls Lucknow Finest Escorts Service 8923113531 Availa...
CALL ON ➥8923113531 🔝Call Girls Kesar Bagh Lucknow best Night Fun service 🪡

CALL ON ➥8923113531 🔝Call Girls Kesar Bagh Lucknow best Night Fun service 🪡
Synthesis report template 3 asperin
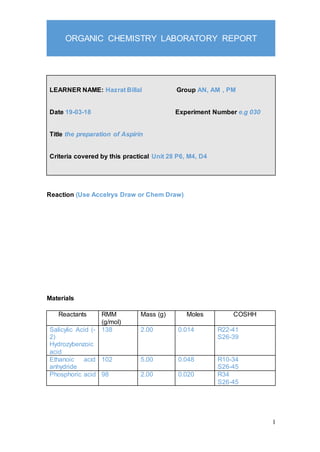
- 1. 1 ORGANIC CHEMISTRY LABORATORY REPORT LEARNER NAME: Hazrat Billal Group AN, AM , PM Date 19-03-18 Experiment Number e.g 030 Title the preparation of Aspirin Criteria covered by this practical Unit 28 P6, M4, D4 Reaction (Use Accelrys Draw or Chem Draw) Materials Reactants RMM (g/mol) Mass (g) Moles COSHH Salicylic Acid (- 2) Hydrozybenzoic acid 138 2.00 0.014 R22-41 S26-39 Ethanoic acid anhydride 102 5.00 0.048 R10-34 S26-45 Phosphoric acid 98 2.00 0.020 R34 S26-45
- 2. 2 ORGANIC CHEMISTRY LABORATORY REPORT Method Salicylic acids (2g) was added in a pear shape flask together with ethanoic anhydride (5cm3) and 5 drop of phosphoric acids were also added. Then the mixture was heated for 10 minutes then the ice was added and the mixture putted to water white solid was collected. The flask was placed in cold water bath to cool down and was left therefore, the crystal was able to form the crude crystals was poured through a Buckner funnel so it can be filtered, the dry aspirin was putted with (20ml) of boiling ethanol to dissolve the crystals 10ml of ethanol was added then the filtration process was done the precipitate was filtered to remove the impurities. Then it was putted into a beaker and a half of the volume of the filtered distilled water was added. It was then measured and recorded and left to be cooled down. Results One mole of SA gives 1 mole of ASA 2/138 moles of SA gives 2/138 moles Therefore, theoretical yield of ASA= 0.014 moles Mass/ RMM= Moles Mass = 0.0014×180= 2.6 Percentage yield A/T×100 2.14/2.16×100= 82% Percentage yield = 82 %