Thermodynamics III
- 1. THERMODYNAMICS - III Dr.P.GOVINDARAJ Associate Professor & Head , Department of Chemistry SAIVA BHANU KSHATRIYA COLLEGE ARUPPUKOTTAI - 626101 Virudhunagar District, Tamil Nadu, India
- 2. Third law of thermodynamics Determination of absolute entropy • At constant pressure equation (1) becomes dS = 𝑑𝑞 𝑇 -------(1) (𝜕S)p = 𝜕𝑞 𝑝 𝑇 -------(2) 𝜕𝑆 𝜕𝑇 p = 𝜕𝑞 𝜕𝑇 p x 1 𝑇 --------(3) At the absolute zero of temperature, the entropy of every substance may become zero and it does become zero in the case of a perfectly crystalline structure. Statement • The mathematical expression for change in entropy is • Divide on both side of equation (2) by dT
- 3. dS = Cp x 𝑑𝑇 𝑇 -------(5) 0 𝑆 𝑇 𝑑𝑆 = 0 𝑇 Cp 𝑑𝑇 𝑇 -------(6) ST = 0 𝑇 Cp d( ln 𝑇) -------(7) Where ST is the absolute entropy Third law of thermodynamics 𝜕𝑆 𝜕𝑇 p = Cp x 1 𝑇 -------(4) • Since 𝜕𝑞 𝜕𝑇 P is equal to Cp , the equation (3) becomes • Integrate equation (5) within the limits • Since d( ln 𝑇) is equal to 𝑑𝑇 𝑇 , the equation (6) becomes
- 4. Third law of thermodynamics • The area of the curve between T = 0 and any required temperature T gives the value of ST • The integral value 0 𝑇 Cp d( ln 𝑇) can be obtained by plotting Cp against ln T
- 5. Exceptions of Third law According to third law of thermodynamics the entropy of the crystalline solid is zero at 0 K. But for some gases like CO, NO and N2O, the entropies of their crystalline solid state are not zero at 0 K Explanation • These gases are having two alternative arrangements of molecule in their crystal lattices structure as shown as CO and OC NO and ON NNO and ONN • So that the crystals of these gases does not have ordered structure (or) perfect structure. It means that the entropies of the crystalline solids of these gases might not be zero at 0 K Third law of thermodynamics
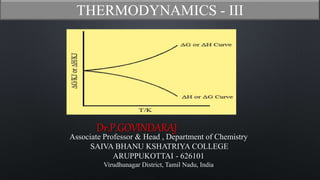
- 6. Third law of thermodynamics Statement The value of 𝜕(∆G) 𝜕𝑇 approaches zero gradually as the temperature is lowered towards the absolute zero and the value of ∆G approach close to the value of ∆H . This is graphically represented as Nernst Heat theorem Mathematically, Lt T=0 𝜕(∆G) 𝜕𝑇 = Lt T=0 𝜕(∆ 𝐻) 𝜕𝑇 = 0
- 7. Third law of thermodynamics • Gibbs Helmholtz equation is ∆G = ∆ H + T 𝜕(∆G) 𝜕𝑇 P ∆G - ∆H = T 𝜕(∆G) 𝜕𝑇 P -------(1) • At absolute zero temperature i.e., (T = 0 K), equation (1) becomes ∆G - ∆ H = 0 ∆G = ∆ H Explanation ∆G - ∆H = 0 x 𝜕(∆G) 𝜕𝑇 P
- 8. Significance of Nernst heat theorem 𝜕(∆G) 𝜕𝑇 P = - ∆S --------(1) 𝜕(∆ 𝐻) 𝜕𝑇 P = ∆Cp --------(2) • Where ∆S is the change of entropy for a reaction ∆Cp is the difference in the heat capacities of the product and reactant Third law of thermodynamics • From second law of thermodynamics, ∆S and ∆Cp for the reaction is expressed as • For a chemical reaction Reactant Product
- 9. • According to Nernst heat theorem Lt T = 0 𝜕(∆G) 𝜕𝑇 P = Lt T = 0 ∆S = 0 Lt T = 0 𝜕(∆ 𝐻) 𝜕𝑇 P = Lt T = 0 ∆Cp = 0 • i.e., Lt T = 0 ∆S = 0 Lt T = 0 ∆Cp = 0 • i.e., the entropy change of a reaction tends to approach zero and that the difference between the heat capacities of product and reactant tends to approach zero as the temperature is lowered towards the absolute zero Third law of thermodynamics
- 10. THANK YOU
